Translate this page into:
Nano-herbal combination targets apoptosis in oral squamous cell carcinoma
⁎Corresponding author at: Department of Biology, Faculty of Mathematics and Natural Science, Universitas Sumatera Utara, Medan 20155, Indonesia. syafruddin6@usu.ac.id (Syafruddin Ilyas)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Background
This study evaluates the synergistic effects of the nano-herbal combination of Bischofia javanica leaves and Phaleria macrocarpa fruits on oral squamous cell carcinoma (OSCC), a metastatic keratinocyte malignancy.
Materials and Methods
Thirty-six male Wistar rats were divided into six groups: a negative control group (K0), an OSCC-induced control group (K1), and four treatment groups (P1-P4), which received oral treatments for four weeks. Blood samples were analyzed for superoxide dismutase (SOD), malondialdehyde (MDA), and vascular endothelial growth factor (VEGF) levels, while immunohistochemistry was used to assess the expression of heat shock protein 70 (HSP-70), caspase-3, caspase-8, caspase-10, and MDA.
Results
Nano-herbal treatment significantly reduced MDA levels, particularly in groups P3 and P4, with reductions comparable to those seen in the Vitamin C treatment group (p < 0.05). SOD levels were highest in group P3 (p < 0.05), similar to the negative control group (K0), while VEGF levels were significantly elevated in the positive control group (K1) (p < 0.05). Group P3 exhibited the highest expression of HSP-70 (p < 0.05). Caspase-3 expression was significantly higher in P3 compared to K1 (p = 0.0007), while caspase-8 and caspase-10 expressions were also significantly elevated in groups P2, P3, and P4 (p < 0.05). Group P3 showed the most pronounced effects in promoting apoptosis, with caspase-3 and caspase-8 levels markedly higher compared to K1.
Conclusion
The combined nano-herbal formulations of Bischofia javanica leaves and Phaleria macrocarpa fruits effectively mitigates oxidative stress, induces apoptosis, and holds potential as a therapeutic approach for managing oral squamous cell carcinoma (OSCC).
Keywords
OSCC
MDA
SOD
VEGF
HSP-70. Apoptosis
Caspase-3
Caspase-8
Caspase-10
Nano-herbal Combination
- OSCC
-
oral squamous cell carcinoma
- CMC-Na
-
Natrium Carboxymethylcellulose
- ELISA
-
Enzyme-linked immunosorbent assay
- IOA
-
Interobserver Agreement
- IRS
-
Immunoreactive Score
- K0
-
Group of negative controls
- K1
-
Group of OSCC-induced controls
- P1
-
Group of OSCC treated with nano-herbal formulations derived from Bischofia javanica leaves
- P2
-
Group of OSCC treated with nano-herbal formulations derived from Phaleria macrocarpa fruit
- P3
-
Group of OSCC treated with the combined nano-herbal formulations of Bischofia javanica leaves and Phaleria macrocarpa fruits
- P4
-
Group of OSCC treated with Vitamin C
- Ns
-
Not significant
- SOD
-
superoxide dismutase
- MDA
-
malondialdehyde
- VEGF
-
vascular endothelial growth factor
- HSP-70
-
heat shock protein 70
- AhR
-
aryl hydrocarbon receptor
- HEM
-
High Energy Milling
- BaP
-
Benzo(alpha)pyrene
Abbreviations
1 Introduction
Oral squamous cell carcinoma (OSCC) is a malignancy originating from keratinocyte cells, which can metastasize through lymphatic or hematogenous pathways. The development of OSCC is driven by a complex interplay between genetic predispositions and environmental carcinogens, particularly the use of tobacco, consumption of alcohol, and the practice of betel quid (areca nut) chewing (WHO, 2023). In North America and Europe, the incidence of OSCC is increasing among younger populations due to human papillomavirus (HPV) infections (WHO, 2023). Data from the Global Cancer Observatory in 2020 indicate that OSCC accounts for 94 % of all oral cavity malignancies globally, with regional prevalence ranging from 3-6 % in Western countries to as high as 30 % in Eastern countries, including Indonesia, where It is ranked as the eighth most prevalent cancer globally (GLOBOCAN, 2020).
A significant challenge in treating OSCC is its resistance to conventional therapies, which often fail to adequately induce apoptosis, a critical mechanism for controlling cancer progression. This underscores the need for novel treatments that effectively stimulate apoptosis in OSCC cells. One notable environmental risk factor for OSCC is exposure to Benzo(alpha)pyrene (BaP), a polycyclic aromatic hydrocarbon present in cigarette smoke, air pollution, and smoked foods (Jiang et al., 2019). BaP induces oxidative stress and DNA mutations, promoting carcinogenesis by interacting with the aryl hydrocarbon receptor (AhR), increasing cytochrome P450 enzymes, and elevating free radical production. The oxidative damage caused by BaP leads to dysregulation of key tumor suppressor genes, such as P53 and BRCA, thereby facilitating OSCC progression (Poetsch, 2020).
Previous studies have investigated various natural compounds possessing antioxidant and anticancer properties, including those derived from traditional herbal medicines. However, while some research has indicated the potential of plant-based treatments to promote apoptosis in cancer cells, the integration of various herbal extracts is utilized —particularly in nano-formulations—remains underexplored. This study hypothesizes that the combined nano-herbal formulations of Bischofia javanica leaves and Phaleria macrocarpa fruits will demonstrate greater effectiveness in inducing apoptosis and reducing oxidative stress in OSCC compared to individual treatments. An essential component of this study involves the conversion of these herbal into nanoparticle formulations. Nanoparticles offer several advantages in drug delivery and therapeutic efficacy, particularly in the context of cancer treatment. The diminutive size and elevated surface area-to-volume ratio significantly improve bioavailability and stability of active compounds, improve cellular uptake, and facilitate targeted delivery to cancerous tissues (Elumalai et al., 2024; Rumahorbo et al., 2024a; Simanjuntak and Rumahorbo, 2024). Additionally, nanoparticle formulations can enhance the solubility of hydrophobic substances present in Bischofia javanica and Phaleria macrocarpa, facilitating enhanced penetration through biological membranes. This nanoscale delivery system ensures that bioactive compounds effectively reach the tumor microenvironment, thereby augmenting their ability to induce apoptosis and reduce oxidative stress in OSCC cells. Consequently, the incorporation of these plant extracts into nano-formulations may significantly enhance their therapeutic efficacy compared to conventional herbal treatments.
Although Bischofia javanica and Phaleria macrocarpa have been studied separately for their anticancer properties, limited research has explored their combined synergistic effects, particularly in nano-formulations. Bischofia javanica contains bioactive compounds, such as betulinic acid, friedelin, and quercetin, which demonstrate anticancer activity across various cell lines, including those associated with oral cancer (Lee et al., 2021; Shen et al., 2017). In contrast, Phaleria macrocarpa is recognized for its lignans and flavonoids, including mangiferin, which exhibit both antioxidant and anticancer properties (Christina et al., 2022; Maharani et al., 2021). Given the diverse bioactive compounds present in these plants, combination therapy in nano-formulations may provide a more targeted and potent approach to the treatment of OSCC.
This study addresses the shortcomings of previous research, which often focuses on individual herbal treatments while neglecting the potential of combinatory nano-herbal therapies. By examining the combined effects of both plants, we aim to investigate whether this combination can enhance apoptosis through the caspase pathway, mitigate oxidative stress, and improve therapeutic outcomes for patients with OSCC. Practically, our findings could advance therapeutic strategies for the treatment of OSCC by integrating nano-herbal formulations into clinical practice. The potential of both plants to function as combined therapies may result in more effective treatment modalities that address the limitations of conventional cancer therapies.
Research Objective Questions:
-
How do the combined nano-herbal formulations of both plants influence apoptosis in OSCC cells compared to individual treatments?
-
What effects do these nano-herbal formulations exert on oxidative stress markers, including MDA, SOD, VEGF, and HSP-70 in OSCC?
-
In what ways does the formulation of these herbal extracts into nanoparticles enhance their therapeutic efficacy against OSCC?
2 Materials and Methods
2.1 Reagents
The antibodies utilized for immunohistochemistry included the following: Monoclonal Antibody HSP-70 (Tyr41) Polyclonal Antibody PE Conjugated (Biossusa; Catalog Number: BS-10451R-PE), Caspase-8 Polyclonal Antibody (Elabscience; Catalog Number: E-AB-62025), Caspase-10 Polyclonal Antibody (Elabscience; Catalog Number: E-AB-13820), Caspase-3 Polyclonal Antibody (Elabscience; Catalog Number: E-AB-63510), and Rabbit Polyclonal Antibody for Malondialdehyde (MDA) (BioAssay; Catalog Number: 357267–96 T). The antibodies that were used for ELISA assays included the Malondialdehyde (MDA) BioAssay™ ELISA Kit (Universal) (BT LAB; Catalog Number: SH0020), Superoxide Dismutase (SOD) ELISA Kit (BioSource; Catalog Number: MBS705758), and Rat VEGF ELISA Kit (Catalog Number: ab100786).
2.2 Preparation of the combined nano-herbal formulations of Bischofia javanica and Phaleria macrocarpa
The primary materials utilized in this study are the leaves of Bischofia javanica and the berries of Phaleria macrocarpa, which were purposively obtained from Simalungun Regency, North Sumatra, without comparison to similar plants from other regions. The mature dark green leaves of Bischofia javanica and the mature red fruits of Phaleria macrocarpa were collected from a local community garden, where both plants grow wild and are not cultivated by the local community. The leaves were cleaned and dried at ambient temperature for around seven days, while the flesh of Phaleria macrocarpa was separated from its seeds, washed, thinly sliced, and dried for approximately fourteen days. Both materials were subsequently ground separately and processed into nano-formulations.
Nano-herbal formulations were created utilizing High Energy Milling (HEM) at NanoTech Indonesia in Tangerang. The HEM apparatus employed stainless steel milling balls with diameters of 10 mm and 20 mm, with the larger balls placed in the milling jar first, followed by the smaller ones, ensuring that the total weight of the powdered samples did not exceed two-thirds of the jar's volume. The powdered of both plants, which were pre-processed in a blender, were added separately. No solvents or coolants were utilized to preserve the bioactive compounds. The milling process commenced at 350 rpm, consisting of intermittent phases: three hours of grinding, a one-hour pause, six hours of grinding, another one-hour pause, and a final nine-hour grinding phase, resulting in nanopowders. The same procedure was applied to a one-to-one weight combination of both materials, utilizing two kilograms of each dried substance, in accordance with protocols established in previous research (Rumahorbo et al., 2024b).
2.3 Subjects and study design
This pre-clinical study utilized Rattus norvegicus as the animal model and received approval from the Animal Research Ethics Committee of the Faculty of Mathematics and Natural Sciences at Universitas Sumatera Utara (AREC) (Submission Number: 0455/KEPH-FMIPA/2023). Thirty-six male Wistar rats, each weighing between 180 and 250 g, were divided into six groups of six rats each and acclimatized for one week. One group, consisting of six rats, served as the negative control (K0) without induction of oral squamous cell carcinoma (OSCC). Specific conditions were maintained to prevent the development of OSCC in the negative control group (K0). The rats were maintained under standard laboratory conditions, including a 12-hour light/dark cycle, temperatures ranging from 22 to 24 °C, and humidity levels between 40 and 60 %. They received a standard rodent diet that was free of carcinogens and had ad libitum access to water. The environment was regularly cleaned to minimize stress and contamination.
In contrast, thirty rats were induced with OSCC by injecting 0.04 mg/0.04 ml of benzo[a]pyrene in corn oil into the oral mucosal tissue three times each week throughout a four-week interval. This dosage was based on prior studies that indicated its effectiveness in inducing carcinogenic effects (Bukowska et al., 2021; Yang et al., 2023). Following a swab test and Papanicolaou (PAP) smear, the OSCC-positive rats were organized into five groups: K1 (positive control), P1 (800 mg/kg body weight nano-herbal from Bischofia javanica), P2 (500 mg/kg body weight nano-herbal from Phaleria macrocarpa), P3 (650 mg/kg body weight nano-herbal combination), and P4 (40 mg/kg body weight vitamin C). The vitamin C dosage of 40 mg/kg body weight was based on prior studies in humans and animal models, then adjusted for the rats in this study (Böttger et al., 2021; Zasowska-Nowak et al., 2021). The dosage determination was based on previous studies that examined the toxicity effects of these two plants, including cytotoxicity tests, acute toxicity tests, chronic toxicity tests, and phytochemical characterization analyses of the two nano-herbals. These studies can be accessed in (Simanjuntak and Rumahorbo, 2022; Rumahorbo et al., 2023a–c). Treatments were administered daily via oral gavage in a 0.3 % CMC-Na solution for four weeks, with dosages determined based on previous toxicity studies. Blood samples were collected to assess superoxide dismutase (SOD), malondialdehyde (MDA), and vascular endothelial growth factor (VEGF) levels using ELISA, while oral mucosal tissue was processed for immunohistochemistry.
HSP-70, malondialdehyde (MDA), Caspase-3, Caspase-8, and Caspase-10 expression were analyzed by counting positive cells using the Per Hundred Cells method at 400x magnification. This method quantifies the percentage of cells expressing specific proteins within a sample of one hundred cells in a defined area. Two observers evaluated the positive cells with Interobserver Agreement (IOA), where an agreement of 70–80 % was considered acceptable; values below 70 % were subject to further review. Each group was assessed nine times using the Immunoreactive Score (IRS) system (Table 1).
Frequency of Appearance (A)
Intensity of Staining (B)
IRS Score
0 = Not Visible
0 = negative
0–1 = negative
1= <10 % positive
1 = weak
2–3 = weak
2 = 10–50 % positive
2 = moderate
4–8 = moderate
3 = 51–80 % positive
3 = strong
9–12 = strong
4= >80 % positive
IRS Score (A x B): 0–12
2.4 Statistical analysis of data
The data were initially presented as both the mean and standard deviation and subsequently analyzed using ANOVA, followed by Duncan's post-hoc test. For non-parametric data, the Kruskal-Wallis test was applied, with the Mann-Whitney test utilized for follow-up comparisons. Statistical significance was indicated by asterisks in the tables: *p < 0.05, **p < 0.01, ***p < 0.001, and ****p < 0.0001, while “ns” denoted not significant (p > 0.05). The testing process involved formulating hypotheses, presenting data, calculating test statistics, and drawing conclusions.
3 Results
3.1 The presence of MDA within the blood serum and the oral cavity mucosa
Statistical analysis revealed significant differences in Malondialdehyde (MDA) levels among the groups (p < 0.05, F = 0.000). Groups K1 and P2 exhibited the highest MDA levels, indicating increased oxidative stress, while group K0 (normal control) presented the lowest levels. This observation aligns with the understanding that elevated MDA levels result from lipid peroxidation due to oxidative stress in malignant tissues (Maurya et al., 2021). The reduction in MDA levels in groups P1 and P3, which were treated with nano-herbals combination of both plantas, signifies strong antioxidant effects that effectively mitigate oxidative damage. The efficacy of the combination treatment was comparable to that of Vitamin C, underscoring the potential of these nano-herbals in cancer therapy.
The Kruskal-Wallis test revealed significant differences among the groups (p < 0.05, F = 0.0029). Mann-Whitney tests indicated significant reductions in MDA levels, particularly between K1 (OSCC) and P1 (Bischofia javanica), with P4 (Vitamin C) demonstrating the largest difference from K1. This observation suggests that Vitamin C is effective but may be less impactful than the synergistic effects of the nano-herbals (Table 2). A significant difference between P1 and P3 (p = 0.0416) implied that the use of combined nano-herbals enhances MDA reduction more than Bischofia javanica alone. The absence of a significant difference between P2 and P3 (p = 0.2208) implies that Phaleria macrocarpa may exert comparable effects independently. These results underscore the therapeutic potential of both individual and combined treatments in reducing oxidative stress in OSCC.
Groups
Mean ± SD
IRS
Groups
K0
K1
P1
P2
P3
P4
K0
18.61 ± 4.65
Weak
0.0014**
0.0242*
0.0304*
0.2135ns
0.4496ns
K1
66.67 ± 7.59
Moderate
0.2224ns
0.0530ns
0.0175*
0.0019**
P1
50.89 ± 5.60
Moderate
0.5048ns
0.0416*
0.0229*
P2
39.78 ± 6.78
Moderate
0.2208ns
0.0420*
P3
28.78 ± 2.68
Weak
0.8168ns
P4
20.89 ± 3.76
Moderate
The IRS results indicated weak MDA expression in the negative control group (K0) and moderate levels in K1, P1, P2, and P4, suggesting the presence of oxidative stress in OSCC tissues. The P3 group, which received the combined treatment, demonstrated a significant reduction in MDA levels (Fig. 2). The visualization of MDA expression in the oral mucosa corroborates these findings, underscoring the effects of the treatments on oxidative stress. Overall, the results illustrate the efficacy of the nano-herbal formulations in reducing oxidative stress, highlighting the potential of both plants in cancer therapy and justifying further research into their mechanisms and clinical applications.
3.2 The SOD levels in blood serum after the administration of nano-herbal
The results indicated a significant difference in Superoxide Dismutase (SOD) levels among the groups (p < 0.05, F = 0.0015). The control group (K0) exhibited the highest SOD levels, while the OSCC-induced group (K1) displayed the lowest, suggesting oxidative stress due to reactive oxygen species (ROS) accumulation (Fig. 1). SOD contributes significantly to antioxidant defense by converting superoxide radicals into hydrogen peroxide and oxygen molecules. The decreased SOD activity implies that excessive ROS production overwhelms the cellular antioxidant capacity, thereby contributing to cancer progression (Van Loenhout et al., 2020). Furthermore, oxidative stress induced by carcinogens disrupts cellular homeostasis, a key factor in the pathogenesis of OSCC (Iqbal et al., 2024).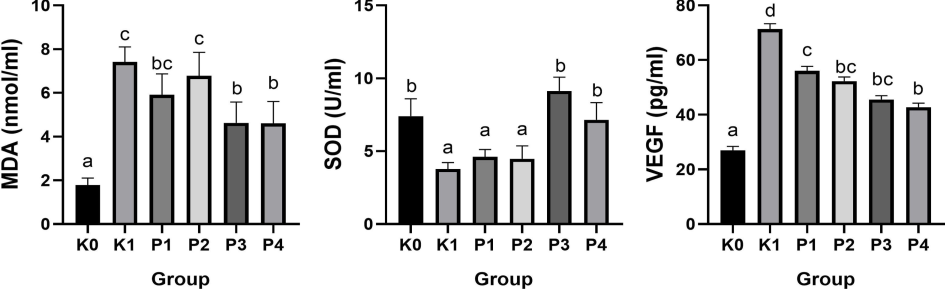
Serum MDA, SOD, and VEGF levels. a, b, c, d signify significant disparities between the groups.
The administration of nanoherbal treatments with Bischofia javanica and Phaleria macrocarpa in groups P1, P2, and P3 significantly restored SOD levels compared to the OSCC group (K1), with the P3 group exhibiting the highest SOD activity (Fig. 1). This improvement suggests that antioxidant compounds in these herbs, such as flavonoids and polyphenols, enhance SOD activity and mitigate the effects of ROS. Bischofia javanica is rich in polyphenols, which are known to boost antioxidant enzyme activity, while Phaleria macrocarpa contains bioactive compounds like mangiferin and gallic acid, recognized for their free radical-scavenging properties (Lee et al., 2021; Ahmad et al., 2023). The synergistic action of both herbs in the P3 group more effectively reduces oxidative damage than individual treatments, as reflected by the increased SOD levels. The rise in SOD levels in the P3 group indicates enhanced antioxidant defense and a reversal of oxidative damage in cancerous tissues, likely due to the improved bioavailability of nano-sized herbal particles, which facilitates better cellular uptake and antioxidant efficacy. (Chowdhury et al., 2020; Elumalai et al., 2024). While Vitamin C treatment (P4) also elevated SOD levels compared to K1, the combination of both plants in P3 demonstrated greater efficacy. This observation aligns with findings indicating that polyphenolic-rich plant extracts frequently outperform synthetic antioxidants such as Vitamin C. In conclusion, the increase in SOD levels, particularly in the P3 group, underscores the effectiveness of Bischofia javanica and Phaleria macrocarpa in restoring antioxidant balance in oral squamous cell carcinoma (OSCC). These nanoherbals may serve as complementary therapies, potentially exceeding the efficacy of Vitamin C. Future research should focus on elucidating their molecular mechanisms and exploring their clinical applications in cancer therapy.
3.3 The VEGF levels in blood serum after the administration of nano-herbal
VEGF analysis revealed Significant contrasts observed among the groups (p < 0.05, F = 0.000), with group K1 exhibiting the highest levels and group K0 the lowest. The nano-herbal treatments resulted in a reduction of VEGF levels compared to K1, with group P1 showing slightly higher levels than P2, indicating varied responses to the treatments. VEGF plays a critical role in angiogenesis and tumor growth, and its levels were elevated in OSCC (K1) (Fig. 1). The nano-herbals derived from both plants effectively inhibited VEGF production, likely attributable to their bioactive compounds, including flavonoids, tannins, mangiferin, and gallic acid (Yuliet et al., 2022).
The nanoformulation significantly enhances bioavailability, thereby improving the absorption of bioactive compounds (Elumalai et al., 2024). Vitamin C (P4) effectively lowered VEGF levels compared to the OSCC group; however, it was less effective than the nano-herbals (Fig. 1). The absence of a significant difference in VEGF levels between groups P1 and P2 indicates that both herbs possess strong anti-angiogenic effects, although the slight increase observed in P1 may be attributed to the unique constituents of Bischofia javanica. The combination treatment (P3) likely amplifies these effects, demonstrating the therapeutic potential of nano-herbals in targeting cancer progression through modulation of the tumor microenvironment (Lu et al., 2024).
In summary, the nano-herbals of both plants effectively reduced VEGF levels in OSCC-induced rats, suggesting their potential to inhibit angiogenesis (Maharani et al., 2021; Tandrasasmita et al., 2015). Their enhanced bioavailability further supports the targeting of cancer progression pathways, positioning them as promising complementary therapies to conventional antioxidants like Vitamin C. Further research is necessary to uncover the molecular pathways involved in their anti-angiogenic effects and to evaluate their clinical relevance in cancer therapy.
3.4 The presence of HSP-70 protein within the oral cavity mucosa
Statistical analysis using the Kruskal-Wallis test showed notable differences in HSP-70 expression among the experimental groups (p < 0.05, F = 0.018), a finding further confirmed by the Mann-Whitney follow-up test (see Table 3). Group P3 exhibited a notable increase in HSP-70 levels, with moderate expression observed across all OSCC-induced groups. Histological images demonstrated heightened HSP-70 expression under stress, characterized by distinct dark brown staining in squamous cells, particularly evident in group P3 (See Fig. 3). HSP-70 (Heat Shock Protein 70) functions as a molecular chaperone, protecting cells from stress-induced damage by preventing protein aggregation and facilitating the repair of misfolded proteins. Its expression increases in response to various stressors, including oxidative stress, heat, and inflammation, and is closely associated with cancer growth responses.
Groups
Mean ± SD
IRS
Groups
K0
K1
P1
P2
P3
P4
K0
17.61 ± 5.05
Weak
0.0424*
0.0061**
0.0598**
0.0025**
0.002**
K1
61.00 ± 8.70
Moderate
0.7475ns
0.0521ns
0.0072**
0.0371ns
P1
55.28 ± 6.61
Moderate
0.9154ns
0.0181*
0.0193*
P2
53.17 ± 7.56
Moderate
0.9135*
0.8833ns
P3
68.72 ± 7.43
Moderate
0.6499*
P4
17.61 ± 5.05
Weak
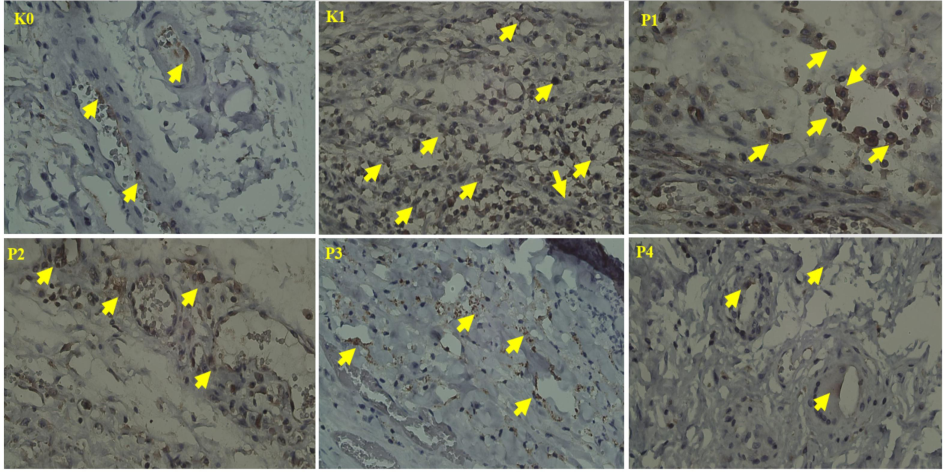
Expression of MDA in the oral mucosa. Arrows indicate positive expression of MDA (400x).
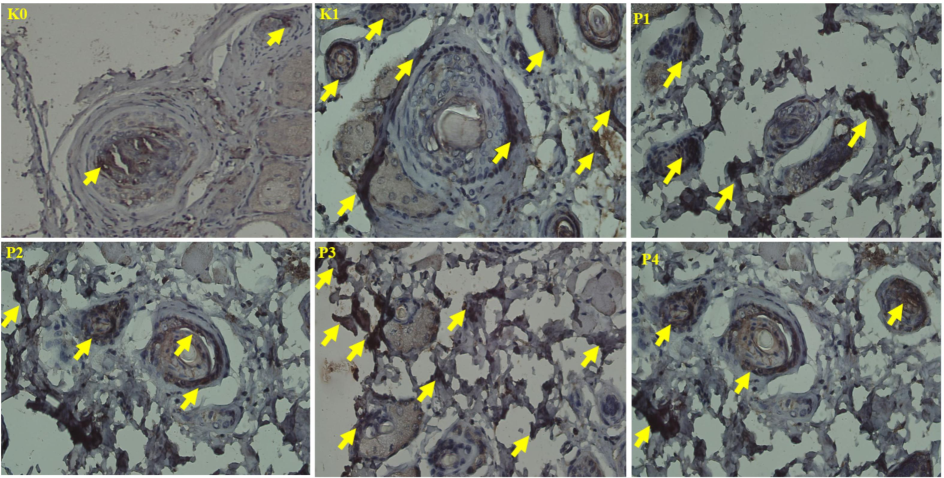
HSP-70 expression in the oral mucosa. Arrows indicate positive HSP-70 expression. (400x).
In oral squamous cell carcinoma (OSCC), elevated levels of HSP-70 indicate cellular stress resulting from uncontrolled cancer growth (Elhendawy, 2023). The positive control group (K1) exhibited moderate HSP-70 expression, reflecting the tumor cells' reliance on protective mechanisms under hypoxic and oxidative conditions. In contrast, group P3, treated with a combination of both plants, demonstrated a significant increase in HSP-70 expression. This rise is associated with cancer-induced stress and the body’s protective response, as the nano-herbal therapy activates stress response pathways to mitigate oxidative stress and inflammation caused by the tumor.
Variations in HSP-70 levels among the treatment groups (P1, P2, P4) can be attributed to the differing bioactive compounds present in the nano-herbals. Bischofia javanica contains antioxidants such as tannins and flavonoids, which reduce oxidative stress and activate HSP-70 (Rumahorbo et al., 2024a). Similarly, Phaleria macrocarpa, rich in phenolic compounds like mangiferin, also decreases oxidative stress and induces HSP-70 expression (Castro-Muñoz et al., 2024). The increased HSP-70 expression observed in group P3 compared to the positive control (K1) highlights the potential of these nano-herbals to modulate stress-response pathways. The nano-sizing of these compounds enhances their bioavailability, improving their interaction with intracellular signaling pathways and increasing the accumulation of stress signals, which triggers HSP-70 expression.
Although HSP-70 expression increased in group P3, the nano-herbals may not completely neutralize oxidative stress and inflammation within the OSCC environment. The upregulation of HSP-70 may function as a final defense mechanism against cancer and stress, highlighting the complex interplay between treatment and cellular responses. Histological analysis reveals minimal stress in the negative control group (K0) and moderate HSP-70 expression in the OSCC-induced group (K1). The higher HSP-70 staining observed in group P3 suggests a more robust stress response resulting from oxidative stress and the therapeutic effects of the nano-herbals. This elevation in HSP-70 indicates cellular stress associated with both OSCC progression and treatment. While the nano-herbals enhance bioavailability and intracellular interactions, they also provoke a stress response. Further research is necessary to elucidate how nano-herbals influence stress-response proteins and their role in cancer therapy.
3.5 The presence of Caspase-3 within the oral mucosa
Caspase-3 expression analysis revealed significant differences among the groups, particularly in P3 and P4 when compared to the positive control (K1). The highest expression observed in P3 (p = 0.0007***) underscores the potent pro-apoptotic effects of the nano-herbals derived from both plants (Lingadurai et al., 2011; Altaf et al., 2013) (see Table 4). These nano-herbals enhance apoptosis by activating the intrinsic pathway through Caspase-3, which is a crucial executor of programmed cell death.
Groups
Mean ± SD
IRS
Groups
K0
K1
P1
P2
P3
P4
K0
10.78 ± 4.02
Moderate
0.0255*
0.0002***
<0.0001****
<0.0001***
<0.0001****
K1
21.56 ± 4.62
Weak
0.038*
0.018*
0.0007***
0.0006***
P1
41.44 ± 5.94
Moderate
0.1682ns
0.0480*
<0.0001****
P2
57.67 ± 6.29
Moderate
0.6048ns
0.0311*
P3
66.67 ± 8.06
Moderate
0.3070ns
P4
79.89 ± 8.35
Moderate
Caspase-3 plays a crucial role in apoptosis by cleaving substrates and facilitating DNA fragmentation (Ghorbani et al., 2024). The increased expression of Caspase-3 in OSCC tissues treated with nano-herbals indicates effective induction of apoptosis, which is essential for countering cancer cell proliferation. Significant differences in Caspase-3 levels between the treated groups and K1 suggest that nano-herbals can restore the downregulated apoptotic machinery in cancer cells. The combination of both plants enhances pro-apoptotic pathways through their bioactive compounds, including flavonoids and polyphenols. These compounds modulate oxidative stress and induce apoptosis, particularly in cancer cells, where increased oxidative stress activates the intrinsic apoptotic pathway (An et al., 2024). Furthermore, the nano-sized formulation improves bioavailability, allowing deeper penetration into tumor tissues, as evidenced by the significant rise in Caspase-3 expression in P3 compared to P1 (p = 0.0480*).
Histopathological analysis (Fig. 4) corroborates these findings by demonstrating distinct morphological changes in the nano-herbal treatment groups, including the presence of apoptotic bodies, nuclear condensation, and cell shrinkage in P3 and P4, indicating the activation of the apoptosis cascade (Chen et al., 2024). In contrast, the untreated group (K1) exhibited fewer apoptotic features, suggesting that cancer cells in this cohort evaded apoptosis—a hallmark of tumor progression (Chen et al., 2024). The upregulation of Caspase-3 in the treatment groups is associated with the nano-herbals' ability to modulate oxidative stress. Enhanced levels of ROS found in cancer cells can either promote survival or trigger apoptosis, depending on the specific cellular context (An et al., 2024). The nano-herbals, possessing both antioxidant and pro-oxidant properties, may favor oxidative stress-induced apoptosis by inducing mitochondrial dysfunction and facilitating the release of cytochrome c, which activates Caspase-3 through the intrinsic pathway (Mishra et al., 2022). Furthermore, nano-herbals may enhance the functions of pro-apoptotic proteins, such as p53, through direct interaction (Dehelean et al., 2021).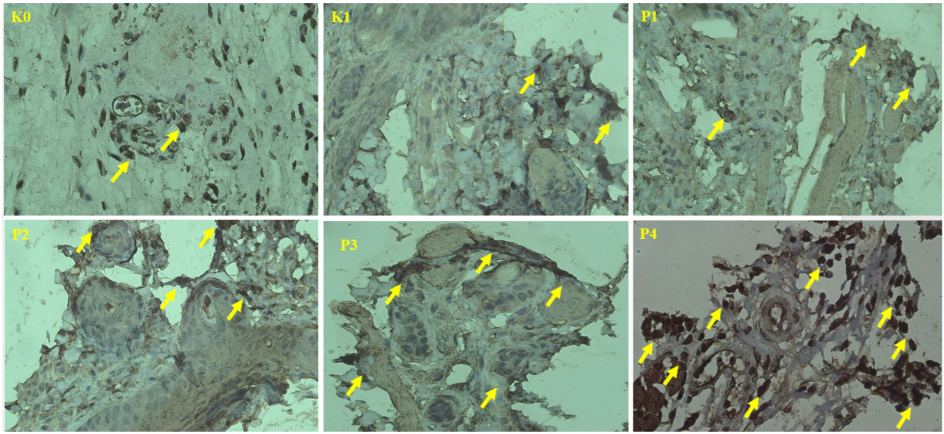
Caspase-3 expression in the oral mucosa. Arrows indicate positive Caspase-3 expression. (400x).
This study reinforces existing research on the significance of combined herbal therapies in enhancing apoptotic responses in cancer cells. The synergistic effects of Bischofia javanica and Phaleria macrocarpa on Caspase-3 activation position them as promising candidates for anti-cancer therapy, particularly relevant to OSCC. The elevated Caspase-3 expression observed in the P3 group suggests that this combination may outperform individual herbal treatments, presenting a robust strategy for inducing apoptosis in cancer cells and potentially improving patient outcomes. These findings establish a solid scientific foundation for the development of nano-herbal formulations as potential cancer therapies, emphasizing their capacity to modulate oxidative stress and deliver pro-apoptotic compounds directly to tumor cells. Further research is warranted to optimize dosage regimens and evaluate long-term efficacy in clinical settings.
3.6 Caspase-8 expression in the oral cavity mucosa
The Kruskal-Wallis test indicated significant differences in Caspase-8 expression among the experimental groups (p < 0.05, F = <0.0001). Follow-up analysis using the Mann-Whitney test revealed that the nano-herbal-treated groups, specifically P2 (p = 0.029*), P3 (p = 0.00478**), and P4 (p < 0.0001****), exhibited significantly higher Caspase-8 expression compared to the untreated K1 group (see Table 5). These findings underscore the potent apoptotic effects of the combined plants, which likely exert their action through the extrinsic apoptotic pathway (Lingadurai et al., 2011; Altaf et al., 2013).
Groups
Mean ± SD
IRS
Groups
K0
K1
P1
P2
P3
P4
K0
12.78 ± 1.41
Weak
0.0148*
0.0009***
0.0002***
0.0096**
<0.0001****
K1
24.00 ± 3.58
Moderate
0.0651ns
0.029*
0.00478**
<0.0001****
P1
30.55 ± 7.62
Moderate
0.1547ns
0.8139ns
0.0014**
P2
37.11 ± 4.79
Moderate
0.8812ns
0.0177*
P3
42.44 ± 5.65
Moderate
0.2474ns
P4
54.78 ± 1.61
Moderate
Caspase-8 is a crucial initiator of the extrinsic apoptotic pathway, activated by the binding of death ligands to cell membrane receptors. Once activated, it cleaves effector caspases such as Caspase-3, leading to cell death (Zhang et al., 2024). The significant expression of Caspase-8 observed in the P3 and P4 groups of our study indicates that the nano-herbals effectively activated this apoptotic pathway, resulting in cancer cell death. Histological analysis further supports this finding, revealing typical apoptotic features in carcinoma cells from the treated groups, including cell shrinkage, nuclear condensation, and cytoplasmic swelling.
The activation of Caspase-8 by Bischofia javanica and Phaleria macrocarpa is associated with their phytochemical constituents. Bischofia javanica contains compounds such as beta-amyrin and betulinic acid, which induce pro-apoptotic effects through death receptor pathways (Chauhan et al., 2023). Similarly, the polyphenolic compounds found in Phaleria macrocarpa, including flavonoids and tannins, enhance cell sensitivity to death receptor signaling (Lay et al., 2014). These bioactive compounds may interact with death receptors, such as Fas and TNF receptors, to initiate the extrinsic pathway via Caspase-8 (Zhang et al., 2024). Furthermore, the nano-formulation of these extracts improves their bioactivity by enhancing cellular uptake and interaction with cancer cells (Elumalai et al., 2024), resulting in significantly higher Caspase-8 expression in group P4, where the nano-herbal combination exhibited the most pronounced apoptotic effect.
Histological examination confirms that Caspase-8-mediated apoptosis occurs in groups P3 and P4, where carcinoma mucosa cells display overlapping and irregular formations with prominent apoptotic features, such as swollen cytoplasm and polymorphic nuclei (see Fig. 5). In contrast, the untreated K1 group exhibits fragmented squamous cells without Caspase-8 expression, indicating inactive apoptosis despite cancer progression. This underscores the ability of nano-herbals to restore apoptotic signaling in cancer cells resistant to cell death (Chaudhary and Rajora, 2024). The significant upregulation of Caspase-8 in the treated groups is attributed to the interaction of nano-herbals with the death receptor pathway. The nano-sized herbal particles enhance penetration into tumor tissues, allowing efficient binding to death receptors and subsequent Caspase-8 activation. This increased bioavailability ensures the direct delivery of herbal compounds to cancer cells, effectively triggering apoptosis, while the antioxidant properties of nano-herbals may protect surrounding normal tissues from oxidative stress, thus facilitating targeted cancer therapy (Elumalai et al., 2024).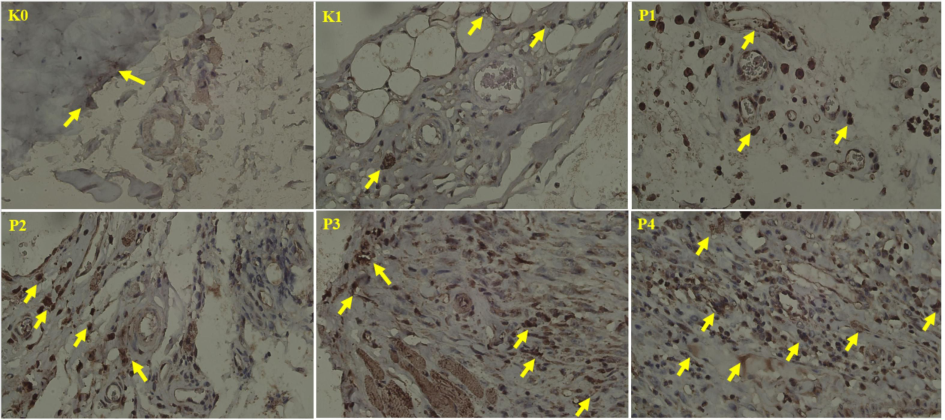
Expression of Caspase-8 in the oral mucosa. Arrows indicate positive Caspase-8 expression). (400x).
The ability of the both plants nano-herbals to upregulate Caspase-8 expression and induce apoptosis via the extrinsic pathway demonstrates significant therapeutic potential for OSCC treatment. This combination enhances cancer cells' sensitivity to apoptosis induced by death receptors, providing a strategy to overcome resistance to cell death. The synergistic effects observed in group P4 suggest that this therapy may be more effective than conventional single-agent treatments. In conclusion, the study demonstrates that nano-herbal formulations significantly enhance Caspase-8 expression in OSCC cells, thereby activating the extrinsic apoptotic pathway. This elucidates the mechanisms through which these nano-herbals induce apoptosis, supporting their development as effective anti-cancer therapies and presenting a novel approach to targeting OSCC for improved patient outcomes.
3.7 Expression of Caspase-10 in the oral mucosa
Statistical analysis using the Kruskal-Wallis test uncovered notable differences in Caspase-10 expression among the experimental groups (p < 0.05, F < 0.0001). The Mann-Whitney tests further confirmed significant differences in Caspase-10 expression in the treatment groups P2, P3, and P4 compared to the control group K1. Immunohistochemical (IHC) staining showed moderate Caspase-10 expression in all groups except K0, which exhibited weak expression (Table 6). Fig. 6 illustrates Caspase-10 expression across the groups, emphasizing cellular fragmentation and inflammatory changes, particularly in group P1.
Groups
Mean ± SD
IRS
Groups
K0
K1
P1
P2
P3
P4
K0
8.22 ± 3.83
Weak
0.0138*
0.0019**
<0.0001****
0.0002***
<0.0001****
K1
13.55 ± 3.58
Moderate
0.2104ns
0.0012**
0.0036**
<0.0001****
P1
16.22 ± 4.39
Moderate
0.0465*
0.0303*
0.0002***
P2
20.33 ± 5.36
Moderate
0.2290ns
0.0009***
P3
26.11 ± 5.46
Moderate
0.1413ns
P4
34.22 ± 2.96
Moderate
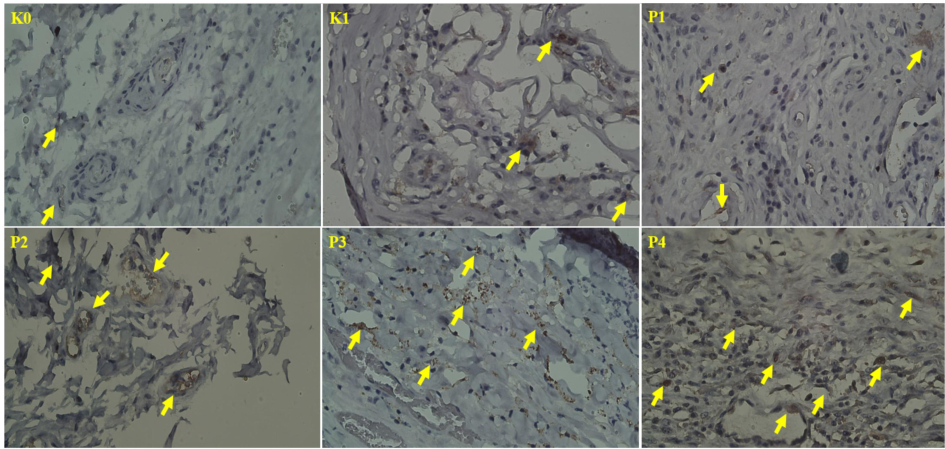
Expression of Caspase-10 in the oral mucosa. Arrows indicate positive expression of Caspase-10. (400x).
Caspase-10, akin to Caspase-8, functions as an initiator caspase within the extrinsic apoptosis pathway, engaging with death receptors such as Fas and TNF receptors to activate apoptosis by subsequently triggering downstream effector caspases, including Caspase-3. This caspase provides redundancy in the extrinsic pathway, serving as an alternative mechanism for apoptosis in certain cancer types (Sahoo et al., 2023). In the present study, the moderate expression of Caspase-10 observed in treatment groups P3 and P4 suggests that the nano-herbal treatment effectively activates both Caspase-8 and Caspase-10. This indicates that the combination of Bischofia javanica and Phaleria macrocarpa enhances apoptotic effects via both pathways, thereby reinforcing the overall apoptotic response.
The moderate Caspase-10 expression in groups P2, P3, and P4 suggests that the nano-herbal formulations of both plants enhance Caspase-10-mediated apoptosis (Ahmad et al., 2023; Lee et al., 2021). Phytochemicals like betulinic acid and mangiferin in these herbs activate death receptor pathways, upregulating both Caspase-8 and Caspase-10 to promote apoptosis in cancer cells. The improved bioavailability and cellular uptake from the nano formulation contribute to the strong apoptotic response, particularly in P4, where the combination of both nano-herbals resulted in the highest Caspase-10 expression. This nanoparticle delivery system enhances penetration into tumor cells, facilitating efficient binding of herbal compounds to death receptors and activating the apoptosis pathway (Chaudhary and Rajora, 2024).
Histological analysis of group P1 revealed significant cell fragmentation and inflammation, characterized by the presence of macrophages, which indicates active Caspase-10-mediated apoptosis, albeit at lower levels than those observed in group P4. The infiltration of macrophages is likely attributable to the recruitment of immune cells for the phagocytosis of apoptotic bodies. Despite the fragmentation noted in P1, the lower Caspase-10 expression compared to P4 suggests a more pronounced apoptotic effect resulting from the dual nano-herbal treatment in P4. In contrast, while moderate Caspase-10 expression was observed in the untreated group K1, there was no significant cell death, indicating incomplete engagement of the apoptotic process. This may be due to the overexpression of anti-apoptotic proteins, such as FLIP, in cancer cells, which inhibit caspase activation despite the presence of death receptor signaling (Boice and Bouchier-Hayes, 2020). The moderate Caspase-10 staining index across the groups, except for K0, indicates the resistance of cancer cells to apoptosis. In oral squamous cell carcinoma (OSCC), this resistance is frequently associated with downregulated caspase activity or upregulated inhibitors of apoptosis proteins (IAPs). The enhanced expression of Caspase-10 in group P4 suggests that the phytochemicals present in Bischofia javanica and Phaleria macrocarpa may modulate pro-apoptotic gene expression or inhibit anti-apoptotic pathways, thereby effectively overcoming this resistance. Furthermore, the nano-formulation likely improves the cellular uptake of bioactive compounds, facilitating better interaction with death receptors and activation of the extrinsic pathway (Elumalai et al., 2024). The ability to induce apoptosis through both the Caspase-8 and Caspase-10 pathways underscores the therapeutic potential of these nano-herbals against cancer cells that exhibit resistance to single apoptotic pathways.
The findings indicate that nano-herbal formulations of both plants activate multiple apoptotic pathways in OSCC, as evidenced by the upregulation of both Caspase-8 and Caspase-10. This dual activation may assist in overcoming the apoptosis resistance frequently encountered in cancer therapy, thereby positioning these nano-herbals as effective adjuvants or alternatives to conventional chemotherapy. They promote cancer cell death through diverse apoptotic mechanisms.
4 Conclusion
In conclusion, this study demonstrates that the nano-herbal from Bischofia javanica and Phaleria macrocarpa in combination significantly induces apoptosis in oral squamous cell carcinoma (OSCC) and reduces oxidative stress in experimental animals induced with oral cancer by benzo alpha pyrene. This combination outperforms the effects of each extract individually. The findings also underscore the potent antioxidant properties of Vitamin C. However, the study's limitations include its focus on a specific animal model, which may not fully translate to humans, as well as the exclusion of other potential synergistic herbal combinations. Future research should involve clinical trials to evaluate the effectiveness and safety of this nano-herbal combination in humans. Additionally, investigations should explore interactions with standard chemotherapy agents, long-term effects, and pharmacokinetics while expanding to include other cancer types and bioactive compounds.
CRediT authorship contribution statement
Cheryl Grace Pratiwi Rumahorbo: Writing – review & editing, Writing – original draft, Visualization, Software, Resources, Methodology, Investigation, Formal analysis, Data curation, Conceptualization. Syafruddin Ilyas: Writing – review & editing, Validation, Software, Resources, Project administration, Funding acquisition, Conceptualization.
Acknowledgements
The authors would like to express their profound gratitude to Universitas Sumatera Utara for the opportunity to undertake this post-doctoral program under the work agreement number 21299/UN5.1.R/KPM/2024. Special thanks are extended to Lembaga Pengelola Dana Pendidikan (LPDP) and the Enhancing Quality Education for International University Recognition (EQUITY) Project for providing financial support for this program. The authors also wish to extend their heartfelt appreciation to Kementerian Pendidikan Tinggi dan Riset for its invaluable support and contributions to advancing research and higher education.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Phaleria macrocarpa (Scheff.) boerl.: An updated review of pharmacological effects, toxicity studies, and separation techniques. Saudi Pharm J.. 2023;31(6):874-888.
- [CrossRef] [Google Scholar]
- Phytochemistry and medicinal properties of Phaleria macrocarpa (Scheff.) Boerl. Extracts,” Pharmacogn Rev.. 2013;7(13):73-80.
- [CrossRef] [Google Scholar]
- Oxidative cell death in cancer: mechanisms and therapeutic opportunities. Cell Death Dis. 2024;15:556.
- [CrossRef] [Google Scholar]
- Targeting apoptotic caspases in cancer. Biochim Biophys Acta Mol Cell Res.. 2020;1867(6):118688
- [CrossRef] [Google Scholar]
- High-dose intravenous vitamin C, a promising multi-targeting agent in the treatment of cancer. J Exp Clin Cancer Res.. 2021;40(1):343.
- [CrossRef] [Google Scholar]
- Benzo[a]pyrene-environmental occurrence, human exposure, and mechanisms of toxicity,. Int. J. Mol. Sci.. 2022;23(11):6348.
- [CrossRef] [Google Scholar]
- Mangiferin: A comprehensive review on its extraction, purification and uses in food systems. Adv Colloid Interface Sci.. 2024;329:103188
- [CrossRef] [Google Scholar]
- Phytosomes: a critical tool for delivery of herbal drugs for cancer. Phytochem Rev 2024
- [CrossRef] [Google Scholar]
- Chauhan, H. K., Oli, S., Bisht, A. K., Bhat, I. D., 2023. Chapter 5 - Bischofia javanica Blume. Himalayan Fruits and Berries Bioactive Compounds, Uses and Nutraceutical Potential. Pages 39-46.
- Chen, Y., Li, X., Yang, M., 2024.Research progress on morphology and mechanism of programmed cell death. Cell Death Dis 15, 327 (2024). DOI: 10.1038/s41419-024-06712-8.
- An integrated exploration of pharmacological potencies of Bischofia javanica (Blume) leaves through experimental and computational modeling. Heliyon. 2020;6(9):e04895.
- [Google Scholar]
- The combination of Elephantopus scaber and Phaleria macrocarpa leaves extract promotes anticancer activity via downregulation of ER-α, Nrf2 and PI3K/AKT/mTOR pathway. J Ayurveda Integr Med.. 2022;13(4):100674
- [CrossRef] [Google Scholar]
- Plant-derived anticancer compounds as new perspectives in drug discovery and alternative therapy. Molecules. 2021;26(4):1109.
- [CrossRef] [Google Scholar]
- Clinical implications of heat shock protein 70 in oral carcinogenesis and prediction of progression and recurrence in oral squamous cell carcinoma patients: a retrospective clinicopathological study. Eur J Med Res. 2023;28:464.
- [CrossRef] [Google Scholar]
- Review of the efficacy of nanoparticle-based drug delivery systems for cancer treatment. Biomed Tech.. 2024;5:109-122.
- [CrossRef] [Google Scholar]
- How does caspases regulation play role in cell decisions? apoptosis and beyond. Mol Cell Biochem. 2024;479:1599-1613.
- [CrossRef] [Google Scholar]
- Interplay of oxidative stress, cellular communication and signaling pathways in cancer. Cell Commun Signal. 2024;22:7.
- [CrossRef] [Google Scholar]
- Tobacco and oral squamous cell carcinoma: A review of carcinogenic pathways. Tob Induc Dis.. 2019;12(17):29.
- [CrossRef] [Google Scholar]
- Lay, M. M., Karsani, S.A., Banisalam, B., Mohajer, S., Abd Malek, S. N., 2014. Antioxidants, phytochemicals, and cytotoxicity studies on Phaleria macrocarpa (Scheff.) Boerl seeds. Biomed Res Int. 2014, 410184, DOI: 10.1155/2014/410184.
- Antioxidant and anti-inflammatory effects of bischofia javanica (Blume) leaf methanol extracts through the regulation of Nrf2 and TAK1. Antioxidants (basel).. 2021;10(8):1295.
- [CrossRef] [Google Scholar]
- Antileukemic activity of the leaf extract of Bischofia javanica blume on human leukemic cell lines. Indian J Pharmacol.. 2011;43(2):143-149.
- [CrossRef] [Google Scholar]
- Nanoparticles in tumor microenvironment remodeling and cancer immunotherapy. J Hematol Oncol. 2024;17:16.
- [CrossRef] [Google Scholar]
- Phytochemical characteristics from Phaleria macrocarpa and its inhibitory activity on the peritoneal damage of endometriosis. J Ayurveda Integr Med. 2021;12(2):229-233.
- [CrossRef] [Google Scholar]
- Serum malondialdehyde as a biomarker of oxidative stress in patients with primary ocular carcinoma: impact on response to chemotherapy. Clin Ophthalmol.. 2021;26(15):871-879.
- [CrossRef] [Google Scholar]
- Application of nanotechnology to herbal antioxidants as improved phytomedicine: An expanding horizon. Biomed. Pharmacother.. 2022;153:113413
- [CrossRef] [Google Scholar]
- The genomics of oxidative DNA damage, repair, and resulting mutagenesis. Comput. Struct. Biotechnol. J.. 2020;18:207-219.
- [CrossRef] [Google Scholar]
- Bischofia javanica and Phaleria macrocarpa nano herbal combination on blood and liver-kidney biochemistry in Oral Squamous Cell Carcinoma-induced rats. Pharmacia. 2024;71:1-8.
- [CrossRef] [Google Scholar]
- Chronic toxicity assessment of nano-formulated Bischofia javanica leaves: Implications for pharmacological use. Pharmacia. 2023;70(4):1101-1109.
- [CrossRef] [Google Scholar]
- Advancing sustainable herbal medicine: Synthesizing nanoparticles from medicinal plants. E3S Web Conf. 2024;519:03038.
- [CrossRef] [Google Scholar]
- Oral chronic toxicity test of nano herbal P. macrocarpa. Pharmacia. 2023;70(2):411-418.
- [CrossRef] [Google Scholar]
- The improvement of the physiological effects of nanoherbal sikkam leaves (Bischofia javanica) Rasayan J. Chem.. 2023;16(2):766-772.
- [CrossRef] [Google Scholar]
- A review on caspases: key regulators of biological activities and apoptosis. Mol Neurobiol. 2023;60:5805-5837.
- [CrossRef] [Google Scholar]
- Betulinic acid inhibits cell proliferation in human oral squamous cell carcinoma via modulating ROS-regulated p53 signaling. Oncol. Res.. 2017;25(7):1141-1152.
- [CrossRef] [Google Scholar]
- Acute toxicity test nano-based herbal Mahkota dewa fruit (P. macrocarpa) Pharmacia. 2022;69(4):1063-1074.
- [CrossRef] [Google Scholar]
- Effectiveness of nano-herbal Phaleria macrocarpa on physiological evaluation in Rattus norvegicus. Pharmacia. 2024;71:1-8.
- [CrossRef] [Google Scholar]
- Anti-inflammatory, antiangiogenic, and apoptosis-inducing activity of DLBS1442, a bioactive fraction of Phaleria macrocarpa, in a RL95-2 cell line as a molecular model of endometriosis. Int J Womens Health.. 2015;3(7):161-169.
- [CrossRef] [Google Scholar]
- The Global Cancer Observatory: All Cancer, https://gco.iarc.fr/today/data/factsheets/cancers/39-all-cancers-fact-sheet.pdf; 2020 [accessed 14 January 2024].
- Oxidative stress-inducing anticancer therapies: taking a closer look at their immunomodulating effects. Antioxidants Basel). 2020;9(12):1188.
- [Google Scholar]
- World Health Organization. Oral health, https://www.who.int/news-room/fact-sheens/detail/oral-health; 2023 [accessed 12 September 2023].
- Involvement and targeted intervention of benzo(a)pyrene-regulated apoptosis related proteome modification and muti-drug resistance in hepatocellular carcinoma. Cell Death Dis. 2023;14:265.
- [CrossRef] [Google Scholar]
- Immunomodulatory activity of Pepolo (Bischofia javanica Blume) stem bark ethanolic extract in Staphylococcus aureus-stimulated macrophages and anticancer activity against MCF- 7 cancer cells. J Appl Pharm Sci.. 2022;12(10):109-116.
- [CrossRef] [Google Scholar]
- Zasowska-Nowak, A., Nowak, P. J., Ciałkowska-Rysz, A., 2021. High-Dose Vitamin C in Advanced-Stage Cancer Patients. Nutrients. 2021 Feb 26;13(3):735, DOI: 10.3390/nu13030735.
- Caspase-8 in inflammatory diseases: a potential therapeutic target. Cell Mol Biol Lett. 2024;29:130.
- [CrossRef] [Google Scholar]
Appendix A
Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.jksus.2024.103548.
Appendix A
Supplementary data
The following are the Supplementary data to this article:







