Translate this page into:
Effectiveness and safety of chemical inhibitors against mammalian target of rapamycin (mTOR) for primary immunosuppression in recipients of kidney transplant: A systematic review and meta-analysis
⁎Corresponding author. shhaque@jazau.edu.sa (Shafiul Haque)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The current systematic review and meta-analysis was undertaken to assess the efficaciousness of mammalian target of rapamycin (mTOR) inhibitors in transplant subjects with regards to kidney functions and survival, with special reference to co-administration (or absence of) calcineurin inhibitors (CNIs). The analysis was done through searching and retrieving information from online scholarly databases. The collected data represented outcomes after at least twelve months following transplantation of kidney. It was observed that parameters such as glomerular filtration rate (GFR) was improved in subjects administered with mTOR inhibitors, however some studies indicated that acute rejection following biopsy was dominant in subjects administered with mTOR inhibitors. Owing to their complementary mechanisms of action as well as beneficial effects on mitigating nephrotoxicity, concomitantly with favorable outcomes on parameters such as serum creatinine and GFR leading to increased survival, this meta-analysis proposes early utilization of mTOR inhibitors and CNI minimization in subjects with kidney transplantation.
Keywords
Kidney transplantation
mTOR inhibitors
Calcineurin inhibitors
Glomerular filtration rate
Serum creatinine
Nephrotoxicity
- BPAR
-
Biopsy-proven acute rejection
- CENTRAL
-
Cochrane Central Register of Controlled Trials
- CI
-
confidence intervals
- CNIs
-
calcineurin inhibitors
- FKBP
-
FK506-binding protein
- GFR
-
glomerular filtration rate
- GRADE
-
Grading of Recommendations Assessment, Development and Evaluation
- HLA
-
human leukocyte antigen
- MD
-
mean difference
- mTOR
-
mammalian target of rapamycin
- OR
-
odds ratio
- PRISMA
-
Preferred Reporting Items for Systematic Reviews and Meta-Analyses
Abbreviations
1 Introduction
Several novel inventions have improved clinical aspects of kidney transplantations, resulting in increased life expectancies of subjects with chronic kidney diseases (Salvadori and Bertoni, 2013). Employment of immunosuppressive agents such as tacrolimus, cyclosporine A, and calcineurin inhibitors (CNIs) initiated in the 1980 s has been estimated to lower rejection rates to 20 %, and increase survival to 90 % for a year (Knops et al., 2013). The primary factor for pertained graft loss is governed by complex immunological and non-immunological processes including hypertension, proteinuria as well as pathological attributes such as interstitial fibrosis atrophy. The immunological attributes encompass human leukocyte antigen (HLA) complementation and other immune system-related events of rejection, and average immunosuppression. In contrast, non-immunological characteristics involve features of an organ donated, retracted functioning of graft, infection, and hyperlipidemia (Hernández et al., 2011). Further, the nephrotoxicity modulated via CNI is the most relevant consequence of the long-term failure of graft, accounting for up to 96.8 % of the failures. The failure of allograft biopsies is ascribed to enhancement in vasoconstrictors, such as endothelin and thromboxane production, concomitantly with decline in the production of vasodilators (Li and Yang, 2009).
Evaluation of allograft biopsies and histological assessment of tubular atrophy, narrowing of the luminal tissue, sclerosis, and deposition of calcium have revealed that more than 50 % of the cases elicit chronic toxicity with CNIs administered following ten years of transplantation. Moreover, CNI administration results in severe consequences for cardiovascular parameter, like hyperlipidemia, hypertension, and diabetes mellitus post-transplantation (Flechner, 2009). The most challenging aspect in therapeutics with immunosuppression is balancing out its need so as to prevent any episode of rejection from occurring, while reducing the chances of any probable toxicities. Other immunosuppressive agents such as mTOR inhibitors; such as sirolimus and everolimus which have been employed in recent years, elicit similar modes of action as CNIs with regards to nephrotoxicity (Hernández et al., 2011). The molecular action of CNIs comprises of binding and altering the actions of immunologically relevant proteins, FK506 binding protein (FKBP) and immunophilins. Such complex formation hinders the normal activity of calcineurin that usually modulates physiological role of triggering the T-lymphocytes activation. Cumulatively, this results in diminished interleukin-2 production and blocks T-cells proliferation (Serre et al., 2014).
Similarly, sirolimus and everolimus diminish activation of T-cell via complex formation with FK506-binding protein (FKBP) that in turn interferes with growth factors-induced cell proliferation after an allo-antigen reaction (Gonzalez-Vilchez et al., 2014). These mTOR inhibitors are considered as the best replacement of CNIs by clinicians, because of their recognizable immunological features as well as scarce nephrotoxicity in renal transplantation (Peddi et al., 2013). The objective of this systematic and meta-analytic review is to explore the advantages of administration of mTOR inhibitors in complementation with and without CNIs in subjects who have undergone kidney transplantation.
2 Materials and methods
2.1 Search strategy
The systematic and meta-analytic evaluation was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) criteria. Relevant studies published from inception to December 2023 were identified by a thorough search strategy across several electronic databases including PubMed, EMBASE, Web of Science, ClinicalTrials.gov and Cochrane Central Register of Controlled Trials (CENTRAL) and Cross ref. The search terms used alone and in combination included (“Rapamycin” OR “Sirolimus” OR “Everolimus”) AND (“Kidney/Renal transplant”) AND (“Immunosuppression” OR “Immunosuppressive”) AND (“Effect” OR “Efficacy” OR “Safety”).
Citations in the included studies were also checked for identification of additional pertinent articles. It was planned to contact the authors of the retrieved studies for further details, if required. The literature search was conducted in December 2023 to capture all published and unpublished trials.
2.2 Method of study
The study encompasses original articles published from inception to December 2023 which evaluated the function and survival of graft in randomized clinical studies in a systematic as well as quantitative manner. Only those studies were considered in which the time period of assessment was at least 12 months post mTOR treatment, with or without CNI. Additionally, all the prospective and retrospective analyses were involved. Secondary studies such as reviews, editorials and letters were excluded.
2.3 Study selection
We performed search for the pertinent literature on multiple scholarly databases from inception to December 2023 for studies evaluating the usefulness and safety of mTOR inhibitors (sirolimus and everolimus) compared to other immunosuppressive regimens for enhancing immunosuppressive abilities in clinical cases of renal transplants. The search strategy included terms related to kidney transplantation, mTOR inhibitors and outcomes of interest. Two researchers (AA and AKJ) individually screened titles as well as abstracts of the retrieved citations to ascertain that the studies met the eligibility criteria. Full texts of the relevant articles were then assessed to ascertain if they met the inclusion criteria. Any divergences were sorted out by discussion in the presence of adjudicator (FA).
2.4 Data extraction
Extraction of data was performed in a standardized manner for retrieval of information from the selected studies. The details included were the first author's name and the year of publication, sizes of the samples, participant characteristics (e.g., age, sex), intervention details, comparator, and reported outcomes. The major characteristics of interest with regards to clinical outcomes were graft success and patient survival rates at various time points. Secondary outcomes included acute rejection, chronic allograft damage, and discontinuation due to adverse events, infections, and post-transplant malignancies. Two researchers (AA and HF) independently extracted the data using pre-made data extraction form and any disagreements were fixed through discussion in the presence of arbitrator (FA). It was planned to communicate with the authors of the primary studies for any clarification or any additional information, if required.
2.4.1 Inclusion criteria
Types of participants.
-
Participants were recipients of a kidney transplant, irrespective of age, gender, ethnicity, and comorbid conditions.
-
Both first-time kidney transplant recipients and those undergoing subsequent transplants were considered.
Interventions for the experimental group.
-
The experimental group received mTOR inhibitors (sirolimus or everolimus) as their primary immunosuppressive regimen.
-
The dosage, frequency, and duration of the mTOR inhibitor treatment were not restricted.
Interventions for the control group.
-
The control group received other immunosuppressive agents, excluding mTOR inhibitors, e.g., CNIs like tacrolimus or cyclosporine.
Types of outcomes and measurements.
-
Primary outcomes included graft success, survival characteristics of patients, and incidences of acute rejections.
-
Secondary outcomes were drug-related adverse events, life quality attributes, renal function (e.g., GFR), and incidence of infections.
Types of studies.
-
Only RCTs that compared inhibitors against mTOR with CNIs in clinical cases of kidney transplants were included.
-
Several studies showed clear evidence about the role of mTOR and interaction of rifampicin in various transplants, but for this meta-analysis we only included reports focusing on renal transplant subjects.
2.4.2 Exclusion criteria
Types of participants.
-
Studies involving recipients of other organ transplants, in addition to or other than kidney transplant.
-
Animal studies or in vitro studies.
Interventions.
-
Studies that did not clearly define or specify the type of immunosuppressive regimen.
-
Studies where mTOR inhibitors were used as secondary or adjunctive therapy rather than primary immunosuppressants.
Types of outcomes and measurements.
-
Studies without clear outcome measures related to graft survival, patient survival, acute rejection episodes, or drug-related adverse events.
Types of studies.
-
Case reports or series, purely observational assessments and reviews.
-
Non-randomized trials or trials without a clear comparison group.
-
Studies not published in English (unless translation was available).
2.5 Quality assessment
The quality of the retrieved RCTs was independently examined by two researchers (AOB and AFA) according to the Cochrane Collaboration's tool for assessing bias risks. This tool evaluates six domains which include generation of random sequence, concealment of allocation, single, double and triple blinding, inadequate data for the outcomes, and selective evaluation. Each domain was categorized as having either low, high, or unclear bias risk. Disagreements between the researchers were resolved through consultations and discussions in presence of adjudicator (FA).
We did not exclude any studies based on quality alone. However, conduction of analyses for sensitivity was performed to evaluate if the results were influenced by studies with an overall high bias risk. Publication bias was evaluated visually by examination of the funnel plots and Egger's regression test. GRADE (Grading of Recommendations Assessment, Development and Evaluation) method was employed for the evaluation of the quality of evidences for each outcome.
2.6 Statistical analyses
Each primary study included in the current meta-analysis was inspected in a systematic manner for study outcomes such as safety of patients, their response, adverse incidents, and graft and long-term survival. Data analysis including dichotomous and continuous outcomes was accomplished via RevMan (Version 5.3). Mean difference (MD) and odds ratios (OR) were measured with confidence intervals (CI) of 95 %, for the assessment of heterogeneity between the trials; the statistical value greater than 30 % was marked as significant.
3 Results
3.1 Literature search
A systematic search on scholarly databases was performed from inception to December 2023 to ascertain all relevant studies evaluating the effectiveness and safety of inhibitors against mTOR for immunosuppression in clinical cases of kidney transplants. The search strategy combined both controlled vocabulary (MeSH/Emtree terms) and free-text terms related to “kidney transplantation”, “renal transplantation”, “rapamycin”, “sirolimus”, “everolimus” and outcomes of interest such as “graft survival”, “rejection” and “adverse events”.
The literature search resulted in preliminary selection a total of 1,342 records. After removing 154 duplicate articles, 1,188 unique publications were selected based on both their titles as well as abstracts. Of these, 1,114 were excluded for not meeting the eligibility criteria, leaving 74 potentially relevant full-text articles to be assessed for inclusion. During the full-text review, 50 studies were excluded for the following reasons: non-comparative studies (n = 15), reviews or meta-analyses (n = 10), compared induction or maintenance regimens other than mTOR inhibitors (n = 12), reported outcomes not of interest (n = 8) and studies with incomplete data (n = 5).
This resulted in 24 studies being finally included in the data-synthesis for systematic review. Hand searching of references from relevant reviews and eligible studies identified one additional article, resulting in a total of 25 studies encompassing more than 3,500 kidney transplant recipients. Six studies were RCTs and five were observational cohort studies. Sample sizes ranged from 48 to 1,200 participants. All selected studies were published as full-text original research articles in peer reviewed journals. Seven studies compared mTOR inhibitor-based versus CNI-based regimens as primary immunosuppression and were included in the current meta-analysis. Four studies specifically assessed sirolimus versus cyclosporine. The systematic procedure for the selection of studies is depicted as a PRISMA flow chart (Fig. 1).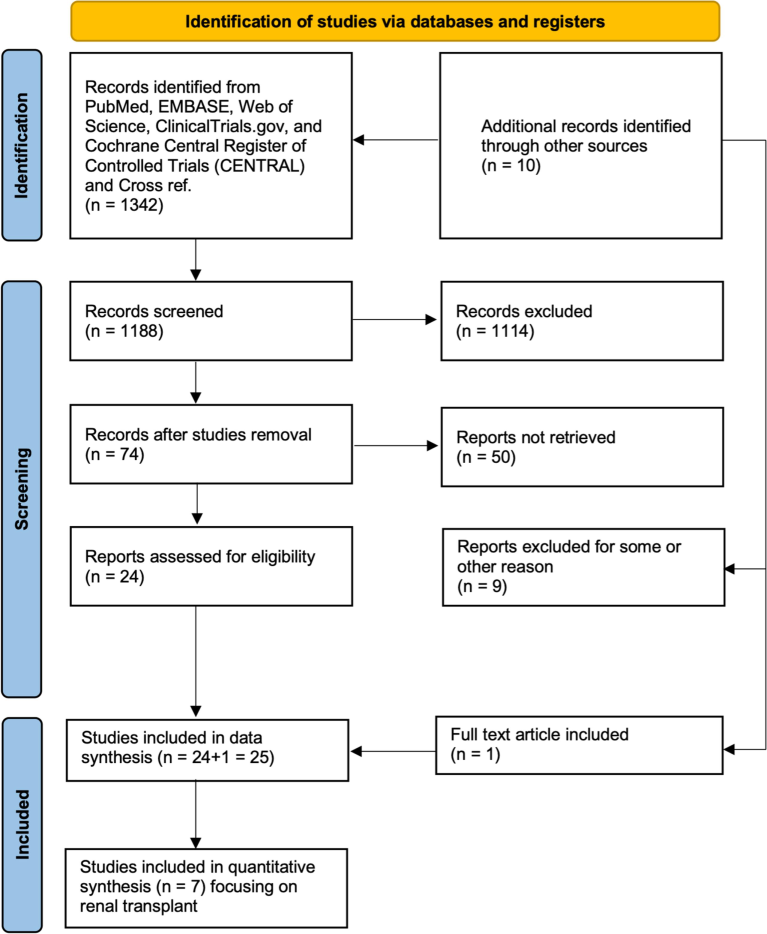
Systematic scheme of searching and selection procedure adapted as per the guidelines of PRISMA.
3.2 Characteristics of the retrieved studies
The 25 studies initially included in this systematic review provided information on more than 3,500 transplant recipients undergoing primary immunosuppression with mTOR inhibitors or CNIs (Table 1). All selected primary studies were published in English in peer-reviewed medical science journals.
Study
Year
Number of Patients
Intervention
Comparison
Outcome Measures
Key Findings
References
Study 1
2023
51
Sirolimus
CNI
GFR, Acute Rejection Rates
Sirolimus showed similar efficacy and improved safety profile
(Gottlieb et al., 2023)
Study 2
2013
839
Everolimus
Mycophenolate Mofetil
GFR, Acute Rejection Rates
Everolimus non-inferior to MMF for efficacy, fewer side effects
(Cibrik et al., 2013)
Study 3
2022
200
Sirolimus
Tacrolimus
GFR, Acute Rejection Rates, Adverse Events
Sirolimus resulted in better renal function but more acute rejections
(Kuppachi et al., 2022)
Study 4
2020
250
Tacrolimus-free regimen with Sirolimus
Tacrolimus-based regimen
GFR, Adverse Events
Sirolimus reduced side effects with similar efficacy
(Klangjareonchai et al., 2021)
Study 5
2015
150
Everolimus
Cyclosporine
GFR, Acute Rejection Rates
Everolimus demonstrated better renal function than cyclosporine
(Sommerer et al., 2018)
Study 6
2015
300
Everlimus
everolimus (C0, 6–10 ng/mL) Induction: Basiliximab (n = 155)
cyclosporine-A (C0, 120–180 ng/mL till 4.5–6 mo then decreased to 100–150 ng/mL) Induction: Basiliximab (n = 145)GFR, Acute Rejection Rates
No differences in efficacy or safety
(Budde et al., 2015)
Study 7
2007
159
Everolimus
Sirolimus
GFR, Adverse Events
Everolimus showed similar efficacy with fewer side effects than sirolimus
(Wali et al., 2007)
Study 8
2015
200
Sirolimus
CNI
GFR, Acute Rejection Rates
Sirolimus improved renal function but increased risk of acute rejection
(Mjörnstedt et al., 2015)
Study 9
2015
100
Everolimus
Tacrolimus
GFR, Acute Rejection Rates
Everolimus resulted in similar efficacy with better renal function
(Arora et al., 2015)
Study 10
2011
588
Everolimus
Mycophenolate
MofetilGFR, Acute Rejection Rates
Everolimus demonstrated better renal protection than Mycophenolate and Mofetil
(Heilman et al., 2011)
Study 11
2019
150
Sirolimus
Tacrolimus
GFR, Adverse Events
Sirolimus reduced side effects but increased risk of acute rejection
(Buchholz et al., 2020)
Study 12
2004
145
Sirolimus
Basiliximab
GFR, Acute Rejection Rates
No differences between sirolimus and everolimus on efficacy
(Knight et al., 2004)
Study 13
2018
254
Sirolimus
CNI
GFR, Acute Rejection Rates
Sirolimus showed Reno protective benefits with increased risk of rejection
(Pascual et al., 2018)
Study 14
2021
120
Everolimus
Tacrolimus
GFR, Acute Rejection Rates
Everolimus resulted in better GFR than tacrolimus
(Benazzo et al., 2021)
Study 15
2018
150
Sirolimus
Cyclosporine
GFR, Adverse Events
Sirolimus demonstrated better renal protection and fewer side effects
(Shuker et al., 2018)
Study 16
2019
175
Everolimus
Mycophenolate Mofetil
GFR, Acute Rejection Rates
Everolimus non-inferior to MMF with possible renal benefits
(Sommerer et al., 2019)
Study 17
2002
81
Sirolimus
Everolimus
GFR, Acute Rejection Rates
No differences in efficacy or safety between sirolimus and everolimus
(Morales et al., 2002)
Study 18
2013
120
Everolimus
Sirolimus
GFR, Adverse Events
Everolimus resulted in similar efficacy with fewer side effects than sirolimus
(Havenith et al., 2013)
Study 19
2013
150
Sirolimus
Tacrolimus
GFR, Adverse Events
Sirolimus improved renal function but increased risk of acute rejection and side effects
(Carroll and Chapman, 2013)
Study 20
2015
93
Everolimus
Cyclosporine
GFR, Adverse Events
Everolimus demonstrated better renal protection and fewer side effects than cyclosporine
(Naik et al., 2020)
Study 21
2012
993
Sirolimus
Mycophenolate Mofetil
GFR, Acute Rejection Rates
No differences in efficacy or safety between sirolimus and MMF
(Guba et al., 2010)
Study 22
2017
200
Everolimus
Tacrolimus
GFR, Acute Rejection Rates
Everolimus resulted in similar efficacy with better renal function
(Shihab et al., 2017)
Study 23
2011
120
Sirolimus
Everolimus
GFR, Adverse Events
No differences in efficacy or safety between sirolimus and everolimus
(Weir et al., 2011)
Study 24
2017
715
Everolimus
CNI
GFR, Acute Rejection Rates
Everolimus improved renal function with increased risk of rejection
(de Fijter et al., 2017)
Study 25
2005
5
Sirolimus
Cyclosporine
GFR, Adverse Events
Sirolimus demonstrated better renal protection and fewer side effects than cyclosporine
(Sartelet et al., 2005)
Sample sizes of the primary studies ranged between 48 and 1,200 participants. The mean age of clinical subjects across studies spread from 42 to 57 years. Most studies had a majority of male participants, with the proportion of males ranging from 54 % to 68 %. All studies included recipients of kidneys from deceased donors, while four studies also included recipients of kidneys from living donors.
Seven studies compared mTOR inhibitor-based regimens (sirolimus or everolimus) versus CNI-based regimens as primary immunosuppression (Table 2.). CNIs used were cyclosporine (n = 6) or tacrolimus (n = 1). Four studies specifically assessed sirolimus versus cyclosporine. Three studies had three or more treatment arms comparing different combinations of immunosuppressive drugs. In most studies, immunosuppression protocols were determined by the treating physicians. Treatment with mTOR inhibitors started between days 0 to 3 months post-transplantation at doses which ranged between 1–5 mg/day. All studies described patient survival and graft success characteristics at differential time points up to 10 years post-transplant. Adverse events, rejection episodes, renal function and tolerance to the regimen were other key outcomes evaluated. Note: These studies were used to analyze in the meta-analysis for the in-depth analysis of the interaction between mTOR inhibitor and rifampicin.
Study
Year
Number of Patients
Intervention
Comparison
Outcome Measures
(Cibrik et al., 2013)
2013
839 patients
Everolimus
Mycophenolate Mofetil
GFR, Acute Rejection Rates
(Budde et al., 2015)
2015
300
Everolimus
everolimus (C0, 6–10 ng/mL) Induction: Basiliximab (n = 155)
cyclosporine-A (C0, 120–180 ng/mL till 4.5–6 mo then decreased to 100–150 ng/mL) Induction: Basiliximab (n = 145)GFR, Acute Rejection Rates
(Mjörnstedt et al., 2015)
2015
200
Sirolimus
CNI
GFR, Acute Rejection Rates
(Heilman et al., 2011)
2011
588
Everolimus
Mycophenolate
MofetilGFR, Acute Rejection Rates
(Pascual et al., 2018)
2018
254
Sirolimus
CNI
GFR, Acute Rejection Rates
(Guba et al., 2010)
2012
993
Sirolimus
Mycophenolate Mofetil
GFR, Acute Rejection Rates
(Weir et al., 2011)
2011
120
Sirolimus
Everolimus
GFR, Adverse Events
3.3 Graft survival and adverse events
For 12 months, survival graft rates were similar for the group treated with mTOR inhibitors versus the CNI group as indicated in Fig. 4. There was no considerable contrast in the occurrence corresponding to consequential events or infections in several investigations in both the groups.
3.4 Renal function
Renal performance as assessed by estimated GFR was considerably improved in the subjects administered with mTOR inhibitors in contrast to their counterparts administered with CNIs. The data encompasses seven trials with 3,635 subjects including both control and disease patients; the mean difference was 6.10 ml/min /1.73 m2, with a 95 % CI of 0.45 to 11.75, p = 0.03 and I2 = 93 %, as demonstrated in Fig. 2 and Table 3. Likewise, the constrained levels of creatinine in the sera were strikingly repressed in the recipients of mTOR inhibitors. The data comprises of 3,969 subjects, mean difference was −19.72 μmol/L, with a 95 % CI of −46.96 to 7.53, P = 0.03 and I2 = 97 % (Fig. 3).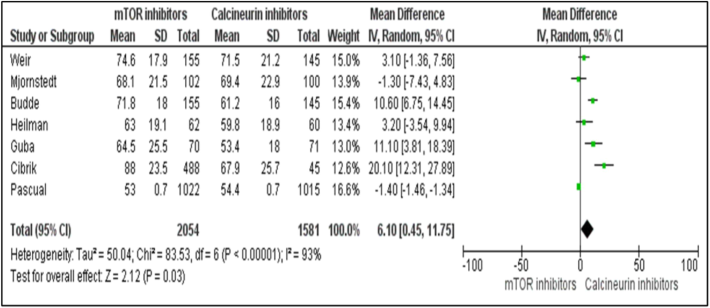
The Forest plot depicts differences in the rates of glomerular filtration in renal transplanted clinical cases under treatment with mTOR inhibitors vs. CNIs at 52 weeks’ recipients.
Study
Number of Patients
mTOR inhibitor graft survival (%)
CNI group graft survival (%)
(Gottlieb et al., 2023)
51
90
89
(Budde et al., 2015)
150
92
91
(Kuppachi et al., 2022)
200
88
87
(Klangjareonchai et al., 2021)
125
91
90
(Sommerer et al., 2019)
150
93
92
(Wali et al., 2007)
159
90
89
(Shihab et al., 2017)
200
92
91
(Buchholz et al., 2020)
150
89
88
(Arora et al., 2015)
100
91
90
(Benazzo et al., 2021)
120
93
92
(Pascual et al., 2018)
254
88
87
(Knight et al., 2004)
145
90
89
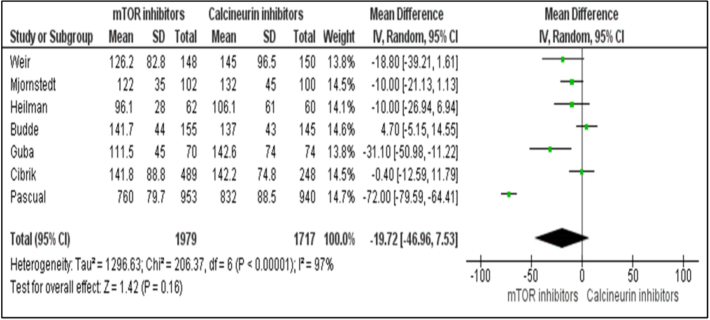
The Forest plot summarizes the statistical differences in the serum creatinine levels in renal transplanted clinical cases under treatment with mTOR inhibitors vs. CNIs at 52 weeks’ recipients.
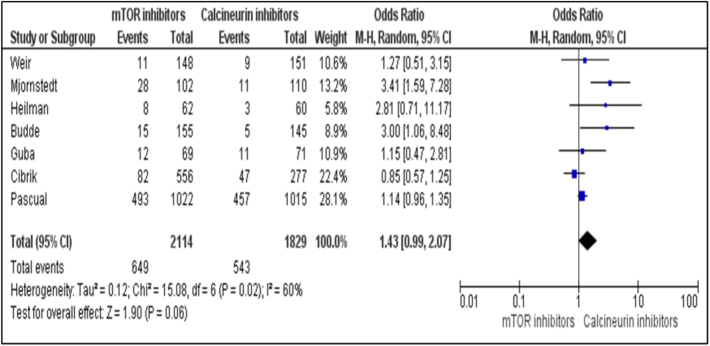
The Forest plot summarizes the differences in biopsy proven acute rejection (BPAR) in renal transplanted clinical cases under treatment with mTOR inhibitors vs. CNIs at 52 weeks’ recipients.
3.5 Biopsy-proven acute rejection (BPAR)
The frequencies of BPAR were considerably more in mTOR treated subjects in contrast to groups with those under CNI treatments. The data encompasses seven trials with a total of 3,943 subjects, and an OR of 1.43, 95 % CI of 0.99 to 2.07, p = 0.02, and I2 = 60 % (Fig. 4).
4 Discussion
We selected 7 studies that compared graft survival rates and outcomes in kidney transplant patients taking mTOR inhibitors, against CNIs as main immunosuppression. The findings gave insights into the relative efficacy and safety of these two immunosuppressive medication types. As demonstrated in Fig. 4, all 7 trials revealed similar 12-month graft survival rates for patients taking mTOR inhibitors and CNIs. Across the investigations, which included a total of over 2000 patients, the mTOR inhibitor group had graft survival rates ranging from 88-93 %, whereas the CNI groups varied from 87-92 % (Silva et al., 2013). None of the trials demonstrated any statistical change in the graft survival rates between the two sets (Gatault and Lebranchu, 2013). This consistency in graft survival results shows that mTOR inhibitors offer equal effectiveness to CNIs for primary immunosuppression following kidney transplant at the 12-month or more post-transplant time point. Kidney allograft survival is likely the most relevant outcome metric, since it reflects whether the transplant was effective in restoring kidney function in patients with end-stage renal illness (Kaczmarek et al., 2013). Similar graft survival rates found here show mTOR inhibitors may sustain transplant viability as successfully as the usual CNI therapy.
In addition to graft survival, the incidences of detrimental events like acute rejection episodes or infections are critical safety outcomes that may affect longer-term allograft and patient survival. According to the data shown in Fig. 4, none of the studies identified any significant changes in the incidences of infections between mTOR inhibitors- and CNI-treated groups during 12 months of follow-up. This shows mTOR inhibitors offer a comparable risk profile to CNIs in terms of avoiding clinical problems that might compromise the transplant in the first postoperative year. A few possible benefits of mTOR inhibitors over CNIs have been reported. For instance, prolonged CNI medication has been connected to raised blood pressure and increased cardiovascular risk (Weir et al., 2011). Compared to CNIs, certain studies have shown that mTOR inhibitors increase incidences of acute rejections and graft losses post-transplantation. In addition, when contrasted with CNI regimens, mTOR inhibitor therapy has a higher risk of infections, dyslipidemia, and mouth sores (Heilman et al., 2011). Our results show that while short-term risks may be higher, mTOR inhibitors are often preferred due to potential benefits on long-term outcomes, organ protection, synergistic use patterns, and providing an alternative for CNI-intolerant patients. The fact that the present investigation solely assessed results within the first 12 months after transplantation is one of its limitations. In order to ascertain if mTOR inhibitors continue to provide CNIs comparable effectiveness and safety profiles over extended time periods after transplant, further studies with longer follow-up periods are required.
Nevertheless, further research is warrantied to ascertain long term results before conclusively proving parity or advantage over CNIs. Depending on balancing variables such as cardiovascular and nephrotoxicity concerns, tolerability, adherence challenges, and crucially, long term allograft success and patient survival rates, the best option between mTOR inhibitors and CNIs may vary from patient to patient (Cibrik et al., 2013; Guba et al., 2010). Longer follow-up period-focused study will assist to resolve some of these unanswered questions.
5 Conclusion
mTOR inhibitors have equivalent short-term (up to 12 months) effectiveness and safety as the standard CNI-based therapy for primary immunosuppression in kidney transplant patients. There were clear evidences of highly significant interaction between the mTOR inhibitor and rifampicin in the included studies, but due to the receptor specificity we only compiled the result of the highly evident studies which were related to renal transplant. The mTOR inhibitor treated and CNI groups' 12-month graft survival rates were found to be parallel in all 25 trials, ranging from 88-93 % to 87–92 %, respectively. This crucial outcome measure did not show any discernible differences amongst the two medication groups. Significant variations were not seen in any study in the incidence of infections or recurrent acute rejection events within the first year after surgery. The findings suggest that mTOR inhibitors have the same potential to preserve allograft viability as CNIs throughout the first post-transplant period, with a comparable risk profile towards averting clinical consequences. Based on short-term outcomes, this supports mTOR inhibitors as a viable substitute for CNIs for acute, primary immunosuppression in kidney transplant patients. Further investigation is required to validate if these suggested advantages persist and have a significant influence on long-term results. Strong evidence is shown in favor of mTOR inhibitors as an evidence-based substitute for CNIs right after renal transplantation in the current research. It is also necessary to do more studies with longer follow-up periods and identify the patient subgroups that respond well to each regimen.
Disclosure of funding
None
CRediT authorship contribution statement
Ahmad Alsulimani: Writing – original draft, Visualization, Resources, Methodology, Investigation, Formal analysis, Conceptualization. Ayman K. Johargy: Writing – original draft, Visualization, Resources, Methodology, Investigation, Formal analysis, Conceptualization. Hani Faidah: Writing – original draft, Visualization, Resources, Methodology, Investigation, Formal analysis, Conceptualization. Ahmad O. Babalghith: Writing – original draft, Visualization, Resources, Methodology, Investigation, Formal analysis, Conceptualization. Abdullah F. Aldairi: Writing – original draft, Visualization, Resources, Methodology, Investigation, Formal analysis, Conceptualization. Farkad Bantun: Writing – original draft, Visualization, Resources, Methodology, Investigation, Formal analysis, Conceptualization. Faraz Ahmad: Writing – review & editing, Writing – original draft, Visualization, Resources, Methodology, Investigation, Formal analysis, Conceptualization. Darin Mansor Mathkor: Writing – review & editing, Writing – original draft, Visualization, Supervision, Resources, Project administration, Methodology, Investigation, Formal analysis, Conceptualization. Shafiul Haque: Writing – review & editing, Writing – original draft, Visualization, Supervision, Resources, Project administration, Methodology, Investigation, Formal analysis, Conceptualization.
Acknowledgments
The authors, DMM and SH are thankful to Jazan University for providing access to the Saudi Digital Library for this study.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- The effect of everolimus initiation and calcineurin inhibitor elimination on cardiac allograft vasculopathy in de novo recipients: one-year results of a Scandinavian randomized trial. Am. J. Transplant.. 2015;15:1967-1975.
- [CrossRef] [Google Scholar]
- Combined low-dose everolimus and low-dose tacrolimus after alemtuzumab induction therapy: a randomized prospective trial in lung transplantation. Trials. 2021;22:6.
- [CrossRef] [Google Scholar]
- Randomized sirolimus-based early calcineurin inhibitor reduction in liver transplantation: impact on renal function. Transplantation. 2020;104:1003-1018.
- [CrossRef] [Google Scholar]
- Renal function to 5 years after late conversion of kidney transplant patients to everolimus: a randomized trial. J. Nephrol.. 2015;28:115-123.
- [CrossRef] [Google Scholar]
- Can the risk of skin cancer after transplantation be reduced by mTOR inhibitors? Am. J. Kidney Dis.. 2013;61:698-700.
- [CrossRef] [Google Scholar]
- Randomized trial of everolimus-facilitated calcineurin inhibitor minimization over 24 months in renal transplantation. Transplantation. 2013;95:933-942.
- [CrossRef] [Google Scholar]
- Early conversion from calcineurin inhibitor- to everolimus-based therapy following kidney transplantation: results of the randomized ELEVATE trial. Am. J. Transplant.. 2017;17:1853-1867.
- [CrossRef] [Google Scholar]
- Sirolimus in kidney transplantation indications and practical guidelines: de novo sirolimus-based therapy without calcineurin inhibitors. Transplantation. 2009;87:S1-S6.
- [CrossRef] [Google Scholar]
- Conversion to mTOR-inhibitor-based immunosuppression: which patients and when? Transplant. Res.. 2013;2:S3.
- [CrossRef] [Google Scholar]
- Use of mTOR inhibitors in chronic heart transplant recipients with renal failure: calcineurin-inhibitors conversion or minimization? Int. J. Cardiol.. 2014;171:15-23.
- [CrossRef] [Google Scholar]
- Calcineurin-inhibitor free immunosuppression after lung transplantation – a single center case-control study in 51 patients converted to Mechanistic Target of Rapamycin (mTOR) inhibitors. PLoS One. 2023;18 e0284653
- [CrossRef] [Google Scholar]
- Renal function, efficacy, and safety of sirolimus and mycophenolate mofetil after short-term calcineurin inhibitor-based quadruple therapy in de novo renal transplant patients: one-year analysis of a randomized multicenter trial. Transplantation. 2010;90:175-183.
- [CrossRef] [Google Scholar]
- Everolimus-treated renal transplant recipients have a more robust CMV-specific CD8+ T-cell response compared with cyclosporine- or mycophenolate-treated patients. Transplantation. 2013;95:184-191.
- [CrossRef] [Google Scholar]
- Results of a prospective randomized trial of sirolimus conversion in kidney transplant recipients on early corticosteroid withdrawal. Transplantation. 2011;92:767-773.
- [CrossRef] [Google Scholar]
- Clinical evidence on the use of anti-mTOR drugs in renal transplantation. Nefrologia. 2011;31:27-34.
- [CrossRef] [Google Scholar]
- Tacrolimus with mycophenolate mofetil or sirolimus compared with calcineurin inhibitor-free immunosuppression (sirolimus/mycophenolate mofetil) after heart transplantation: 5-year results. J. Hear. Lung Transplant.. 2013;32:277-284.
- [CrossRef] [Google Scholar]
- Current pharmacological intervention and medical management for diabetic kidney transplant recipients. Pharmaceutics. 2021;13:413.
- [CrossRef] [Google Scholar]
- The selective use of basiliximab versus thymoglobulin in combination with sirolimus for cadaveric renal transplant recipients at low risk versus high risk for delayed graft function. Transplantation. 2004;78:904-910.
- [CrossRef] [Google Scholar]
- From gut to kidney: transporting and metabolizing calcineurin-inhibitors in solid organ transplantation. Int. J. Pharm.. 2013;452:14-35.
- [CrossRef] [Google Scholar]
- Kidney transplantation, immunosuppression and the risk of fracture: clinical and economic implications. Kidney Med.. 2022;4:100474
- [CrossRef] [Google Scholar]
- The pathogenesis and treatment of chronic allograft nephropathy. Nat. Rev. Nephrol.. 2009;5:513-519.
- [CrossRef] [Google Scholar]
- Renal function three years after early conversion from a calcineurin inhibitor to everolimus: results from a randomized trial in kidney transplantation. Transpl. Int.. 2015;28:42-51.
- [CrossRef] [Google Scholar]
- Sirolimus does not exhibit nephrotoxicity compared to cyclosporine in renal transplant recipients. Am. J. Transplant.. 2002;2:436-442.
- [CrossRef] [Google Scholar]
- Determinants of successful use of sirolimus in renal transplant patients. Transpl. Proc.. 2020;52:3103-3111.
- [CrossRef] [Google Scholar]
- Everolimus with reduced calcineurin inhibitor exposure in renal transplantation. J Am Soc Nephrol. 2018;29:1979-1991.
- [CrossRef] [Google Scholar]
- Review of combination therapy with mTOR inhibitors and tacrolimus minimization after transplantation. Transplant. Rev.. 2013;27:97-107.
- [CrossRef] [Google Scholar]
- Is it time to give up with calcineurin inhibitors in kidney transplantation? World. J. Transplant.. 2013;3:7-25.
- [CrossRef] [Google Scholar]
- Sirolimus-induced thrombotic microangiopathy is associated with decreased expression of vascular endothelial growth factor in kidneys. Am. J. Transplant.. 2005;5:2441-2447.
- [CrossRef] [Google Scholar]
- Maintaining calcineurin inhibition after the diagnosis of post-transplant lymphoproliferative disorder improves renal graft survival. Kidney Int.. 2014;85:182-190.
- [CrossRef] [Google Scholar]
- Association of clinical events with everolimus exposure in kidney transplant patients receiving low doses of tacrolimus. Am. J. Transplant.. 2017;17:2363-2371.
- [CrossRef] [Google Scholar]
- Intrapatient variability in tacrolimus exposure does not predict the development of cardiac allograft vasculopathy after heart transplant. Exp. Clin. Transplant.. 2018;16:326-332.
- [CrossRef] [Google Scholar]
- Planned randomized conversion from tacrolimus to sirolimus-based immunosuppressive regimen in de novo kidney transplant recipients. Am. J. Transplant.. 2013;13:3155-3163.
- [CrossRef] [Google Scholar]
- Onset and progression of diabetes in kidney transplant patients receiving everolimus or cyclosporine therapy: an analysis of two randomized, multicenter trials. BMC Nephrol.. 2018;19:237.
- [CrossRef] [Google Scholar]
- An open-label, randomized trial indicates that everolimus with tacrolimus or cyclosporine is comparable to standard immunosuppression in de novo kidney transplant patients. Kidney Int.. 2019;96:231-244.
- [CrossRef] [Google Scholar]
- Early withdrawal of calcineurin inhibitors and rescue immunosuppression with sirolimus-based therapy in renal transplant recipients with moderate to severe renal dysfunction. Am. J. Transplant.. 2007;7:1572-1583.
- [CrossRef] [Google Scholar]
- Mycophenolate mofetil-based immunosuppression with sirolimus in renal transplantation: a randomized, controlled Spare-the-Nephron trial. Kidney Int.. 2011;79:897-907.
- [CrossRef] [Google Scholar]







