Translate this page into:
Network pharmacology and molecular docking analysis of Catharanthus roseus compounds: Implications for non-small cell lung cancer treatment
⁎Corresponding author. mo.saeed@uoh.edu.sa (Mohd Saeed)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The prevalence of non-small cell lung cancer (NSCLC), particularly in the Eastern Region of Saudi Arabia, is concerning and highlights the need for novel treatments, despite recent progress in NSCLC treatment. Plant-derived natural compounds, such as those found in Catharanthus roseus, have shown promising results in cancer research. By employing cutting-edge computational analysis techniques, such as molecular docking and network pharmacology, the study aimed to understand the mechanism of action of compounds isolated from C. roseus. The study identified several bioactive and drug-like compounds in C. roseus that displayed diverse interactions with important proteins associated with cancer. The compounds PubChem CIDs 5316490, 425978, and 261578 were found to form significant interactions within the binding site of the B-cell-lymphoma-2 (BCL-2) protein. Compound 5316490, in particular, interacted with critical residues within the target protein, highlighting its potential as a therapeutic agent against lung cancer. The understanding of the molecular targets and mechanism of action of C. roseus bioactive compounds obtained from this study provides hope for improving current treatment options. In conclusion, the findings provide additional details on the evidence supporting the potential of C. roseus as a treatment for NSCLC and suggest that further research could lead to the development of innovative, potent therapies.
Keywords
Catharanthus roseus
BCL-2 receptor
Non-small cell lung cancer (NSCLC)
Network pharmacology
Molecular docking
1 Introduction
Despite significant advancements in both therapeutic and preventive measures, cancer continues to pose a significant global health challenge. Cancerous growths, known as tumors, can spread to various anatomical sites and are characterized by uncontrolled cellular proliferation. While modern therapeutic approaches, such as chemotherapy, radiation therapy, and pharmaceuticals, are effective in treating a range of illnesses, they often come with severe side effects that can negatively impact the overall health of a patient. As a result, there is a growing interest in finding alternative cancer treatments and therapies that are highly effective and have low toxicity (Ochwang'i, 2014).
B-cell lymphoma-2 (Bcl-2) is a crucial protein that is a member of the Bcl-2 family. The influence of this protein on non-small cell lung cancer (NSCLC) is substantial. In human NSCLC models, its overexpression has been linked to both resistance to conventional chemotherapy and the induction of tumor growth (Choi, 2005). Recent studies indicate that the inhibition of Bcl-2 can reverse the development of drug resistance that has been observed in numerous cancer cell lines (Sun et al., 2017). A meta-analysis of different types of cancers revealed that Bcl-2 expression varied, indicating that the expression level of Bcl-2 in cancers is complex and that its impact on patient outcomes is multifaceted. The complex role of Bcl-2 in apoptosis control demonstrates its importance as a therapeutic target in the fight against NSCLC (Hwang, 2001).
The occurrence of lung cancer has risen in the Eastern Region of Saudi Arabia, necessitating the implementation of management, screening, and prevention approaches. Given that over half of the Saudi Arabian population is under 29 years of age, initiatives have been undertaken to evaluate cancer incidence and mortality rates within the country. The aim of these endeavors is to devise efficient methods for managing this disease. Lung cancer patients often have a dismal prognosis, despite the progress made in the detection and treatment of the disease. This underscores the need for innovative therapeutic strategies. The investigation of natural compounds obtained from medicinal plants as potential anticancer agents has gained attraction among investigators (Dhyani, 2022; Mishra, 2023).
Catharanthus roseus, previously identified as Vinca rosea L. and classified under the Apocynaceae family, presents itself as a captivating botanical specimen within this particular framework (Kumar et al., 2022). C. roseus possesses a significant historical background within the realm of traditional medicine, which suggests it has been utilized to effectively address a diverse range of health issues such as diabetes, cancer, hypertension, fever, and hemostasis (Kumar et al., 2022). According to estimates, C. roseus is believed to possess around 120 terpenoid indole alkaloids, many of which demonstrate noteworthy therapeutic properties (Amaral, 2017).
Given its broad therapeutic significance, we wish to examine the anticancer properties of phytochemicals found in C. roseus, with a particular emphasis on lung cancer and the BCL-2 protein. To accomplish this, advanced computational biology tools will be used to investigate the molecular impact and therapeutic mechanisms of compounds obtained from C. roseus. By gaining a deeper understanding of the compound's interaction with the BCL-2 protein, we may be able to design new and effective treatments for this life-threatening illness. We wish to use network pharmacology and molecular docking to identify how bioactive compounds interact with the target protein. The ultimate goal is to slow cancer progression and improve treatment outcomes by combining traditional herbal remedies with modern approaches.
2 Material and methods
2.1 Computational analysis of C. roseus compounds for bioactivity
The three-dimensional structure of C. roseus compounds in “.sdf” format was retrieved through the Indian Medicinal Plants, Phytochemistry, and Therapeutics 2.0 (IMPPAT 2.0) (Vivek-Ananth, 2023), and PubChem. The Molinspiration tool (https://molinspiration.com/cgi/properties) was used to evaluate bioactivity of C. roseus compounds. The structure of the compounds was retrieved in.sdf format and prepared for further docking analysis using Discovery Studio (DS) [Dassault Systems, BIOVIA Corp., San Diego, CA, USA, v 21.1].
2.2 Pharmacokinetic and toxicological assessment (ADMET Analysis)
The ADMET prediction analysis of the compounds derived from C. roseus was conducted using the ADMET and Lipinski Rule modules of DS, with the primary objective of assessing the pharmacokinetic and pharmacodynamic properties of these compounds (Ferreira and Andricopulo, 2019).
2.3 Targets prediction for C. roseus compounds
The Swiss Target Prediction tool was employed to predict the targets for the bioactive compounds obtained from C. roseus (Daina et al., 2019). This method enables us to understand these compounds and investigate their therapeutic potential by exploring specific targets. These target proteins serve to identify key compounds and their biological functions.
2.4 Molecular docking study
Molecular docking explores how small molecules interact with the target protein at its binding sites. It serves as a foundational computational tool in drug design (Agu, 2023). In this study, molecular docking studies play a crucial role in predicting binding modes and affinities of the C. roseus compounds with the BCL-2 protein, significantly contributing to understanding their potential pharmacological activities.
2.4.1 Receptor preparation
The three-dimensional conformation of the protein BCL-2 (PDB ID: 6O0K_A), which is associated with B-cell lymphoma 2, was obtained from the RCSB Protein Data Bank (Burley, 2020). The structure consists of a total of 166 amino acid residues. The BCL-2 structure was solved by X-ray crystallography at a resolution of 1.62 Å. The structure is bound with Venetoclax in its binding pocket, whereas aonaethylene glycol is bound in another pocket. The obtained structure was prepared by removing water molecules, co-factors, and ligands irrelevant to the study. Hydrogen atoms were added, and the protonation state of the ionizable residues was adjusted to match physiological conditions. The iGEMDock tool (Hsu, 2011) was used to perform the molecular docking analysis and evaluate the potential interaction between 6O0K_A and the filtered compounds from C. roseus.
2.4.2 Ligand preparation
The compounds obtained from C. roseus were prepared for molecular docking studies by optimization and refinement of the ligands. This process enables the compound to adopt suitable conformation and other properties accurate and reliable for molecular docking (Kumar et al., 2022).
2.4.3 Binding pocket selection
The BCL-2 protein binding pocket was carefully chosen to achieve biologically significant results. Our study focused on the area where Venetoclax, an FDA-approved medication, had previously shown binding properties (Chandrasekar, 2022). The coordinates defining the binding pocket are x = –15.323057, y = 2.213738, and z = −9.593467, with a sphere radius of 13.647127 Å.
2.4.4 Docking analysis
Docking analysis was performed utilizing the iGEMDock tool to investigate the interaction between the C. roseus compounds and the BCL-2 protein (PDB ID 6O0k_A). The prediction and characterization of potential binding modes between the C. roseus compounds and the BCL-2 protein involved the assessment of various parameters such as intermolecular interactions, conformational changes, and binding energy.
2.4.5 Scoring and analysis
Following the docking using the iGEMDock tool, the complexes were ranked and scored based on various parameters, including binding energy, van der Waal forces, hydrogen bonding, hydrophobic interactions, and other intermolecular forces. The resulting complexes were analyzed to identify potential ligands with strong binding affinity and favorable interactions with the BCL-2 protein.
3 Results
3.1 Computational analysis of C. roseus compounds for bioactivity
Molecular docking explores the interaction of ligands within the binding pocket of a protein, which requires binding energy and van der Waals (VDW) forces (Agu, 2023). Understanding and improving these interactions is critical to the development of novel treatments and drug discovery, given that precise ligand–protein interactions can result in powerful and specific molecules with therapeutic promise. Table S1 shows that the binding energy of the docked compounds from C. roseus ranges from 1893.88 to −180.2 kJ/mole.
Table S2 provides an overview of the bioactivity scores for each compound derived from C. roseus, as determined by the Molinspiration tool. These scores were calculated for various target classes, including G protein-coupled receptor (GPCR) ligand, Ion Channel Modulator (ICM), Kinase Inhibitor (KI), Nuclear Receptor Ligand (NRL), Protease Inhibitor (PI), and Enzyme Inhibitor (EI). The scores represent the capacity of each compound to interact with and regulate the activity of specific biological targets. The Molinspiration tool classifies biological activity scores into different parameters, offering details about the potential activity of compounds. Compounds with scores greater than 0 are expected to exhibit significant biological activity, while scores in the range of −0.5 to 0 indicate moderate activity, suggesting a lower but measurable level of interaction. Conversely, compounds with scores below −0.5 are considered inactive, indicating that they do not display the expected biological activity (Yergaliyeva, 2022). The biological activity scores play a significant role in the classification of compounds based on their potential activity or efficacy, as well as in determining the suitability of these compounds for inclusion in drug development trials.
The compounds derived from C. roseus displayed a range of bioactivity scores for GPCR ligands, spanning from −3.3 to 0.5. Several compounds showed significant interactions with GPCR ligands. Similarly, a wide range of bioactivity scores were also recorded for ICM (−3.64 to 1.02). The bioactivity ratings of KI varied from −3.66 to −0.07. C. roseus compounds, specifically 5315229, exhibited promising bioactivity scores, particularly as an ICM and a KI, with values of 0.41 and 0.23, respectively. The NRL scores observed in this study exhibited a range from −3.58 to 0.78, indicating the affinities of C. roseus compounds for nuclear receptors. The bioactivity scores of PI varied from −3.0 to −0.02. The observed scores for EI varied from −3.52 to 0.61. Compound A demonstrated significant bioactivity, as evidenced by its EI score of 0.3. Compound 5316490 displayed diverse bioactivity scores among different target classes. It scored 0.23 as a GPCR Ligand. Positive values were also seen in ICM (1.02), NRL (0.28), and EI (0.3). Compound 425978 has a 0.36 ICM and 0.4 NRL bioactivity score. Compound 261578 scored 0.42 and 0.27 in ICM and KI, respectively. GPCR and KI scores for Compound 162971130 were 0.33 and 0.23, respectively. The GPCR Ligand (0.3) and KI (0.07) scores of Compound 162963996 were also positive (Fig. 1).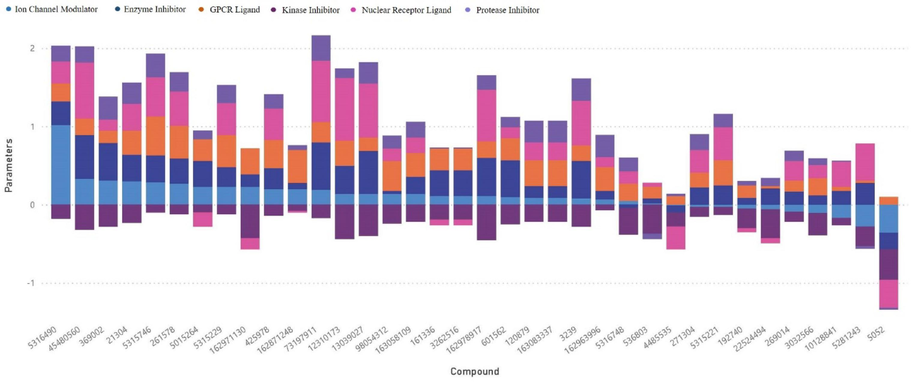
Selection of Optimal Compounds: Balancing Bioactivity and ADMET Filtering. This histogram showcases the process of refining compounds from C. roseus by combining bioactivity assessment with ADMET (Absorption, Distribution, Metabolism, Excretion, and Toxicity) filtering.
3.2 Pharmacokinetic and toxicological assessment (ADMET Analysis)
Table 2 summarizes the results of the ADMET properties analysis and presents details on the properties of the bioactive compounds that are obtained from C. roseus. Some compounds such as 3239, 192740, 261578, 425978, 536803, 3032566, 4485535, 5315221, 5315229, 5315746, and 531971130, have good solubility and bioavailability. Compounds like 120879, 5316748, 12310173, 98054312, 162871248, 162963996, and 163083337 have intermediate solubility. C. roseus compounds 3239, 21304, 120879, 192740, 261578, 425978, 536803, 3032566, 4485535, 5315221, 5315229, 5315746, 5316490, 162871248, 162963996, 162971130, and 163058109 have limited blood–brain barrier permeability, which may restrict their ability to cross the barrier. Most compounds were not found to inhibit CYP2D6. Compound 45480560 may inhibit CYP2D6, affecting medication, metabolism and interactions with other drugs. Compounds 3239, 120879, 192740, 425978, 536803, 4485535, 5315221, 5315229, 5315746, 162963996, 163058109, and 5316490 were found to have better safety profiles and be less hepatotoxic. Most compounds were estimated to have limited absorption. In contrast, compound 45480560 exhibits a higher absorption capacity. An analysis of plasma protein binding (PPB) shows that many compounds have a binding affinity for plasma proteins, which may affect the distribution and bioavailability of these proteins. A significant portion of the compounds that were evaluated by ADMET had positive safety profiles, suggesting a lower probability of toxicity. It is noteworthy that most of the compounds fulfilled many toxicity criteria, including the Ames test for mutagenicity and hERG inhibition. Consequently, the compounds exhibit an enhanced safety profile and have a better chance of proceeding to the next stages of drug development. Analysis of the ADMET plot for the compounds from C. roseus demonstrates that a lot of the compounds fall within the targeted ADMET range, suggesting their possible use as drug candidates. According to the ADMET and Lipinski Rules, most of the compounds were found to possess acceptable pharmacokinetic properties and low toxicity. These observations suggest that compounds from C. roseus have the potential to be utilized in the development of novel drugs. This is supported by the data in Fig. 2 and Tables 1 and 2.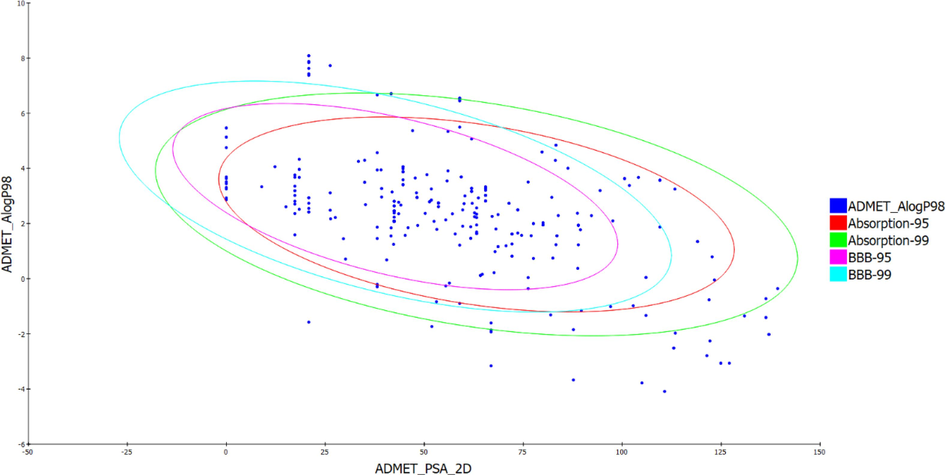
ADMET Profile: Correlation between 2D Polar Surface Area (PSA_2D) and Octanol-Water Partition Coefficient (AlogP98) for C. roseus compounds.
Name
H.B.A.
H.B.D.
M.W.
ALogP
Rot. Bonds
PSA
3239
6
4
480.677
3.572
5
107.22
21304
7
1
426.505
2.03
5
79.3
120879
4
1
336.427
2.094
2
41.57
192740
7
1
398.452
2.497
4
73.16
261578
7
1
426.505
1.951
5
79.3
425978
8
1
456.531
1.935
6
88.54
536803
6
1
396.479
2.326
5
67.87
3032566
6
2
368.426
1.258
2
71.03
4485535
6
0
490.68
3.115
7
47.1
5315221
6
2
368.426
0.816
2
71.03
5315229
7
1
426.505
1.949
5
79.3
5315746
6
2
384.469
1.572
3
73.23
5316490
8
2
481.622
−1.155
7
89.85
5316748
4
1
338.443
2.383
2
41.57
12310173
4
2
332.434
3.504
2
74.59
13039027
5
4
464.678
3.63
5
97.99
45480560
2
2
418.695
6.708
5
40.46
73197911
5
5
466.694
3.672
5
101.15
98054312
4
2
326.433
1.932
1
46.94
162871248
5
2
340.416
2.354
1
57.72
162963996
4
1
336.427
2.072
2
41.57
162971130
9
4
470.515
0.794
4
120.72
163058109
8
1
442.505
1.229
5
88.54
163083337
4
1
336.427
2.094
2
41.57
Name
Solubility
ALogP98
BBB
CYP2D6
Hepatotoxicity
Absorption
PPB
3239
2
0
4
F
T
0
T
21304
3
0
3
F
F
0
F
120879
2
0
2
T
F
0
F
192740
2
0
3
F
T
0
T
261578
3
0
3
F
F
0
F
425978
3
0
3
F
F
0
F
536803
2
0
3
T
F
0
T
3032566
3
0
3
F
F
0
F
4485535
3
0
1
F
F
0
F
5315221
3
0
3
F
F
0
F
5315229
3
0
3
F
F
0
F
5315746
3
0
3
F
F
0
F
5316490
4
0
4
F
T
0
F
5316748
2
0
2
T
F
0
T
12310173
2
0
2
F
F
0
T
13039027
3
0
4
F
F
0
T
45480560
1
0
4
F
F
1
T
73197911
3
0
4
F
T
0
T
98054312
3
0
2
F
F
0
F
162871248
2
0
2
T
T
0
T
162963996
2
0
2
T
F
0
F
162971130
3
0
4
F
F
0
F
163058109
3
0
3
F
F
0
F
163083337
2
0
2
T
F
0
F
3.3 Target prediction for C. roseus compounds
The Swiss Target Prediction tool, a robust computational method, was used to identify the probable target proteins of the bioactive compounds in C. roseus. This method analyzes chemical structure similarities and ligand–protein interaction data to make accurate predictions. The results of this analysis are presented in Table 3. The Compound 3239 was found to interact with target proteins Cytochrome P450 2 J2, cAMP-dependent protein kinase beta-1 catalytic subunit, and Serine/threonine-protein kinase. Compound 21304 specifically targets P-glycoprotein 1, cytochrome P450 3A4, and cytochrome P450 2 J2. The compound 120879 is anticipated to interact with the target proteins Cytochrome P450 2B6, Cytochrome P450 2C19, and Cytochrome P450 2C9. These identified targets have a potential role in cancer pathogenesis.
Compound
Predicted Target1
Predicted Target2
Predicted Target3
3239
Cytochrome P450 2 J2
cAMP-dependent protein kinase beta-1 catalytic subunit
Serine/threonine-protein kinase 38-like
21304
P-glycoprotein 1
Cytochrome P450 3A4
Cytochrome P450 2 J2
120879
Cytochrome P450 2B6
Cytochrome P450 2C19
Cytochrome P450 2C9
192740
P-glycoprotein 1
Cytochrome P450 3A4
Cytochrome P450 2 J2
261578
P-glycoprotein 1
Cytochrome P450 2 J2
Serine/threonine-protein kinase NEK6
425978
P-glycoprotein 1
Cytochrome P450 3A4
Cytochrome P450 2 J2
536803
P-glycoprotein 1
Serine/threonine-protein kinase OSR1
Alpha-2b adrenergic receptor
3032566
P-glycoprotein 1
Cytochrome P450 2 J2
Serine/threonine-protein kinase OSR1
4485535
P-glycoprotein 1
Alpha-2b adrenergic receptor
Muscarinic acetylcholine receptor M1
5315221
Polyadenylate-binding protein 1
Heat shock protein HSP 90-alpha
Eukaryotic translation initiation factor 4H
5315229
Cytochrome P450 2D6
P-glycoprotein 1
Cytochrome P450 2 J2
5315746
Cytochrome P450 3A4
Cytochrome P450 2D6
Proteasome Macropain subunit MB1
5316490
P-glycoprotein 1
Mitogen-activated protein kinase kinase kinase 1
Receptor-interacting serine/threonine-protein kinase 4
5316748
Neuronal acetylcholine receptor protein alpha-7 subunit
Sigma opioid receptor
Serotonin 1a (5-HT1a) receptor
12310173
P-glycoprotein 1
Serine/threonine-protein kinase OSR1
Myosin-IIIB
13039027
Cytochrome P450 2 J2
Heat shock protein HSP 90-alpha
Muscarinic acetylcholine receptor M1
45480560
P-glycoprotein 1
Serine/threonine-protein kinase OSR1
Serine/threonine-protein kinase DCLK1
73197911
Endothelin receptor ET-A
Cytochrome P450 2C9
Cytochrome P450 2 J2
98054312
P-glycoprotein 1
Phosphatidylinositol-4-phosphate 5-kinase type-1 gamma
Cytochrome P450 2 J2
162871248
Cytochrome P450 3A4
Endothelin receptor ET-A
TNF-alpha
162963996
P-glycoprotein 1
Cytochrome P450 3A4
Cytochrome P450 2C19
162971130
Retinoic acid receptor gamma
Retinoic acid receptor beta
Prostanoid IP receptor
163058109
Heat shock protein HSP 90-alpha
Muscarinic acetylcholine receptor M1
P-glycoprotein 1
163083337
P-glycoprotein 1
Serine/threonine-protein kinase ULK1
Mitogen-activated protein kinase kinase kinase 1
3.4 Molecular docking study
The current study aimed to investigate the potential interactions between the ten most significant bioactive compounds of C. roseus (PubChem CID: 5316490, 42495, 621578, 162971130, 162963996, 3239, 73197911, 21304, 5315746, and 5315221) and the BCL-2 protein (PDB ID: 6O0K), a critical target in cancer treatment. The top ten bioactive compounds were determined by selecting the compounds with the lowest binding energies. Post-docking analysis revealed that the ten bioactive compounds exhibited a high affinity for the binding pocket of the BCL-2 protein, particularly in the area where Venetoclax (LBM) binds. The researchers evaluated the binding affinities of the BCL-2 protein and the compounds using hydrophobic contacts, van der Waals interactions, and hydrogen bonding. Fig. 3 displays an intriguing, superimposed image of the bioactive compounds docked in the binding pocket of BCL-2. The study thoroughly analyzed the molecular interactions between the bioactive compound and the BCL-2 protein based on the docking results. The results showed that certain compounds were able to form stable complexes by hydrogen bonding to key amino acid residues in the binding pocket. The formation of Van der Waals forces and hydrophobic contacts contributed to the stabilization of the ligand–protein interactions. The structural orientations of the docking drugs were notably comparable to those of the reference inhibitor, Venetoclax (LBM). The bioactive compounds and Venetoclax display comparable binding interactions, suggesting their potential therapeutic benefit in targeting the BCL-2 protein for cancer treatment. The efficiency and consistency of the compounds binding within the BCL-2 protein binding pocket, which is similar to Venetoclax, are demonstrated in Fig. 3.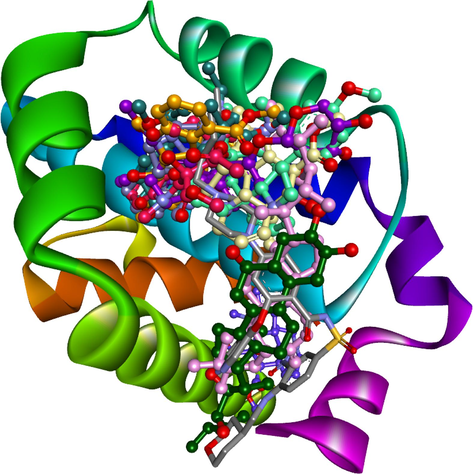
Molecular Docking of the Top Ten Bioactive Compounds from Catharanthus roseus within the Binding Pocket of the BCL-2 Protein, Revealing Potential Anticancer Interactions.
Several close intramolecular interactions were identified by the docking analysis, as shown in Fig. 4. These interactions can be classified as alkyl bonds, carbon-hydrogen bonds, hydrogen bonds, hydrophobic interactions, and electrostatic interactions. Compound 5316490 establishes significant interactions within the binding pockets of the BCL-2 receptor (6O0k). It was observed to form carbon-hydrogen bonds with VAL133:O and GLU136:O. Additionally, a hydrophobic interaction was observed with VAL133. Similarly, Compound 621578 was found to be forming CHB with TYR108:O and hydrophobic interactions with VAL133 (Alkyl), LEU137(Pi-Alkyl), MET115 (Pi-Alkyl), and VAL156 (Pi-Alkyl). Compound 162971130 forms hydrogen bonds with LEU137:O, ALA149:O, GLU136:O and ASP111:O. It also interacts hydrophobically with MET115 (Alkyl), VAL133 (Alkyl), LEU137 (Alkyl), PHE112 (Pi-Alkyl), and ALA149 (Pi-Alkyl). Compound 162963996 forms hydrogen bonds with ALA149:O, ASP111:O, ASP111:OD2 (Sharma, 2010), and MET115 (Pi-Alkyl). It reacts hydrophobically with Pi-Alkyl VAL156. The molecule 3239 forms hydrogen bonds with ARG146:NH1, ASP103:OD2, and LEU137:O. It also forms carbon hydrogen bonds with VAL133:O, ASP111:O, and GLU136:O, and electrostatic interactions with ASP111:OD1 (Pi-Anion). It also interacts hydrophobically with MET115 (Alkyl), VAL133 (Alkyl), LEU137 (Alkyl), and PHE153 (Pi-Alkyl). Compound 73197911 has typical hydrogen bonds with ASN143:ND2, ARG146:NE, ARG146:NH1, ALA100:O, ASP103:OD2, and TYR108. In addition, it also interacts hydrophobically with ALA100. Compound 21304 forms carbon hydrogen bonds with VAL133:O, ASP111:O, GLU136:O, electrostatic interactions with ASP111:OD1 (Pi-Anion), and hydrophobic interactions with MET115 (Alkyl), VAL133 (Alkyl), LEU137 (Alkyl), and PHE153 (Pi-Alkyl). Table 4 shows that the Compound 5315746 forms hydrogen bonds with ARG146:NE, ARG146:NE, ARG146:NH1, LEU137:, and GLU136:O.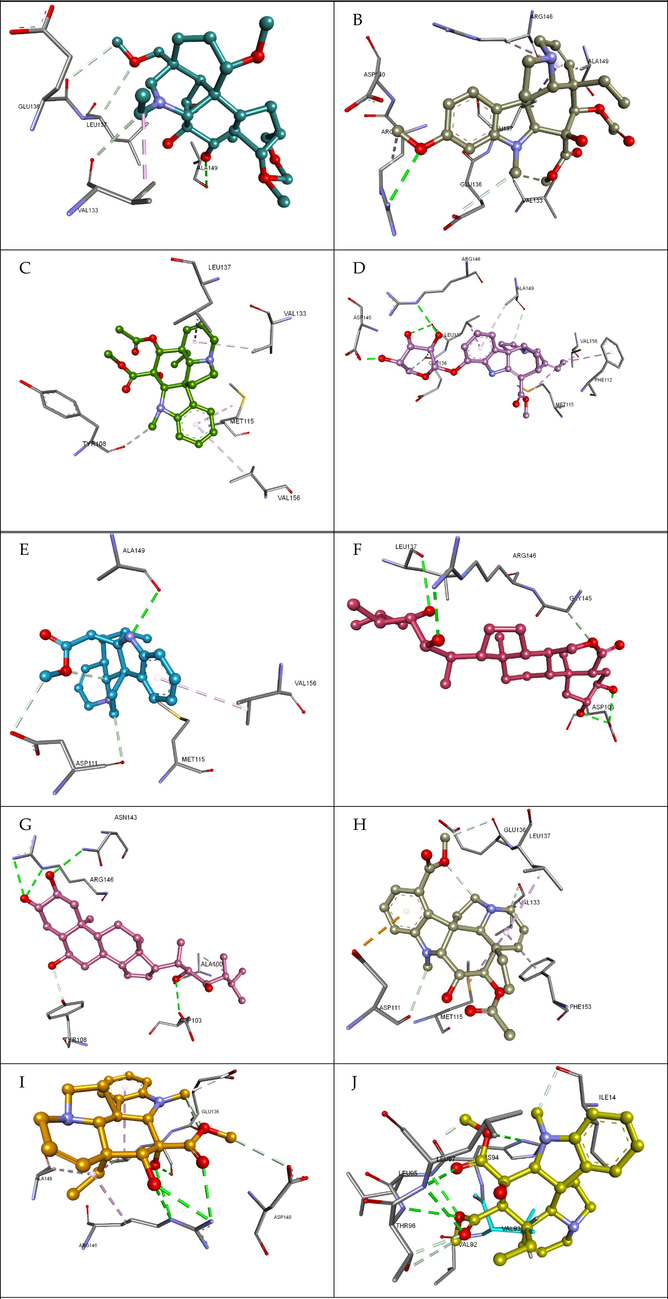
Top ten docked complexes and their close intramolecular interactions. The compounds are shown in balls and sticks, and the interacting binding pocket residues are shown in sticks. The green dashes are representing hydrogen bonds, and the pink dashes are representing hydrophobic interactions (A) 5316490, (B) 425978, (C) 621578, (D) 162971130, (E) 162963996, (F) 3239, (G) 73197911, (H) 21304, (I) 5315746, (J) 5315221. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
Compound
Intra-molecular Interaction
Interacting atoms
Distance in \AA
5316490
Hydrogen Bond
Z:UNK999:O - A:ALA149:O
3.03359
Hydrogen Bond
A:LEU137:CA - Z:UNK999:O
3.73757
Hydrogen Bond
Z:UNK999:C - A:VAL133:O
3.52379
Hydrogen Bond
Z:UNK999:C - A:GLU136:O
3.43654
Hydrophobic
Z:UNK999:C - A:VAL133
3.94712
425978
Hydrogen Bond
A:HOH447:O - Z:UNK999:O
2.88901
Hydrogen Bond
A:ARG139:NH1 - Z:UNK999:O
2.86343
Hydrogen Bond
Z:UNK999:C - A:GLU136:OE2
3.7434
Hydrogen Bond
Z:UNK999:C - A:VAL133:O
3.58818
Hydrophobic
Z:UNK999:C - A:LBM301
3.97739
Hydrophobic
A:LEU137 - Z:UNK999
5.13727
Hydrophobic
A:ARG146 - Z:UNK999
4.57188
Hydrophobic
A:ALA149 - Z:UNK999
4.41559
Hydrophobic
Z:UNK999:C - A:ARG139
3.87608
Hydrophobic
Z:UNK999 - A:LBM301:C44
4.80436
621578
Hydrogen Bond
A:HOH402:O - Z:UNK999:O
2.73942
Hydrogen Bond
A:HOH414:O - Z:UNK999:O
2.89045
Hydrogen Bond
A:HOH414:O - Z:UNK999:O
3.23449
Hydrogen Bond
Z:UNK999:C - A:TYR108:O
3.54313
Hydrophobic
A:VAL133 - Z:UNK999
5.17234
Hydrophobic
A:LEU137 - Z:UNK999
4.11392
Hydrophobic
A:LBM301:C - Z:UNK999
2.88885
Hydrophobic
Z:UNK999 - A:MET115
4.30917
Hydrophobic
Z:UNK999 - A:VAL156
5.10049
162963996
Hydrogen Bond
Z:UNK999:N - A:ALA149:O
2.97507
Hydrogen Bond
Z:UNK999:C - A:ASP111:O
3.19987
Hydrogen Bond
Z:UNK999:C - A:ASP111:OD2
3.23401
Hydrophobic
Z:UNK999 - A:MET115
4.90791
Hydrophobic
Z:UNK999 - A:VAL156
4.87257
162971130
Hydrogen Bond
A:ARG146:NE - Z:UNK999:O
3.09035
Hydrogen Bond
Z:UNK999:O - A:LEU137:O
2.73195
Hydrogen Bond
Z:UNK999:C - A:ALA149:O
3.64435
Hydrogen Bond
Z:UNK999:C - A:GLU136:O
3.79542
Hydrogen Bond
Z:UNK999:C - A:GLU136:O
3.07706
Hydrophobic
Z:UNK999:C - A:MET115
4.11007
Hydrophobic
Z:UNK999:C - A:VAL156
3.09619
Hydrophobic
A:PHE112 - Z:UNK999:C
4.45505
Hydrophobic
Z:UNK999 - A:LEU137
4.6527
Hydrophobic
Z:UNK999 - A:ALA149
4.58809
3239
Hydrogen Bond
A:ARG146:NH1 - Z:UNK999:O
2.50722
Hydrogen Bond
Z:UNK999:O - A:ASP103:OD2
2.49556
Hydrogen Bond
Z:UNK999:O - A:ASP103:OD2
2.61985
Hydrogen Bond
Z:UNK999:O - A:LEU137:O
3.19932
Hydrogen Bond
A:GLY145:CA - Z:UNK999:O
2.87308
73197911
Hydrogen Bond
A:ASN143:ND2 - Z:UNK999:O
2.92139
Hydrogen Bond
A:ARG146:NE - Z:UNK999:O
2.92515
Hydrogen Bond
A:ARG146:NH1 - Z:UNK999:O
2.81663
Hydrogen Bond
Z:UNK999:O - A:ALA100:O
2.55856
Hydrogen Bond
Z:UNK999:O - A:ASP103:OD2
2.9587
Hydrogen Bond
Z:UNK999:O - A:TYR108
3.35901
Hydrophobic
A:ALA100 - Z:UNK999
3.91063
21304
Hydrogen Bond
Z:UNK999:C - A:VAL133:O
3.58849
Hydrogen Bond
Z:UNK999:C - A:ASP111:O
3.25814
Hydrogen Bond
Z:UNK999:C - A:GLU136:O
3.66659
Electrostatic
A:ASP111:OD1 - Z:UNK999
3.83608
Hydrophobic
A:MET115 - Z:UNK999
5.44964
Hydrophobic
A:VAL133 - Z:UNK999
4.7011
Hydrophobic
A:LEU137 - Z:UNK999
4.09456
Hydrophobic
A:PHE153 - Z:UNK999
4.87986
5315746
Hydrogen Bond
A:ARG146:NE - Z:UNK999:O
3.00097
Hydrogen Bond
A:ARG146:NE - Z:UNK999:O
2.85746
Hydrogen Bond
A:ARG146:NH1 - Z:UNK999:O
3.06821
Hydrogen Bond
A:ARG146:NH1 - Z:UNK999:O
2.65798
Hydrogen Bond
Z:UNK999:O - A:LEU137:O
2.59558
Hydrogen Bond
Z:UNK999:C - A:GLU136:O
3.30066
Hydrogen Bond
Z:UNK999:C - A:GLU136:OE2
3.70192
Hydrogen Bond
Z:UNK999:C - A:ASP140:OD2
3.0496
Hydrophobic
A:ARG146 - Z:UNK999
4.06054
Hydrophobic
A:ALA149 - Z:UNK999
4.20951
Hydrophobic
Z:UNK999 - A:LEU137
5.31094
Hydrophobic
Z:UNK999 - A:LEU137
4.53145
5315221
Hydrogen Bond
A:HIS94:ND1 - Z:UNK999:O
3.21487
Hydrogen Bond
A:LEU95:N - Z:UNK999:O
2.72989
Hydrogen Bond
A:THR96:N - Z:UNK999:O
3.08657
Hydrogen Bond
A:LEU97:N - Z:UNK999:O
2.70156
Hydrogen Bond
A:LEU97:N - Z:UNK999:O
3.28436
Hydrogen Bond
A:THR96:CB - Z:UNK999:O
2.50998
Hydrogen Bond
Z:UNK999:C - A:ILE14:O
2.46888
Hydrogen Bond
Z:UNK999:C - A:THR96:OG1
3.40658
4 Discussion
Catharanthus roseus, commonly referred to as the Madagascar periwinkle or Vinca rosea, is a well-known botanical species due to its extensive array of chemical compounds and therapeutic properties.
Formal rephrasing: GPCRs are a class of transmembrane receptors that play a crucial role in cellular signal transduction. Our research investigated the potential of phytochemicals derived from C. roseus in treating NSCLC by targeting the BCL-2 protein. BCL-2 is an apoptosis regulator that allows cancer cells to evade programmed cell death. By inhibiting apoptosis through the dysregulation of BCL-2 in NSCLC, cancer cells are able to survive, promote tumorigenesis, and develop resistance to therapy. By targeting BCL-2, there is a unique opportunity to eliminate cancer cells while sparing healthy cells (López-Otín and Overall, 2002). Our research identified and assessed the bioactive compounds in C. roseus. The focus of these evaluations was on the therapeutic benefits of the selected compounds. C. roseus phytochemicals were subjected to ADMET screening to identify compounds with promising drug-like properties and low toxicity. This precise selection criterion allowed us to scrutinize the favourable compounds. The molecular docking analysis revealed that bioactive compounds with drug like properties have strong interactions with the BCL-2 protein binding site. These interactions suggest a therapeutic intervention mechanism and support the use of these compounds to improve NSCLC treatment by targeting the BCL-2 protein (López-Otín and Overall, 2002).
Identifying compounds that target GPCRs may be useful in cancer treatment because GPCRs are essential for tumor growth, metastasis, and the complex signalling networks that exist within cancer cells (Chaudhary and Kim, 2021). GPCRs can be targeted to disrupt these processes and alter the tumor microenvironment, thereby preventing cancer (Arakaki et al., 2018). The identified bioactive compounds derived from C. roseus can selectively bind to GPCRs and modify their signalling pathways by either activating or inhibiting them. These compounds can act as ligands for GPCRs (Yang, 2021). These compounds reveal a broad range of pharmacological effects on several physiological processes, including immune system function, hormone response, and neurotransmission modulation. Ion channels are types of proteins that allow ion transport across cell membranes in order to regulate signaling and excitation. C. roseus contains bioactive compounds that can modulate ion channels and causes changes in the transport of ions across cellular membranes, including both influx and efflux mechanisms (Tiwari, 2023). Ion channel modulation can affect many different physiological processes, including muscle contraction and neuronal activity (Rosendo-Pineda et al., 2020). KI is a type of compound that selectively targets and inhibits its function. Kinases are essential for regulating various cellular signalling cascades. As a result, these compounds have the potential to serve as therapeutic agents. These compounds efficiently target and modify the signalling cascades associated with various clinical disorders by inhibiting kinases. C. roseus bioactive compounds can bind to nuclear receptors and regulate gene expression. The ligands under consideration have the potential to regulate immune system responses, hormonal signalling pathways, and metabolic processes (Sar, 2023). The extracts of C. roseus contain bioactive compounds that exhibit protease inhibition, which suggests potential therapeutic applications in diseases characterized by uncontrolled protease activity, such as cancer and viral infections (López-Otín and Overall, 2002). Furthermore, it has been discovered that C. roseus contains compounds with enzyme-inhibitory properties. Enzyme inhibitors have therapeutic potential in the treatment of microbial infections, metabolic disorders, cancers, and inflammatory disorders. They also provide a way to regulate various biochemical pathways. Altogether, C. roseus provides a significant source of bioactive substances with a wide variety of pharmacological properties.
Several compounds derived from C. roseus have demonstrated promising results in a variety of therapeutic areas. These compounds have variable degrees of bioactivity, indicating their ability to interact with diverse biological systems. In terms of bioactivity, 5316490 is particularly noteworthy, displaying high performance in a variety of assays such as GPCR ligand, ICM, NRL, and PI. This compound’s versatility indicates that it could be employed as a multi-target drug. Furthermore, its bioactivity scores in PI and KI indicate that it has the ability to treat cancer, which is an important aspect of drug development in this field. Compounds with broad activity profiles are especially useful in complex diseases that involve multiple pathways, making them a valuable resource for drug discovery (Hazra et al., 2022). Compound 5315229 offers a promising prospect as an ion channel modulator and kinase inhibitor, both of which are critical in cancer treatment. Ion channels are of particular interest in the treatment of neurological disorders due to their ability to modulate neuronal signalling. Kinase inhibitors, on the other hand, are essential in blocking abnormal signalling pathways in cancer cells. The Compound 425978 has displayed positive results as both an ion channel modulator and a nuclear receptor ligand. Its interaction with nuclear receptors suggests that it regulates gene expression, which may affect endocrine and metabolic processes (Rosendo-Pineda et al., 2020). Compound displays biological activity as a GPCR ligand, ICM, and NRL, suggesting its potential as a therapeutic agent due to its broad target engagement. Furthermore, the fact that GPCRs are linked to a diverse range of disorders highlights the importance of compounds like 261578 in drug discovery (Lappano and Maggiolini, 2011). Altering ion channels can change how cancer cells spread, and nuclear receptor interactions may play a role in controlling cancer-related genes (Capatina et al., 2022). These compounds hold promise for application in cancer research and treatment. They might affect numerous aspects of cancer biology by modifying certain signalling pathways and influencing crucial cellular processes.
Following the ADMET analysis, the compounds obtained from C. roseus exhibit considerable promise as a potential candidate for drug development. As seen in Table 2, these compounds have many applications in pharmaceutical research. Oral administration of these compounds is feasible due to their efficient gastrointestinal absorption. A significant number of compounds can permeate biological membranes and spread throughout the body by following Lipinski's Rule of Five. This finding suggests that these compounds have a high oral bioavailability, which is an important consideration in the development of novel medicines (Ferreira and Andricopulo, 2019; Tasleem, 2021). The ideal distribution of drugs to target tissues and organs requires a balance between hydrophilic and lipophilic properties. Several compounds are shown to disperse efficiently in tissues that are rich in lipids, as shown by their lipophilicity and logP values. The compounds were found to be less vulnerable to hepatic metabolism, as evidenced by their moderate to low rates of metabolism supported by cytochrome P450 enzymes. This suggests that these compounds may be able to stay in the body for longer periods of time with less excretion, which is a desired trait for potential medicinal candidates (Bertram-Ralph and Amare, 2023). The results indicate that a majority of the compounds decreased efflux transporter function, which resulted in greater renal clearance and reduced the possibility of active cell transportation. This feature is essential for increasing drug levels and prolonging drug action, both of which are crucial for achieving therapeutic efficacy. Additionally, these compounds have good safety parameters, such as meeting the toxicity criteria for hERG inhibition and showing negative results in the Ames test for mutagenicity. These findings suggest that these substances have great potential as candidates for pharmaceutical research and development (Shugarts and Benet, 2009).
Identifying the target proteins of bioactive compounds and their potential therapeutic uses necessitates an understanding of the pharmacological mechanisms involved. A predictive study with Swiss Target Prediction identified numerous possible target proteins for bioactive compounds derived from C. roseus. Cytochrome P450 has been shown to interact with Compound 3239. This enzyme has been linked to cancer because it metabolizes medicines and procarcinogens, which can activate and detoxify carcinogens. By inhibiting or modifying the activity of cytochrome P450, it may be possible to inhibit these processes, making it an attractive target for cancer prevention and treatment strategies. The compounds with identification codes 21304, 192740, 261578, 425978, 5315229, 5316490, 13039027, 73197911, 98054312, and 162963996 are expected to interact with P-gp, an efflux transporter known to play a role in multidrug resistance in cancer cells. Inhibiting P-glycoprotein (P-gp) may increase the intracellular accumulation of chemotherapeutic agents, potentially increasing their efficacy in cancer treatment (Seelig, 2020). According to the results, the compounds 5315221 and 163058109 have been found to interact selectively with Heat Shock Protein 90-alpha, a vital molecular chaperone required for the stability and proper functioning of a variety of client proteins. Cancer cells rely on these client proteins for their growth and survival. HSP 90 inhibitors have been investigated as potential cancer treatments (Li and Luo, 2023). Based on current predictions, Compound 3239 is anticipated to interact with AMP-dependent protein kinase, a crucial regulator of autophagy and cellular proliferation. Furthermore, it is believed to participate in interactions with Serine/threonine-protein kinase 38. Given that uncontrolled kinase activation is a common feature of cancer, targeting these enzymes with therapeutic interventions is a promising approach for cancer treatment. Compound 261578 is expected to interact with Serine/threonine-protein kinase NEK6, a protein essential for regulating the cell cycle. As cancer is characterized by the dysregulation of cell cycle checkpoints, investigating the therapeutic potential of targeting kinases like NEK6 is desirable (Yu, 2022). Compound 162971130 is anticipated to interact with retinoic acid receptors, specifically the gamma and beta types. These receptors play an important role in cancer treatment because they regulate cell differentiation and programmed cell death. As such, they have a significant role in the management of acute promyelocytic leukemia (Dhokia and Macip, 2021).
The binding energy is the determinant of the interaction between a compound and the binding pocket of a protein. A low binding energy means that the complex is more stable and has a stronger ligand–protein interaction. Additionally, Van der Waals (VDW) forces, which are non-covalent and crucial for molecular docking, play a significant role in the interaction between the ligand and protein during docking. These forces aid in positioning the ligand in the correct orientation within the binding pocket (Hermann et al., 2017).
Bioactive compounds from C. roseus were found to interact with the BCL-2 receptor via complex molecular pathways. These interactions are essential for using these compounds as anti-cancer therapies. A more comprehensive analysis reveals that the complex interactions between hydrophobic forces and hydrogen bonds produce the ligand-receptor binding patterns. Hydrogen bonds are essential for molecular recognition, and the study stressed their importance (Hubbard et al.). These interactions are classified into several categories: compounds such as 5316490, 73197911, 162971130, 162963996, and 3239 exhibit hydrogen bonding interactions. For instance, LEU137 exhibited its ability to interact with a wide range of ligands by forming conventional hydrogen bonds with molecules 5316490 and 162963996. It was discovered that ASP103 and compound 3239 form hydrogen bonds, which increase the stabilizing effect of ASP103. Direct interactions between hydrogen atoms and electronegative atoms generate strong electrostatic attraction, resulting in conventional hydrogen bonds. These interactions are essential for the formation of stable ligand-receptor complexes. One interesting interaction shown by compound 73197911 is that the π electrons of an aromatic ring in the ligand form a hydrogen bond with an acceptor atom in the receptor (TYR108). This particular interaction serves as an example of how aromatic residues are involved in the identification and binding of specific ligands. Since pi-donor hydrogen bonds depend on the presence of aromatic rings in both the ligand and the receptor, they provide an extra degree of specificity to the binding process. Furthermore, under certain conditions, the carbon atoms of the compounds take part in hydrogen bonding. It was observed that the compounds 5316490, 42495, 621578, 162971130, 162963996, and 3239 form carbon-hydrogen bonds with the binding pocket residues, VAL133, ASP111, and GLU136. These interactions reveal the remarkable flexibility of the receptor by highlighting the heterogeneity of hydrogen bonding in ligand-receptor binding. Hydrogen bonds between carbon atoms are essential for the ligand-receptor complex's stability and selectivity (Herschlag and Pinney, 2018). Hydrophobic interactions, or hydrophobic forces, are crucial for the stability of ligand-receptor complexes. These interactions cause nonpolar groups to become isolated from the surrounding water, which causes them to agglomerate (Franks, 1975). Alkyl interactions with compounds 5316490, 621578, 73197911, 21304, and 5315746 were noted with LEU137, VAL133, MET115, ALA149, ARG146, and ALA100. Alkyl interactions are formed when hydrophobic ligand segments—often with alkyl groups—make contact with hydrophobic receptor residues. These interactions increase binding affinity by thoroughly saturating the hydrophobic segments of the ligand in the hydrophobic binding pocket of the receptor. In Compound 162971130, 162963996, 3239, 73197911, and 5315746, pi-alkyl interactions are mediated by residues MET115, VAL156, LEU137, PHE112, PHE153, and ARG146. The alkyl groups of hydrophobic residues in the receptor and the pi electrons of an aromatic ring in the ligand are involved in these interactions. They strengthen and stabilize the ligand-receptor complex (Ferenczy and Kellermayer, 2022). The variety and complexity of these hydrophobic and hydrogen interactions demonstrate the intricacy of ligand-receptor binding. Since these interactions affect not only the binding strength but also the selectivity and specificity of possible drug candidates, an understanding of their complexities is essential for rational drug design. The distinct interaction profile of each compound adds to the wide range of binding that occurs within the binding pocket of BCL-2 receptor.
5 Conclusions
This study finds C. roseus derived compounds as potential therapeutics for NSCLC. The broad bioactivity of compound 5316490 suggests that it may possess wide range of applications as a pharmaceutical agent. Additionally, compound 425978 and compound 5315229 exhibit unique activity profiles, which bodes well for the treatment of cancer. Compound 261578 has a diverse set of bioactivities, making it a versatile candidate for drug development. These compounds target cancer pathways via specific protein interactions, most notably hydrogen bonding (LEU137, ASP103) and hydrophobic interactions (VAL133, MET115) with the BCL-2 receptor. Furthermore, their favorable pharmacokinetic profiles position them as promising option for treating NSCLC and associated diseases. The study makes a substantial contribution to the knowledge of C. roseus compounds in the context of personalised NSCLC therapies.
Funding
This research has been funded by the Scientific Research Deanship at the University of Ha’il, Saudi Arabia, through project number BA-2215.
CRediT authorship contribution statement
Ghaida Alsaif: Formal analysis, Writing – original draft, Conceptualization, Writing – review & editing. Munazzah Tasleem: Formal analysis, Writing – review & editing, Conceptualization. Raja Rezgui: Formal analysis, Supervision, Writing – review & editing. Khalid Alshaghdali: Writing – review & editing. Amir Saeed: Conceptualization, Writing – review & editing. Mohd Saeed: Formal analysis, Supervision, Writing – review & editing.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Molecular docking as a tool for the discovery of molecular targets of nutraceuticals in diseases management. Sci. Rep.. 2023;13(1):13398.
- [Google Scholar]
- Protein conformational flexibility modulates kinetics and thermodynamics of drug binding. Nat. Commun.. 2017;8(1):2276.
- [Google Scholar]
- GPCRs in Cancer: Protease-Activated Receptors, Endocytic Adaptors and Signaling. Int. J. Mol. Sci.. 2018;19(7)
- [Google Scholar]
- Factors affecting drug absorption and distribution. Anaesthesia Intensive Care Med.. 2023;24(4):221-227.
- [Google Scholar]
- RCSB Protein Data Bank: powerful new tools for exploring 3D structures of biological macromolecules for basic and applied research and education in fundamental biology, biomedicine, biotechnology, bioengineering and energy sciences. Nucleic Acids Res.. 2020;49(D1):D437-D451.
- [Google Scholar]
- Targeting Ion Channels for Cancer Treatment: Current Progress and Future Challenges. Rev. Physiol. Biochem. Pharmacol.. 2022;183:1-43.
- [Google Scholar]
- The BCL-2 Inhibitor Venetoclax Augments Immune Effector Function Mediated by Fas Ligand, TRAIL, and Perforin/Granzyme B, Resulting in Reduced Plasma Viremia and Decreased HIV Reservoir Size during Acute HIV Infection in a Humanized Mouse Model. J. Virol.. 2022;96(24):e0173022.
- [Google Scholar]
- Bcl-2 promotes invasion and lung metastasis by inducing matrix metalloproteinase-2. Cancer Res.. 2005;65(13):5554-5560.
- [Google Scholar]
- SwissTargetPrediction: updated data and new features for efficient prediction of protein targets of small molecules. Nucleic Acids Res.. 2019;47(W1):W357-W364.
- [Google Scholar]
- A master of all trades – linking retinoids to different signalling pathways through the multi-purpose receptor STRA6. Cell Death Discovery. 2021;7(1):358.
- [Google Scholar]
- Anticancer potential of alkaloids: a key emphasis to colchicine, vinblastine, vincristine, vindesine, vinorelbine and vincamine. Cancer Cell Int.. 2022;22(1):206.
- [Google Scholar]
- Contribution of hydrophobic interactions to protein mechanical stability. Comput. Struct. Biotechnol. J.. 2022;20:1946-1956.
- [Google Scholar]
- ADMET modeling approaches in drug discovery. Drug Discov. Today. 2019;24(5):1157-1165.
- [Google Scholar]
- The hydrophobic interaction. In: Water A Comprehensive Treatise: Aqueous Solutions of Amphiphiles and Macromolecules. Springer; 1975. p. :1-94.
- [Google Scholar]
- Natural Phytocompounds from Common Indian Spices for Identification of Three Potential Inhibitors of Breast Cancer: A Molecular Modelling Approach. Molecules. 2022;27(19):6590.
- [Google Scholar]
- First-Principles Models for van der Waals Interactions in Molecules and Materials: Concepts, Theory, and Applications. Chem. Rev.. 2017;117(6):4714-4758.
- [Google Scholar]
- iGEMDOCK: a graphical environment of enhancing GEMDOCK using pharmacological interactions and post-screening analysis. BMC Bioinf.. 2011;12(1):S33.
- [Google Scholar]
- Hubbard, R.E., Kamran Haider, M. Hydrogen Bonds in Proteins: Role and Strength, in Encyclopedia of Life Sciences.
- Apoptosis and bcl-2 expression as predictors of survival in radiation-treated non–small-cell lung cancer. Int. J. Radiation Oncol.* Biol.* Phys.. 2001;50(1):13-18.
- [Google Scholar]
- Catharanthus roseus (L.) G. Don: A review of its ethnobotany, phytochemistry, ethnopharmacology and toxicities. J. Ethnopharmacol.. 2022;284:114647
- [Google Scholar]
- G protein-coupled receptors: novel targets for drug discovery in cancer. Nat. Rev. Drug Discov.. 2011;10(1):47-60.
- [Google Scholar]
- HSP90 inhibitors and cancer: Prospects for use in targeted therapies (Review) Oncol. Rep.. 2023;49(1)
- [Google Scholar]
- Protease degradomics: A new challenge for proteomics. Nat. Rev. Mol. Cell Biol.. 2002;3(7):509-519.
- [Google Scholar]
- Methotrexate-conjugated zinc oxide nanoparticles exert a substantially improved cytotoxic effect on lung cancer cells by inducing apoptosis. Front. Pharmacol.. 2023;14:1194578.
- [Google Scholar]
- Medicinal plants used in treatment and management of cancer in Kakamega County, Kenya. J. Ethnopharmacol.. 2014;151(3):1040-1055.
- [Google Scholar]
- Nuclear receptor: Structure and function. Prog. Mol. Biol. Transl. Sci.. 2023;196:209-227.
- [Google Scholar]
- P-glycoprotein: one mechanism, many tasks and the consequences for pharmacotherapy of cancers. Front. Oncol.. 2020;10:576559
- [Google Scholar]
- A chromatin-mediated reversible drug-tolerant state in cancer cell subpopulations. Cell. 2010;141(1):69-80.
- [Google Scholar]
- The role of transporters in the pharmacokinetics of orally administered drugs. Pharm. Res.. 2009;26(9):2039-2054.
- [Google Scholar]
- Bcl-2 family in non-small cell lung cancer: its prognostic and therapeutic implications. Pathol. Int.. 2017;67(3):121-130.
- [Google Scholar]
- Investigation of Antidepressant Properties of Yohimbine by Employing Structure-Based Computational Assessments. Curr. Issues Mol. Biol.. 2021;43(3):1805-1827.
- [Google Scholar]
- Ethanolic Extract of Artemisia vulgaris Leaf Promotes Apoptotic Cell Death in Non-Small-Cell Lung Carcinoma A549 Cells through Inhibition of the Wnt Signaling Pathway. Metabolites. 2023;13(4):480.
- [Google Scholar]
- IMPPAT 2.0: an enhanced and expanded phytochemical atlas of Indian medicinal plants. ACS Omega. 2023;8(9):8827-8845.
- [Google Scholar]
- G protein-coupled receptors: structure- and function-based drug discovery. Signal Transduct. Target. Ther.. 2021;6(1):7.
- [Google Scholar]
- In silico drug-likeness, biological activity and toxicity prediction of new 3, 5-bis (hydroxymethyl) tetrahydro-4H-pyran-4-one derivatives. Chemical Bulletin of Kazakh National University. 2022;107(4):14-20.
- [Google Scholar]
- NEK6 is an injury-responsive kinase cooperating with STAT3 in regulation of reactive astrogliosis. Glia. 2022;70(2):273-286.
- [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.jksus.2024.103134.
Appendix A
Supplementary material
The following are the Supplementary data to this article:







