Translate this page into:
Phytochemical composition and therapeutic potential of Caralluma edulis a cholistani plant
⁎Corresponding authors at: Jiangxi Provincial Key Laboratory of Ex Situ Plant Conservation and Utilization, Lushan Botanical Garden, Jiangxi Province and Chinese Academy of Sciences, Jiujiang 332900, China (A.N. Shah). mirza.imran@iub.edu.pk (Mirza Imran Shahzad), ans.786@yahoo.com (Adnan Noor Shah)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Abstract
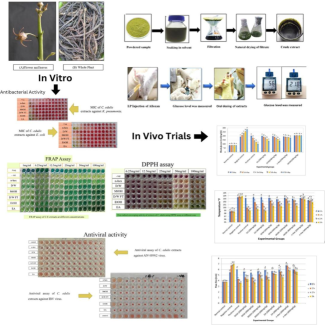
This study explores C. edulis, a plant indigenous to the Cholistan desert, locally known as Pimpa or Seetu, traditionally consumed as a vegetable. Our research aimed to comprehensively analyze its phytochemical constituents, and evaluate its antibacterial, antioxidant, antiviral, anti-inflammatory, antidiabetic, and antipyretic potentials. Utilizing a range of extracts including methanol (MtOH), ethanol (EtOH), ethyl acetate (EA), n-hexane (n-hex), dichloromethane (DCM), and aqueous (Aq), for effective extraction of phytochemicals from C. edulis. Standard biochemical assays and High-Performance Liquid Chromatography-Photodiode Array (HPLC-PDA) were used for analysis of phenolic compounds. Antibacterial effect(s) were confirmed through disc diffusion method and min inhibitory concentrations (MICs). The antioxidant activity was assessed through the Ferric Reducing Antioxidant Power (FRAP) assay and the DPPH radical scavenging method. In vivo antiviral potential was assessed through Hemagglutination (HA) test. Anti-inflammatory, antidiabetic, and antipyretic activities were performed on female albino rats using carrageenan, alloxan monohydrate and yeast-induced methods, respectively. Statistical analysis was done using standard one way ANOVA.Our findings revealed a rich diversity of phenolic compounds and the presence of proteins, alkaloids, and carbohydrates in C. edulis. MtOH and n-hex extracts demonstrated deep antiviral activity against various viral strains. In vivo toxicology studies indicated no significant toxicity at doses up to 5 g/kg. The DCM extract has shown notable anti-inflammatory effects, and EA extract was leading in antipyretic activity. All extracts, except MtOH, exhibited antidiabetic properties.In conclusion, C. edulis emerges not only as a valuable nutritional source but also as a potent alternative medicinal resource, offering wide range of therapeutic benefits.
Keywords
C. edulis
Cholistan
Antioxidant
Antidiabetic
Anti-corona virus
Anti-inflammatory
Antipyretic
1 Introduction
Caralluma belongs to Asclepiadaceous family and are being used as food in South East Asia (Mustafa et al., 2017). It is known for its therapeutic potential, especially against diabetes and gastrointestinal disease. C. edulis is commonly known as 'Settu or Pippu' (Ahmad et al., 2007), and its juicy stem can be used as a seasonal vegetable and its extracts have been used in the treatment of constipation, digestive problems, and obesity. Secondary metabolites not only protect plants against microorganisms, insects, and other potential predators but also act asa source of therapeutic agents. Plants comprise a variety of bioactive substances, such as alkaloids, tannins, glycosides, carotenoids, flavonoids, terpenoids, and steroids. These constituents have been reported for different properties e.g. antimicrobial and hemolytic activities. The HPLC fingerprints of standard phenolic compounds are used as qualitative and quantitative controls for the investigation of unknown compounds from plant samples (Mradu et al., 2012).
The number of chronic and stress-causing species is increasing in human body due to the disproportionate production of free radicals which are causing oxidative Biomolecule damage. The use of antioxidants can neutralize the oxidation procedures and delay oxidative stress. Plant based antioxidants are more useful on the basis of their availability, safety and efficacy (Shi et al., 2019). Similarly, to control viral threats, the poultry industry is using plant based therapeutics (Iqbal et al., 2024). Although control sheds and effective vaccination strategies have been introduced but viral outbreaks in poultry industry are still very common.
Diabetes mellitus is one of the most common metabolic disorders affecting billions of people globally. If it is not treated or controlled, it can cause other acute or chronic infections. Currently, it is considered the fifth leading cause of death. Although there has been significant progress in development of drugs and methods for the treatment of diabetes, but results are still not very much promising. These treatments have many disadvantages associated likedrug resistance, toxicityetc.(Kooti et al., 2016 & Ashraf et al., 2023). Numerous plant-based drugs have been used to treat diabetes and proved as an effectivesource of hypoglycemic drugs.
Medicinal plants possess many anti-inflammatory phytochemicals such as phenolics, alkaloids, polysaccharides, terpenoids, fatty acids, etc. that can inhibit/decrease inflammation. There have been different mechanisms reported through which these anti-inflammatory responses are studied (Oguntibeju, 2018& Mubashir et al., 2022).Pyrexia is defined as an increase in body temperature that can be caused by various reasons including infections, inflammations and other diseased conditions, which enhance the production of chemical mediators such as cytokines. High fever is often associated with other health complications such as dehydration, increased tissue catabolism, and other ailments (Estella et al., 2022).
This study aimed to evaluate the in vitro antibacterial, antioxidant, and antiviral activities, along with the in vivo anti-inflammatory, antipyretic, and antidiabetic effects of different extracts from C. edulis.
2 Experimental procedures
2.1 Sample collection
Caralluma edulis was collected as a whole plant from the Cholistan desert, Bahawalpur, Pakistan. The plant is characterized by its thick, succulent, cylindrical stems often with grayish or greenish n color. Typically, it possesses reduced leaves and small, aromatic greenish-yellow flowers arranged in clusters. Its fruit is fleshy and elongated. Plant identification was done by a taxonomist at the Department of Botany, The Islamia University of Bahawalpur (voucher no. 561).
2.2 Extract preparation
Extracts were prepared by the method provided by (Shah et al., 2018) with little modification. Methanol (MtOH), Ethanol (EtOH), Distilled water (Aq), Aqueous freeze–thaw (Aq F/T) Ethyl acetate(EA),n-hexane (n-hex), and Dichloromethane (DCM) were used as solvents. To prepare the Aq freeze–thaw (Aq F/T) group, plant material was soaked in distilled water in a sealed container. After three days in the freezer, the material thawed in a water bath, and the resulting solution was filtered and dried. This method, involving a unique aqueous freeze–thaw process, facilitated the extraction of bioactive compounds for a thorough exploration of the plant material's chemical composition. The plant was washed, dried at RT for 20 days, and ground into fine powder. 10 g of dried plant was soaked into 100 mL of respective solvent. The mixtures were kept in airtight containers, over a period of 3 days. The filtrates were dried using a rotary evaporator and stored for future use.
2.3 Preliminary phytochemical screening
The extracts were analyzed for basic phytochemical screening using standard biochemical tests. (Shaikh and Patil, 2020). Proteins, alkaloids, carbohydrates, flavonoids, saponins, glycosides, steroids, terpenoids, and phenols were analyzed. These phytochemicals have important medicinal properties such as alkaloids exhibits anti-inflammatory and analgesic properties, phenols and flavonoids are known for their antioxidant properties. Saponins and steroids possess anti-inflammatory and immune modulatory effects. Glycosides Present in C. edulis can exhibit various biological activities, including potential antidiabetic effects. Terpenoids are recognized as plant’s aromatic profile and may possess antimicrobial and anti-inflammatory activities.
2.4 Phytochemical analysis
To assess the presence of various phytochemicals in crude extracts of Caralluma edulis, the following tests were conducted:
Protein Test: Million’s reagent was added to the extracts. The formation of a white precipitate confirmed the presence of proteins.
Alkaloids Test: Wagner’s reagent was used; a color change indicated the presence of alkaloids. Flavonoids Test: Molish reagent and sulfuric acid were added to the extracts. The appearance of a violet ring confirmed the presence of flavonoids. Additionally, a mixture of acetic acid and 2 % NaOH showed an intense yellow color that disappeared upon the addition of the crude extract. Saponins Test: The presence of saponins was confirmed by vigorous shaking of the extracts with water, resulting in stable foam formation.
Glycosides Test: Liebermann’s test was performed by adding chloroform, acetic acid, and concentrated sulfuric acid to the extracts. A color shift to violet, blue, and green confirmed the presence of glycosides.
Steroids and Terpenoids Test: Salkowski’s test involved adding extracts to chloroform and sulfuric acid, with a red color appearing in the lower layer confirming the presence of these compounds.
Phenols Test: The Ferric chloride test used 2 % FeCl3 in the extracts, with a color change from blue to black confirming the presence of phenols.
2.5 HPLC-PDA analysis
2.5.1 Chemicals and solvents
All chemical standards, including gallic acid, catechin, chlorogenic acid, p–OH benzoic acid, vanillic acid, epicatechin, syringic acid, 3–OH benzoic acid, 3–OH–4–MeO benzaldehyde, p–coumaric acid, rutin, sinapinic acid, t–ferulic acid, naringin, 2,3–diMeO benzoic acid, benzoic acid, o–coumaric acid, quercetin, harpagoside, t–cinnamic acid, naringenin, and carvacrol from Sigma Aldrich (Milan, Italy). Methanol and acetonitrile (HPLC–grade), as well as acetic acid (99 %), were sourced from Carlo Erba Reagent (Milan, Italy). Dimethyl sulfoxide (DMSO) was obtained from Honeywell (Japan). Milli–Q water was produced using a Millipore Milli–Q Plus water treatment system (Millipore Bedford Corp., Bedford, MA, USA).
2.5.2 Sample preparation
Samples for HPLC-PDA analysis were prepared as follows: the plant extracts were weighed using an analytical balance and then dissolved in mobile phase A (Milli-Q water + 3 % acetic acid) and mobile phase B (acetonitrile + 3 % acetic acid) in a ratio of 93:7 (v:v), adding 20 % dimethyl sulfoxide (DMSO), except for the aqueous plant extracts, which were perfectly soluble in only mobile phase. The chemical samples were prepared at a concentration of 1 mg per 250 µL. Each sample underwent vortexing for half a minute, followed by a 15-minute sonication process. Subsequently, 20 µL of each prepared sample was injected into the HPLC system for analysis.
2.5.3 HPLC conditions
HPLC analyses were carried out using a validated method from our previous research (Di Sotto et al., 2018) on a Waters liquid chromatograph. This system included a model 600 solvent pump and a 2996 photodiode array detector (PDA), with data acquisition managed through Empower v.2 Software (Waters Spa, Milford, MA, USA). The separation was achieved using a C18 reversed-phase packing column (Prodigy ODS (3), 4.6 × 150 mm, 5 μm; Phenomenex, Torrance, CA, USA), maintained at a temperature of 30 ± 1 °C with the aid of a Jetstream2 Plus column oven. UV/Vis data were collected across a wavelength range of 200 to 500 nm. Quantitative analyses were performed at the respective max wavelengths of each compound, using an injection volume of 20 μL. The mobile phase consisted of water-acetonitrile (93:7, v:v, 3 % acetic acid), which was subjected to on-line degassing via a Biotech DEGASi, model Compact (LabService, Anzola dell’Emilia, Italy). In supplementary material S1 was reported the chemical standards chromatograms (at 275 nm, wavelength where all chemicals are visible) coupled with a table that reports all the analytes with the corresponding retention times (min) and wavelengths (nm) used for quantitative analyses, while gradient elution details are provided in supplementary material S2.
2.5.4 Antibacterial assay
Seven bacterial ATCC strains includingE. coli, P. vulgaris, P. aeruginosa, K. pneumoniaea,S. aureus, MDR P. aeruginosa MDR S. aureuswere used in the assay(Sofi et al., 2016).
2.5.5 Disc diffusion method
The antibacterial activity of the plant extracts was evaluated using the disc diffusion method. Extracts were prepared in their respective solvents at a concentration of 0.5 g/mL (Behravan et al., 2019). Pre-soaked GF-1 grade filter paper discs were employed for the assay. Bacterial inocula were spread on agar plates, which were then incubated for 40 min at 37 °C before the discs were placed. The cultures were subsequently incubated overnight at 37 °C, and the zone of inhibition (ZoI) was measured according to the standard procedures outlined by CLSI guidelines. Neat solvents served as controls. Ampicillin and moxifloxacin were utilized as positive controls for normal and drug-resistant bacterial strains, respectively. The min inhibitory concentration (MIC) was determined following the standardized methods described by (Elisha et al., 2017 & Ren et al., 2005).
2.6 Antioxidant assays
2.6.1 Ferric Reducing antioxidant power (FRAP)
FRAP assay was performed. An ELISA reader took the OD700. The assay was performed in triplicate (Chaves et al., 2020).
2.6.2 Free radicle scavenging activity using 2, 2-diphenyl-1-picryl-hydrazyl (DPPH)
The free radical scavenging was done using DPPH assay(Noreen et al., 2017). An ELISA reader took the OD517. The assay was performed in triplicate. Percentage %RSA was calculated by using the given formula:
IC50 was calculated by graph pad prism.
2.6.3 Antiviral activity
Antiviral activity was evaluated against the Infectious Bronchitis Virus (IBV) and the H9N2 strain of Avian Influenza Virus (AIV). For viral inoculations, 9–11 days old chicken embryonated eggs were taken. Viral strains were injected through the chronio-allantoic route under sterile conditions. Eggs were incubated at 37 °C for 48 h, after which the allantoic fluids were collected.
2.6.4 Heamagglutination (HA) titer
The standard HA test was carried out asthe method given by (Hussain et al., 2003).
2.6.4.1 IC50
The IC50 of each active extract was calculated through the serial dilution method.The dose and time-dependent curves were calculated using linear regression using the Easy Fit program.
2.6.4.2 In vivo studies
Healthy albino Female rats weighing between 150 and 250 g were obtained from animal rearing facility, Faculty of Pharmacy, The Islamia University of Bahawalpur. Following OECD guidelines, all animals were housed in standard lab settings with a 12-hour light/dark cycle, a 35–60 % humidity range, and free access to standard food and water, at the Department of Food Sciences, Faculty of Agriculture and Environment, The Islamia University of Bahawalpur, Pakistan. All the trials were completed with the guiding principle of the Institutional Animal Ethics Committee (IAEC), The Islamia University of Bahawalpur, Pakistan.
2.6.4.3 Acute toxicity study
For acute toxicity test, six groups of three animals each (n = 3) were used and crude extract of C. edulis at increasing dosages of 200, 500, 1000, 2000, 3000, and 5000 mg/kg respectively were given orally. Toxicity signs and behavioral changes such as hyperactivity, convulsions, corneal reflex, sweating, alertness, urination, and mortality in all groups were observed for 24 h under standard environmental conditions, with free excess of food and water (Asif et al.,2014).
2.6.4.4 Anti-inflammatory assay
The animals were divided into 11 groups of five animals in each group (n = 5). By injecting carrageenan (1 % w/v in 0.9 % w/v NaCl, 0.1 mL) into the rat's right hind paw, acute inflammation was triggered. Group, I was the normal control group (Normal saline 5 mL/kg), and groups II was positive control (Standard dose of Diclofenac sodium 15 mg/kg) and group III was negative control (Only carrageenan induced). The remaining groups were treated with plant extracts. The rats' right-hand paw sizes were measured using Vernier caliper. After 30 min of carrageenan administration, extracts were given orally. The standard drug Diclofenac sodium injection was administered intraperitoneally. The paw sizes were measured at regular intervals until 4 h (Jisha et al., 2019).
2.6.4.5 Antipyretic activity (Yeast induced pyrexia)
Different crude extracts were used to evaluate antipyretic potential by yeast-induced pyrexia method. The basal rectal temperature was measured before and after treatment. 10 % Brewer's yeast solution was subcutaneously injected into rats. The animals were separated into 11 groups, and each group had 5 animals (n = 5). Group I was normal control, group II was –ve control (normal saline), group III was +ve control (paracetamol), groups IV & V were treated with MtOH extracts 200 and 400 mg/kg doses, respectively, groups VII &VII treated with EA extracts 200 and 400 mg/kg respectively and group X& XI were treated with n-Hex extracts with different doses such as 200 and 400 mg/kg respectively. Rectal temperatures were measured at regular intervals of 1–4 h (Abdel-Aleem et al., 2019).
2.6.4.6 Alloxan-induced anti-diabetic activity
Diabetes was induced intraperitoneally by giving Alloxan monohydrate injection in dose dependent manner (120 mg per kg of body weight). The diabetic rats (>150 mg/dL) were divided into 7 groups including positive (standard drug Glibenclamide 5 mg/kg) and negative control groups. In each group, five animals (n = 5) were placed. For hyperglycemic evaluation, blood glucose levels were tested every three days for up to one week by glucometer. As per the schedule, the animals were fasted for the entire night in order to get blood samples (Raju et al., 2016).
2.7 Statistical analysis
All data were presented as mean ± Standard Error of Mean (SEM). The statistical package for social sciences (SPSS) was used to perform One Way Analysis of Variance (ANOVA) for comparing the means among various groups. A p-value of less than 0.05 was considered statistically significant, indicating that the observed differences among the group means were unlikely to have occurred by random chance. This approach using ANOVA ensures a robust and valid assessment of the data, especially in determining overall differences across the groups under study. Graphical illustration was carried out using Microsoft excel, 2016.
3 Results
3.1 Phytochemical screening
This screening indicated the presence of proteins, alkaloids, carbohydrates, saponins, glycosides, steroids, terpenoids, phenols, and flavonoids. The extraction of phytochemicals depended on both the polarity of the solvent and the nature of the phytochemicals (Nawaz et al., 2020).
Table 1. Detected = (+), not detected = (−).
Biochemical tests
Extracts
n-hex
Aqueous
MtOH
EtOH
EA
Proteins
−
−
+
−
−
Alkaloids
−
−
+
+
−
Carbohydrates
+
−
+
+
+
Flavonoids
−
+
−
−
−
Saponins
−
−
+
+
−
Glycosides
−
−
+
+
+
Steroids
−
−
+
+
+
Terpenoids
−
−
+
+
+
Phenols
−
+
+
+
−
3.2 HPLC-PDA determination and phenols chemical profiles
Results show that different extracts of C. edulis contain different phenolic compounds. The phenolic compound found in max quantity was Chlorogenic acid (0.85 µg/mg) in EA extract (Fig. 1c). Different concentrations of phenolic compounds were found in other extracts except for n-Hex. Some extracts contain different phytochemicals that were below the limit of quantification of assay. The overall trend was EtOH > EA > MtOH > Aqueous F/T > Aqueous.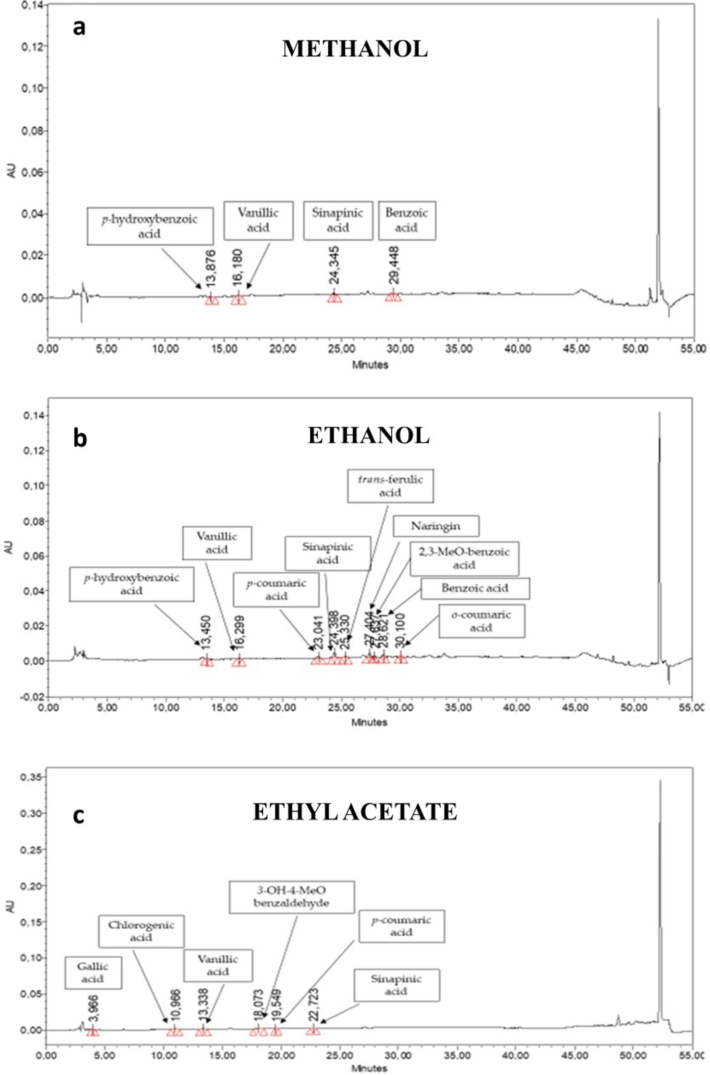
HPLC-PDA determination and phenols chemical profiles.
Table 2 and Fig. 1 a,b,c.
Phenols
n-hex
Aqueous
MeOH
AqueousF/T
EtOH
EA
Gallic acid
BLQ
Chlorogenic acid
0.32 (± 0.02)
0.85(± 0.06)
p-OH benzoic acid
BLQ
BLQ
BLQ (>LOD)
BLQ
Vanillic acid
BLQ
BLQ
3-OH-4-MeO benzaldehide
BLQ
p-coumaric acid
BLQ
BLQ
Sinapinic acid
BLQ
BLQ
BLQ
t-ferulic acid
BLQ
Naringin
0.38 (± 0.03)
2,3-diMeO benzoic acid
0.34(± 0.02)
Benzoic acid
1.74 (± 0.10)
0.32 (± 0.02)
o-coumaric acid
BLQ
3.3 Antibacterial activity
Different extracts ofC. edulis have shown good antibacterial activities against different strains. EtOH extract have shown max ZoI in case of K. pneumoniaea, P. aeruginosa, E. coli, andMDR P. aeruginosa.n-Hex extract heave showed max ZoI against S. aureus and P. vulgaris and other studies have reported similar results (Shailemo et al., 2016). Similarly, EA extract shows max antibacterial potential against MDR S. aureus.MIC of each active extract was calculated in many extracts were found effective against bacteria in low concentrations as well.
Table 3.
Bacterial strains
Extract type
Avg ZoI(mm) ± SEM
MIC (µg/µl)
K. pneumoniaea
n-hex
7.0000 ± 0.8
3125
Aqueous
7.5000 ± 0.5
−
MtOH
9.0000 ± 0.8
12500
Aqueous F/T
7.5000 ± 0.5
−
EtOH
16.0000 ± 1
6250
EA
9.0000 ± 0
3125
Ampicillin
16.0000 ± 0
781.25
P. vulgaris
n-hex
9.5000 ± 0.5
12500
Aqueous
8.0000 ± 0
−
MtOH
9.0000 ± 0.8
12500
Aqueous FT
7.5000 ± 0.5
−
EtOH
8.5000 ± 1
12500
EA
8.5000 ± 0.5
6250
Ampicillin
15.0000 ± 0.8
390.625
S. aureus
n-hex
10.0000 ± 0
3125
Aqueous
9.0000 ± 0
6250
MtOH
8.5000 ± 0.5
3125
Aqueous FT
9.0000 ± 0
6250
EtOH
7.0000 ± 1
1562.5
EA
7.5000 ± 0.5
3125
Ampicillin
10.0000 ± 0
3125
P. aeruginosa
n-hex
7.5000 ± 0.5
12500
Aqueous
8.0000 ± 0
−
MtOH
9.0000 ± 0
12500
Aqueous FT
8.5000 ± 0.5
−
EtOH
14.5000 ± 1
3125
EA
8.0000 ± 0.8
12500
Ampicillin
12.5000 ± 0.5
3125
E. coli
n-hex
7 ± 0
3125
Aqueous
7.5 ± 0.5
−
MtOH
7 ± 0
6250
Aqueous FT
6.5 ± 0.5
−
EtOH
11 ± 1
6250
EA
7.5 ± 0.5
6250
Ampicillin
15 ± 0
3125
MDR P. aeruginosa
n-hex
7.5000 ± 0.5
12500
Aqueous
8.0000 ± 0
−
MtOH
8.5000 ± 0.5
6250
Aqueous FT
8.5000 ± 0.5
−
EtOH
13.5000 ± 0.5
6250
EA
10.0000 ± 0
6250
Moxifloxacin
11.5000 ± 1
1562.5
MDR S. aureus
n-hex
7.5000 ± 0.5
−
Aqueous
7.0000 ± 0
−
MtOH
8.5000 ± 0.5
6250
Aqueous FT
7.0000 ± 0
−
EtOH
8.0000 ± 0.8
12500
EA
11.0000 ± 0.8
6250
Moxifloxacin
15.0000 ± 0
1562.5
3.4 Antioxidant assay
The antioxidant potential of C. edulis extracts was checked by FRAP and DPPH assay. In the case of FRAP, the yellow color of negative control changed into blue or pale green depending upon the quantity of antioxidants in plant extracts. Max potential was shown by EA and EtOH extracts. The overall order of activity was EA > EtOH > MtOH > n-Hex > Aq. In the case of the DPPH assay, almost the same trend was seen. Max antioxidant power was shown by EA extract with IC50 1202.4. The overall order of activity in terms of IC50 was EA > MtOH > Aqueous > EtOH > AqueousF/T.
Figs. 2 and 3.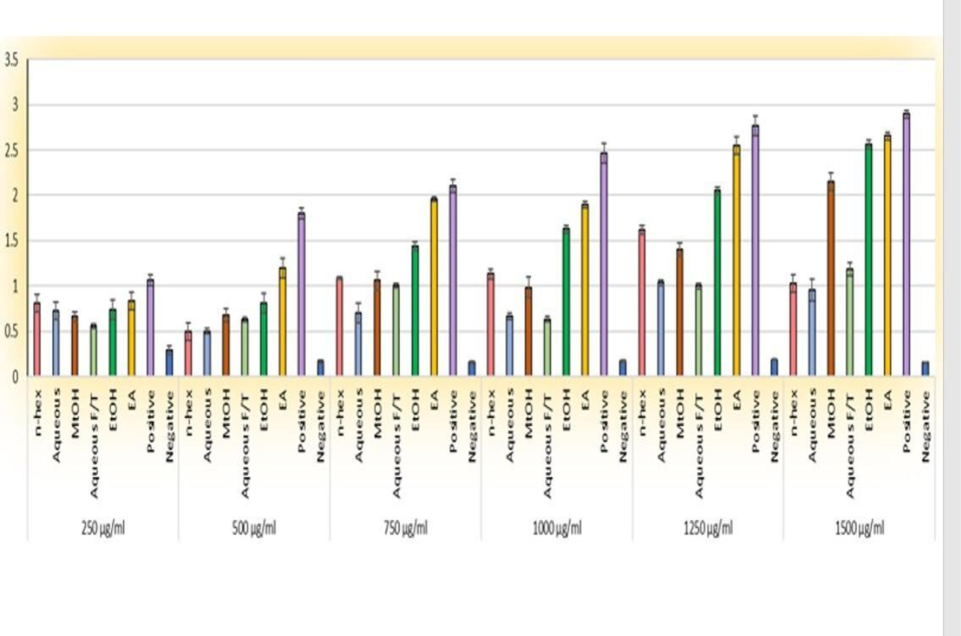
Anti-oxidant assay of C. edulis extract at different concentrations FRAP assay (Absorbance at 700 nm). The value represents mean ± standard error from at least 3 replicates.
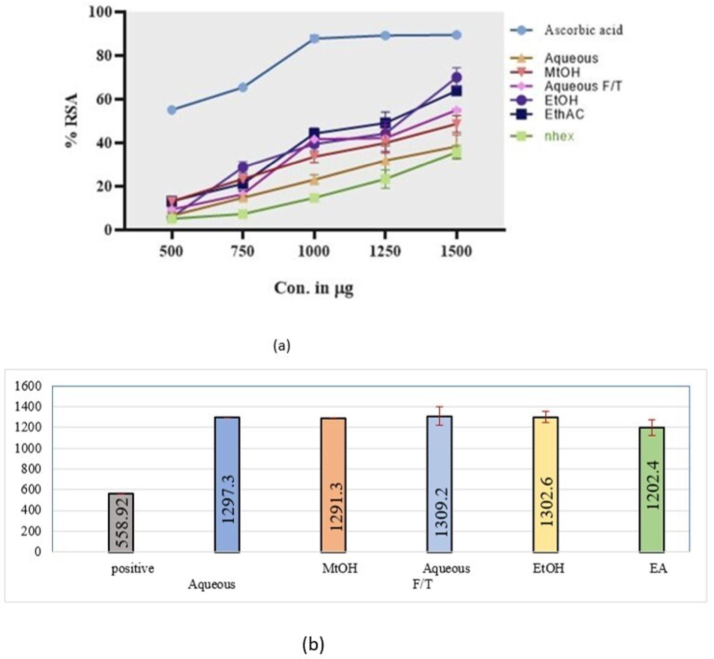
DPPH radical scavenging activity a) %RSA and b) IC50 of all extracts of C. edulis. All experiments were performed in triplicate. Data are expressed as mean ± SD (n = 3).
3.5 Antiviral activity
C. edulis extracts were tested against two most common poultry viruses i.e., AIV-H9N2 and IBV. Almost all extracts have shown good antiviral potential against these strains. The max anti-AIV H9N2 activity was shown by n-Hex and EtOH extracts with HA titers 0 (10 log reduction = 100 %). The overall order of activity was n-Hex = EtOH > MtOH = Aq > F/T > Aqueous > EA. In the case of IBV, all extracts of C. edulis were positive but in varying order. MtOH, Aqueous F/T, and EA extracts have shown max antiviral potentials with HA titer 0 (10 log reduction = 100 %). IC50 of each positive extract was calculated. The overall order of activity was MtOH = AqueousF/T = EA > EtOH = Aqueous > n-Hex.
Table 4. Log reduction = [(log2 control = 10) −(log2 sample)].
Plant extracts
H9N2
IBV
HA Titer
Titer in log2
Log Reduction
IC50
HA Titer
Titer in log2
Log Reduction
IC50
n-hex
0
<log 2
10
6.25
4
2log2
8
25
Aqueous
4
2log2
8
25
2
log2
9
3.125
MtOH
2
log2
9
12.5
0
<log2
10
6.25
AqueousF/T
2
log2
9
3.125
0
<log2
10
3.15
EtOH
0
<log2
10
3.125
2
log2
9
25
EA
8
3log2
7
25
0
<log2
10
12.5
3.6 Acute toxicity
For the acute toxicity study, the crude extracts of C. edulis were administered to female albino rats (n = 3) at doses of 200,500,2000,3000, and 5000 mg/kg BW. These dosages were chosen based on previous research efficacy and OECD guidelines (Jonsson et al., 2013). After administration of doses, animals were monitored closely over the next 14 days for any behavioral changes such as goosebumps, convulsions, corneal reflex, sweating, allergic reaction, hyperactivity, and mortality. No major complication or mortality was observed in the acute toxicity study, up to the 5000 mg/kg dosage level. At dose levels 3000 and 5000 mg/kg, mild changes in goosebumps were observed. So, it is concluded that C. edulis is safe to use up to 5000 mg/kg.
3.7 Anti-inflammatory
Among all extracts, DCM extracts exhibited excellent results at both doses of 200 and 400 mg/kg, comparable to the positive control groups, respectively. In contrast, the n-Hex extract demonstrated the least activity. All the extracts displayed dose-dependent activity. To further reinforce our findings, statistical analysis was conducted, indicating significant differences in the efficacy of DCM extracts compared to the positive control groups at both doses (p < 0.05). Additionally, statistical significance details highlight the lower activity observed in the n-Hex extract (p < 0.05).
The overall trend of different extracts, supported by statistical measures, was as follows: DCM > MtOH > EA > n-Hex.
Fig. 4.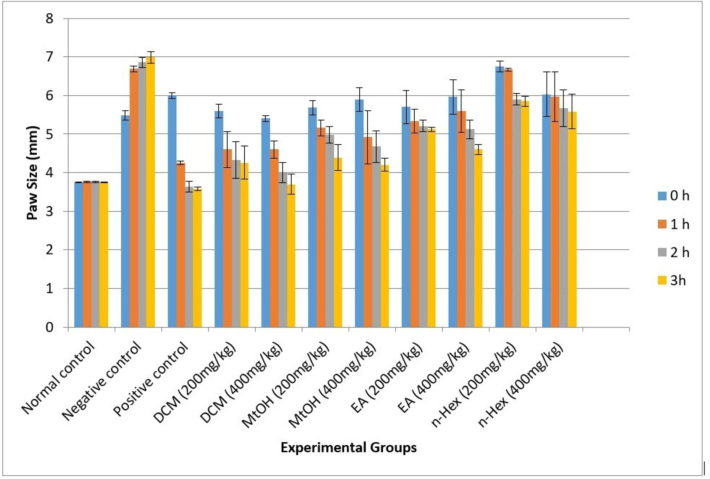
Anti-inflammatory activity of C. edulis extracts. Each value represents as mean SEM (n = 5). p-value < 0.05 was considered statistically significant.
3.8 Antipyretic activity
The EA extract of C. edulis exhibited excellent antipyretic potential, demonstrating significant results after 1 h that were sustained up to 3 h. Max efficacy was observed at the 3-hour mark. Doses, 200 and 400 mg/kg of EA, displayed high effectiveness compared to the Positive control group (Paracetamol, 150 mg/kg), with statistical analysis confirming the significance of these results (p < 0.05). The observed significant differences underscore the robust antipyretic activity of the EA extract.
Furthermore, the calculated p-values provide a quantitative measure of the observed differences, reinforcing the reliability of our findings. The overall trend of antipyretic activity observed was as follows: EA > DCM > MtOH > n-Hex.
Fig. 5.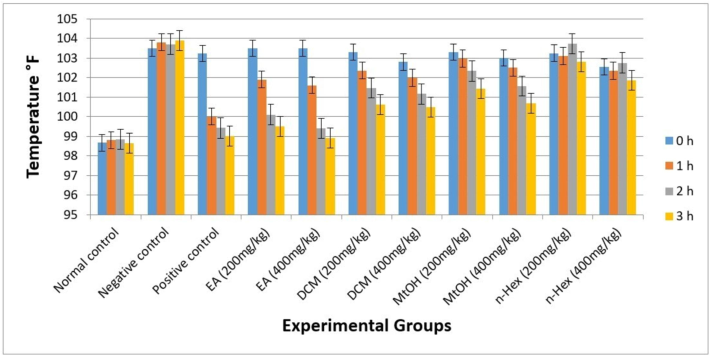
Anti-pyretic activity of C. edulis extracts. Each value represents as mean SEM (n = 5). p-value < 0.05 was considered statistically significant.
3.9 Anti-diabetic activity
The EA extract showed highly significant antidiabetic activity compared to DCM and MtOH extracts. MtOH extract demonstrated non-significant results on the 1st, 3rd, and 5th days when compared to the Positive control Group; however, a slightly positive response was observed on the 7th day. A comprehensive statistical analysis, with calculated p-values, has been conducted, revealing significant differences among the groups. Specifically, the significant changes on certain days and between groups have been identified, with p-values providing a quantitative measure of the observed differences. The overall trend for antidiabetic activity was EA > DCM > n-Hex > MtOH, comparable to the positive control group.
Fig. 6.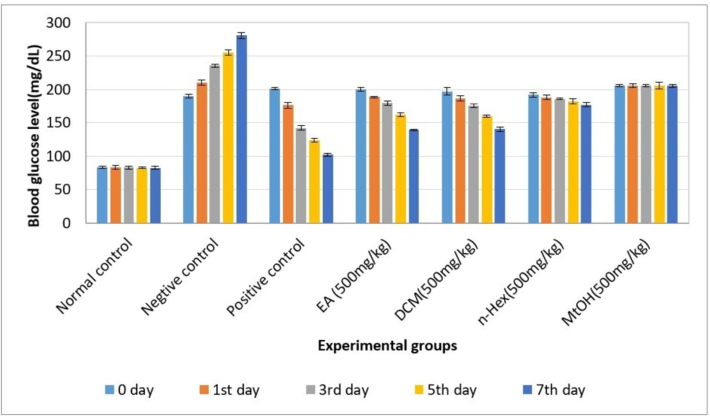
Antidiabetic activity of C. edulis extracts. Each value represents as mean SEM (n = 5). p-value < 0.05 was considered statistically significant.
4 Discussion
Medicinal plants have been used for centuries to treat various diseases and health conditions. Many of these plants contain compounds that have pharmacological activity and can be used to develop drugs for the treatment of various illnesses. The misuse of antibiotics have led to the development of antibiotic resistance in bacteria. Medicinal plants provide an alternative pathway to combat antibiotic resistance (Duțu et al., 2022). In the current investigation, EtOH extract has shown max antibacterial activity against various strains. HPLC-PDA analysis has confirmed the presence of reported antibacterial agents like Benzoic acid, Naringin, 2,3-diMeO Benzoic acid,etc. in EtOH extracts(Adamczak et al., 2020). The reason for the good antibacterial potential of EA extract is the presence of chlorogenic acid which is already reported as a good antibacterial agent (Cai et al., 2019). Studies have confirmed the presence of phytochemicals is directly proportional to the antioxidant potentials of extracts (Borges et al., 2020).As EA extract contains chlorogenic acid that’s why this extract shows max potential. The overall trend was EA > EtOH > MtOH > n-hex > D/W FT > D/W.
Antiviral assay was performed against AIV-H9N2 and IBV virus. Results revealed that extracts of C. edulis had very good antiviral activity. The order of activity against AIV-H9N2 virus was n-hex > EtOH > MtOH > D/W > D/W FT > EA and order of same activity against IBV was MtOH > D/W FT > EA > D/W > EtOH > n-hex. Similar studies were reported from 11 Cholistani plants and their antiviral potential was checked against AIV-H9N2, NDV, IBV and Infectious Bursal Disease (IBD) viruses. Different extracts of these plants were prepared and tested against viruses. The medicinal plants as O. compressa, N. procumbens, and S. surattense had max antiviral potential among tested plants. In short, these studies have demonstrated the value of cholistani plants as a source of antiviral drugs (Shahzad et al., 2020).
Medicinal plants show excellent anti-inflammatory activity due to the presence of secondary metabolites such as phenols, flavonoids, alkaloids, saponins, etc. They may cause a variety of proinflammatory mediators such as mast cells, macrophages, lymphocytes, and neutrophils. Secondary metabolites may be responsible for inhibiting proinflammatory enzymes (COX, PLA2) (Wang et al., 2013). The phytochemical screening of C. edulisalso indicated the existence of saponins, tannins, phenol, alkaloids, terpenoids, and flavonoids. Based on these findings, these phytochemicals may be responsible for exerting their anti-inflammatory effect via the inhibitory mechanism of prostaglandins synthesis via COX enzyme inhibition (Minhas et al., 2018). DCM extract of C. edulis showed the highest anti-inflammatory results given that secondary metabolites are present such as flavonoids that can cause inhibition of COX and 5LOX pathways resulting in a decrease in eicosanoid synthesis. They may also reduce the production of proinflammatory mediators responsible for the inflammatory process (Abdel-Aleem et al., 2019).
The onset of fever in antipyretic activity is triggered by inflammatory mediators such as cytokines released by mononuclear phagocytes as a defense of the immune system. These cytokines also interact with specific receptors and stimulate the release of pyrogenic mediators. The cytokines that promote fever are transported from blood to the brain through specific carriers. In the brain, cyclooxygenase (COX)-2 produces prostaglandins E2(PGE2), a mediator of fever. Prostaglandins act on thermo sensitive neurons of the hypothalamus (Ravishankar et al., 2010). The MtOH, EA, and DCM extracts of C. edulis showed significant antipyretic potential compared to control groups and significantly decreased rectal temperature. Based on these results, it can be suggested that it may due to inhibition of prostaglandin synthesis. And the phytochemical analysis of C. edulis revealed that it has β-sitosterol, glycosides, steroidal glycosides, and flavonoids that may take part to control fever (Aslam et al., 2019& Khani et al., 2023).
Various researchers have identified flavonoids, sterols, alkaloids, and polyphenols as bioactive agents with antidiabetic properties. C. edulis is known to contain saponins, alkaloids, tannins, phenols, glycosides, terpenoids, and flavonoids, all of which exhibit antidiabetic effects. Notably, saponins have been reported to possess hypoglycemic activity. EA extract has dominated in both in vitro antidiabetic (α-glucosidase inhibitory assay) and in vivo antidiabetic activity due to the presence of the phytochemicals which have hypoglycemic effects (Ezejiofor et al., 2013& Ali et al., 2023).
5 Conclusions
In conclusion, C. edulis (Seetu) exhibits a rich profile of antimicrobial, antibiofilm, antioxidant, antiviral, anti-inflammatory, anti-pyretic, and anti-diabetic agents, positioning it as both a valuable vegetable and a source of potent pharmacological compounds. Our in vivo trials on rats highlight the need to transition from preclinical studies to human clinical trials. Our findings reveal a clear correlation between the research aim and the observed results. This research direction would not only enhance our knowledge of the plant's bioactive constituents but also pave the way for the development of targeted interventions or pharmaceutical applications.
CRediT authorship contribution statement
Bushra Gillani: Writing – original draft, Investigation, Data curation, Conceptualization. Sarah Tariq: Resources, Methodology, Formal analysis. Mirza Imran Shahzad: Writing – review & editing, Project administration, Methodology, Conceptualization. Tatheer Fatima: Writing – review & editing, Investigation, Formal analysis. Marcello Locatelli: Writing – review & editing, Software, Investigation. Xinxia Cai: Writing – review & editing, Visualization, Software. Adnan Noor Shah: Writing – review & editing, Resources, Data curation. Ajaz Ahmad: Writing – review & editing, Funding acquisition.
Acknowledgments
The researchers also express their sincere appreciation to the Researchers Supporting Project Number (RSP2024R350) at King Saud University, Riyadh, Saudi Arabia.
We are thankful to Dr Zulfiqar Ahmad, Chairman Department of Food Sciences and Technology, The Islamia University of Bahawalpur.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Total phenolic and flavonoid contents and antioxidant, anti-inflammatory, analgesic, antipyretic and antidiabetic activities of Cordia myxa L. leaves. Clin. Phytosci.. 2019;5(1):29.
- [CrossRef] [Google Scholar]
- Total phenolic and flavonoid contents and antioxidant, anti-inflammatory, analgesic, antipyretic and antidiabetic activities of Cordia myxa L. leaves. Clin. Phytosci.. 2019;5(1):1-9.
- [Google Scholar]
- Antibacterial activity of some flavonoids and organic acids widely distributed in plants. J. Clin. Med.. 2020;9(1):109.
- [Google Scholar]
- GIS, GPS and remote sensing application to investigate agricultural potential in Cholistan. Sociedade & Natureza.. 2007;19(1):55-64.
- [Google Scholar]
- Exploring the potential of next generation sequencing in plant breeding and genetics. Agrobiol. Rec. 2023:1-5.
- [Google Scholar]
- Time and concentration-dependent differential antioxidant potential in the gum of medicinally important Araucaria heterophylla. Agrobiol. Rec. 2023:44-52.
- [Google Scholar]
- Diuretic activity of achyranthes aspera linn crude aqueous extract in albino rats. Trop. J. Pharm. Res.. 2014;13(12):2039-2045.
- [Google Scholar]
- Microscopic investigations and pharmacognostic techniques for the standardization of Caralluma edulis (Edgew.) Benth. Ex Hook. F. Microscopy Research and Technique.. 2019;82(11):1891-1902.
- [Google Scholar]
- Facile green synthesis of silver nanoparticles using Berberis vulgaris leaf and root aqueous extract and its antibacterial activity. Int. J. Biol. Macromol.. 2019;124:148-154.
- [Google Scholar]
- Comparison of techniques and solvents on the antimicrobial and antioxidant potential of extracts from acacia dealbata and olea europaea. Antibiotics. 2020;9(2):48.
- [Google Scholar]
- Antibacterial activity and mechanism of cinnamic acid and chlorogenic acid against Alicyclobacillus acidoterrestris vegetative cells in apple juice. Int. J. Food Sci. Technol.. 2019;54(5):1697-1705.
- [Google Scholar]
- Quantification of the antioxidant activity of plant extracts: Analysis of sensitivity and hierarchization based on the method used. Antioxidants.. 2020;9(1):76.
- [Google Scholar]
- Antiviral and antioxidant activity of a hydroalcoholic extract from humulus lupulus L. Oxid. Med. Cell. Longev.. 2018;2018:5919237
- [Google Scholar]
- Dutu LE, Popescu ML, Purdel CN, Ilie EI, Luță E-A, Costea L, Gîrd CEJD. Traditional medicinal plants—a possible source of antibacterial activity on respiratory diseases induced by chlamydia pneumoniae, haemophilus influenzae, klebsiella pneumoniae and moraxella catarrhalis. 2022;14(2):145.
- The antibacterial activity of extracts of nine plant species with good activity against Escherichia coli against five other bacteria and cytotoxicity of extracts. BMC Complement. Altern. Med.. 2017;17(1):1-10.
- [Google Scholar]
- Evaluation of the analgesic and antipyretic activity of methanol extract of Combretum bauchiense Hutch & Dalziel (Combretaceae) leaves. Phytomed. Plus. 2022;2(1):100166
- [Google Scholar]
- Hypoglycaemic and tissue-protective effects of the aqueous extract of Persea americana seeds on alloxan-induced albino rats. Malaysian J. Med. Sci.: MJMS.. 2013;20(5):31.
- [Google Scholar]
- Detection of serum antibody levels against infectious bursal disease (IBD) virus using indirect hemagglutination (IHA) test in commercial broilers. Int. J. Poult. Sci.. 2003;2(6):442-445.
- [Google Scholar]
- A narrative review on effective use of medicinal plants for the treatment of parasitic foodborne diseases. Agrobiol. Rec.. 2024;16:79-92.
- [Google Scholar]
- Anti-inflammatory efficacy of methanolic extract of Muntingia calabura L. leaves in Carrageenan induced paw edema model. Pathophysiology. 2019;26(3–4):323-330.
- [Google Scholar]
- Application of OECD Guideline 423 in assessing the acute oral toxicity of moniliformin. Food Chem. Toxicol.. 2013;53:27-32.
- [Google Scholar]
- Impact of dietary cation-anion difference on rumen fermentation, digestibility, and blood parameters in Zandi lambs under heat stress. Agrobiol. Rec. 2023:60-67.
- [Google Scholar]
- The role of medicinal plants in the treatment of diabetes: a systematic review. Electron. Physician. 2016;8(1):1832.
- [Google Scholar]
- Anti-inflammatory effect of Caralluma edulis against acute and chronic inflammation. JAPS: Journal of Animal & Plant Sciences. 2018;28(2)
- [Google Scholar]
- HPLC profiles of standard phenolic compounds present in medicinal plants. Int. J. Pharma. Phytochem. Res.. 2012;4(3):162-167.
- [Google Scholar]
- Common medicinal plants effective in peptic ulcer treatment: a nutritional review.International. J. Agri. Biosci.. 2022;11(2):70-74.
- [Google Scholar]
- Mustafa G, Arif R, Atta A, Sharif S, Jamil A. Bioactive compounds from medicinal plants and their importance in drug discovery in Pakistan. 2017.
- Effect of solvent polarity on extraction yield and antioxidant properties of phytochemicals from bean (Phaseolus vulgaris) seeds. Braz. J. Pharm. Sci.. 2020;56
- [Google Scholar]
- Measurement of total phenolic content and antioxidant activity of aerial parts of medicinal plant Coronopus didymus. Asian Pac. J. Trop. Med.. 2017;10(8):792-801.
- [Google Scholar]
- Medicinal plants with anti-inflammatory activities from selected countries and regions of Africa. J. Inflamm. Res.. 2018;11:307.
- [Google Scholar]
- In vivo animal model for screening of anti diabetic activity. Biosci. Biotechnol. Res. Asia. 2016;9(2)
- [Google Scholar]
- Antipyretic activity of Guduchi Ghrita formulations in albino rats. AYU: International Quarterly Journal of Research in Ayurveda.. 2010;31(3)
- [Google Scholar]
- Differential gene expression for investigation of Escherichia coli biofilm inhibition by plant extract ursolic acid. Appl. Environ. Microbiol.. 2005;71(7):4022-4034.
- [Google Scholar]
- Evaluation of antioxidant and antibacterial activities of the stems of Flammulina velutipes and Hypsizygus tessellatus (white and brown var.) extracted with different solvents. J. Food Meas. Charact.. 2018;12(3):1947-1961.
- [Google Scholar]
- Antiviral activities of Cholistani plants against common poultry viruses. Trop. Biomed.. 2020;37(4):1129-1140.
- [Google Scholar]
- Qualitative tests for preliminary phytochemical screening: an overview. Int. J. Chem. Stud.. 2020;8(2):603-608.
- [Google Scholar]
- Antibacterial activity of Moringa ovalifolia and Moringa oleifera methanol, N-hexane and water seeds and bark extracts against pathogens that are implicated in water borne diseases. Green Sustain. Chem.. 2016;6(02):71.
- [Google Scholar]
- Online extraction–HPLC–FRAP system for direct identification of antioxidants from solid Du-zhong brick tea. Food Chem.. 2019;288:215-220.
- [Google Scholar]
- Antioxidant and antimicrobial properties of grape and papaya seed extracts and their application on the preservation of Indian mackerel (Rastrelliger kanagurta) during ice storage. J. Food Sci. Technol.. 2016;53(1):104-117.
- [Google Scholar]
- Naturally derived anti-inflammatory compounds from Chinese medicinal plants. J. Ethnopharmacol.. 2013;146(1):9-39.
- [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.jksus.2024.103519.
Appendix A
Supplementary material
The following are the Supplementary data to this article:







