Translate this page into:
Genetic association between Q192R polymorphism in the paraoxonase 1 gene and female infertility in the Saudi women: Validated using DNA sequencing analysis
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Objective
Infertility may also describe the inability to conceive whether a woman continues to have miscarriages. Female infertility is a complex condition which can suffer from a range of conditions and one-third of all infertility cases occur in women. Obesity is correlated by various complex pathways with infertility among women. Paraoxonase (PON) is an antioxidant and oxidant enzyme that plays an important role in a variety of conditions, including inflammation, oxidative stress, and lipid metabolism. Paraoxonase1 (PON1) gene variants can influence oxidative stress and thus female infertility susceptibility. However, no molecular studies have been conducted in Saudi infertile women and the Q192R polymorphism. The purpose of this study was to investigate into the relationship between the Q192R polymorphism in the PON1 gene and the risk of infertility in Saudi women.
Study design
This case-control study was carried out with 122 female infertile women and 100 fertile women. Genomic DNA was extracted from 222 participants, quantified performed polymerase chain reaction, agarose gel run, and RFLP analysis. Finally, Sanger sequencing was validated in 10% of the samples obtained. Excel was used to collect clinical and genomic data, which was then analyzed statistically.
Results
Anthropmetric and clinical data was assessed using both the cases and control women (p < 0.05). Hardy Weinberg equilibrium was in accordance within the Q192R polymorphism (p = 0.001). Q192R polymorphism confirms the allele (p = 0.003) and genetic association (QR vs QQ; p = 0.001) with different mode of inheritances including dominant and co-dominant models (p = 0.003). ANOVA analysis revealed a positive association between weight and Q192R polymorphism (p = 0.04).
Conclusion
This study concludes as Q192R polymorphism was positively associated in this case-control study performed in the Saudi women diagnosed with female infertility. However, obesity factor might also be playing a role in the infertile women.
Keywords
Female infertility
Q192R
rs662 polymorphism
PON1 gene
Obesity
Saudi women
1 Introduction
Infertility, according to world health organization, is a reproductive system disorder that affects both male and female populations. It is characterized as the failure to reproduce after 12 months of unprotected sexual intercourse (Tantengco, n.d.; Al-Mutawa, 2023). Infertility has been reported to be one of the most common chronic health conditions, regardless of age, affecting 15 % of all couples worldwide, with female infertility accounting for 40 % of all cases (Tomaiuolo, 2020; Brazdova, 2016). A woman (or man) may be considered infertile for one of two reasons: primary or secondary infertility. The inability of a couple to conceive after one year of unprotected intercourse is referred to as primary infertility. Couples with secondary infertility may take a long time to conceive and carry the pregnancy to term, or they may experience a miscarriage (Lindsay and Vitrikas, 2015). Endometriosis, uterine fibroids, premature ovarian failure, pelvic inflammatory disease, and polycystic ovary syndrome (PCOS) are all medical conditions that can contribute to female infertility (Al-Mutawa, 2018). Anovulation, tubal disease, pelvic adhesions, and endometriosis are the most common causes of female infertility. Many couples experience unexplained infertility, which is defined as infertility with no obvious cause (Barbieri, 2019). Ovulation disorders (25 %) are a major contributing factor to the development of female infertility. Furthermore, pelvic/vaginal adhesions (11 %), endometriosis (15 %), hyperprolactinemia (7 %), tubal blockage (11 %), and other tubal anomalies (11 %). When a cause of unexplained infertility is known, the route to care usually becomes evident (Unuane, 2011). Hormonal, genetic, thrombotic, anatomic, autoimmune and unknown infectious factors are the cause of female infertility (Artini, 2013). Environmental factors for the progression of female infertility include stress, various toxins, chemical dusts, pesticides, and physical agents (Mendiola, 2008).
Many causes of female infertility have been identified, with the majority of them being genetic abnormalities such as chromosomal defects or single gene mutations. Several factors have been identified as contributors to female infertility, and because there is no hereditary disorder that could implicate a gene in the pathogenesis of female infertility, and distinct genetic markers are lacking (Liao, 1998). Female infertility, and especially unexplained infertility, has been linked to single nucleotide polymorphisms (SNPs) in a number of genes, suggesting that these variations may have a role in the development of the condition(Fatima, 2019). Numerous genes have been implicated in germ cell pathogenesis and maturation, with defects leading to accelerated cell apoptosis and follicle atresia, both of which contribute to primary ovarian insufficiency (Yatsenko and Rajkovic, 2019). Many genetic studies have identified enormous potential candidate genes for the development of female infertility, and genetic influences have a major effect on several factors for its development. Specific genes and mutations may exacerbate hormonal issues, leading to 46 XX infertility (Siahpoosh, 2021). SNPs, which are common genetic variations found in the human genome and can frequently contribute to abnormal gene function, are frequently used in the treatment and management of infertility. SNPs in the paraoxonase 1 (PON1; OMIM: 168820) gene, which is one of the antioxidant genes, are especially significant in female infertility (Swaminathan, 2016). Diverse studies have discovered a link between the polymorphism and the risk of various diseases, such as previous findings indicating that there is a high variability in PON1 enzymes linked to a variety of issues, including frequent pregnancy loss and female infertility (Pandey, 2020). PON1 is an arylesterase that primarily hydrolyzes highly toxic compounds like paroxon and protects against lipid oxidation in low density, which is encoded by the PON1 gene (Al-Hakeem, 2014). There are approximately 160 polymorphisms in the human PON1 gene's coding sequence (exons), noncoding sequence (introns), and other regulatory regions (Alharbi, 2017). The PON1 gene, which has 9 exons and 8 introns and is located on 7q21-3 (Gene ID: 5444), produces a 1,769-bp mRNA (NM 000446.5) and a 355-aa protein (NP 000437.3) (Attar, 2015). A missense mutation is caused by one of the common SNPs studied in the PON1 gene. A > G in exon 6 causes the transition from glutamine (Q) to arginine (R) (p.Gln192Arg, p.Q192R, A192B), which has been connected to low and high paraoxonase activity, respectively (Matam, 2015).
The rs662 polymorphism was studies in global wide in the numerous human diseases as well as in limited diseases in the Saudi population. Although the rs662 (Q192R) polymorphism in the PON1 gene has been linked to female infertility in certain research, none of these studies have been conducted on Saudi women who are experiencing this issue. Consequently, the purpose of this study was to examine the relation between the PON1 gene's rs662 polymorphism and female infertility in Saudi Arabia.
2 Materials and methods
2.1 Opting of Saudi women with fertile and infertile
This study began with the grant of ethical approval (E-19–4344) from the institutional review board of King Saud University (KSU) in Riyadh, Saudi Arabia. Informed consent form was obtained from the participated women involved in this study. The research was completely followed by Helsinki declaration. This cross-sectional case-control study included 222 Saudi women, 100 of whom were fertile and 122 of whom were infertile. The cases and controls were obtained from Department of Obstetrics and Gynecology at King Khalid University Hospital. Cases of female infertility were included if the woman had a history of infertility in her family and had been trying to conceive for more than two to three years. Hormonal and biochemical testing, STD screening, hysterosalpingography, hysteroscopy, laparoscopy, genetic immunological anomalies, and sperm analysis from a partner were all used to delve into the causes of infertility. Women without family history of infertility, regular menstrual cycle and ability to conceive is considered as exclusion criteria of the female infertility and inclusion criteria of the fertile women (control subjects). The exclusion criteria of control subjects were ovarian lesions, endometriosis and autoimmune diseases (Al-Mutawa, 2018).
2.2 Anthropometric and blood analysis
Among the demographic data collected for this study were participants' ages (in years), heights (in centimeters), weights (in kilograms), and body mass indices (BMIs) (in kilograms per square meter kg/m2) (Alshammary and Khan, 2021). Additionally, women affected with infertility and family history was also recorded. Two milliliters of peripheral blood were taken in an ethylenediaminetetraacetic acid tubse for the purpose of molecular analysis in this study.
2.3 Nucleotide evaluation
Alshammary et al (Alshammary and Khan, 2021) technique was used for extraction of genomic DNA using peripheral blood with Qiagen DNA isolation kits as recommended protocol from Qiagen Company. NanoDrop spectrophotometer was performed to check the DNA quality. All the DNA samples concentration was converted to 10 ng and stored at −80 °C further used for Q192R genotyping. The 5‘-3́ of oligonucleotide sequence of rs662 polymorphism sequence was GGG-ACC-TGA-GCA-CTT-TTA-TGG-C and 3′-5′ sequence of oligonucleotide was CAT-CGG-GTG-AAA-TGT-TGA-TTC-C was used in polymerase chain reaction (PCR). The oligos were optimized for 100 pmoles as stock and converted into 10pmoles as working primers (5′-3′ & 3′-5′). The primers were synthesized from reGene Biologics from capital city of Telangana, India. A 50 µl of PCR reaction was performed using 10X buffer, 50 mM of MgCl2, 0.5 mM dNTP’s, 10pmoles of forward and reverse primers, Taq DNA (10units) and 10 ng of genomic DNA. By adding double distilled water, the final concentration of 50 µl was optimized. PCR was performed on a thermal cycler with initial denaturation at 95 °C/5mins, denaturation at 95 °C/30secs, annealing at 58 °C/30secs, extension at 72 °C/45secs, and final extension at 72 °C/5mins and hold at 4 °C. The total process for completion of PCR was 35 cycles around 1.34 hrs.
2.4 Agarose gel electrophoresis
Three percent of agarose gel was prepared using 7.5-grams of agarose powder (thermo scientific, Lithuania; Ref#R0491) was melted in 250 ml of 1X buffer which was prepared using 25 ml of 10X buffer (10X TBE Electrophoresis Buffer #B52) was dissolved in 225 ml of double distilled water. Additionally, concentration of 10 mg/ml of ethidium bromide (Invitrogen, USA Cat#15585–011) was dissolved to visualize nitrogenous bases of DNA and fluoresces under the ultraviolet (UV) light. A 10-µl of undigested PCR product was loaded with 5 µl of 6X loading dye. The horizontal electrophoresis was performed for 120 mins using 12 W, 100 mA and 100 V. Later, the agarose gel was examined under a UV transilluminator to determine genotyping. The undigested PCR amplicon of Q192R polymorphism was confirmed to be 176 bp by using the 100 bp DNA marker (#SM1143) as to determine the fragment size (Figure-1).
2.5 Digestion with restriction enzyme analysis
Analysis of restriction fragment length polymorphism (RFLP) was implemented to Q192 polymorphism in the PON1 gene. In this study, to digest the ↑GATC sequence, MboI restriction enzyme was used to digest the DNA at the concern restriction site (↑GATC to CTAG↓) at position 192 in rs662 polymorphism. The RFLP analysis was processed using 15 µl of undigested PCR product, 4 µl of 10X buffer and 1 µl of 10U/µl of Mbol restriction enzyme (New England Biolabs from USA). The final concentration of 25 µl of digested product was filled with 5 µl of double distilled water. The digestion was processed for 16 h at 37 °C and 3 % agarose gel (7.5 % of agarose powder in 250 ml of 1X buffer) was used to check digested products. The normal Q allele confirms 145 bp for the long band and 31 bp for the short band, while the variant R allele confirms 117 bp, 31 bp, and 28 bp (Fig. 1).(See Fig. 2).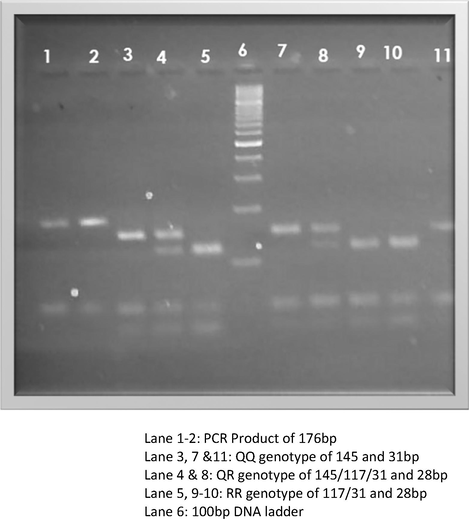
Representation of 3% agarose gel electrophoresis stained with ethidium bromide with digested and undigested Q192R PCR products.
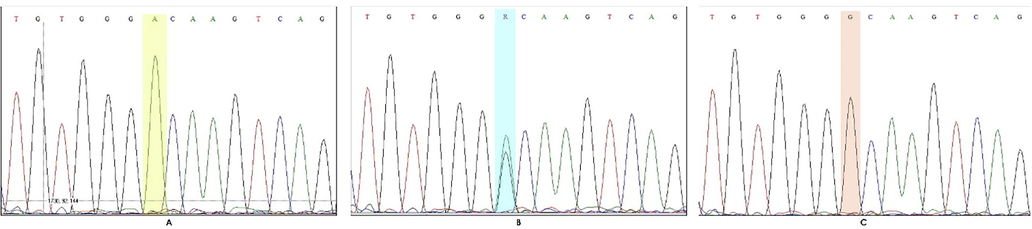
DNA validation of rs662 polymorphism of QQ, QR and RR genotypes through Sanger sequencing analysis.
2.6 Sanger sequencing analysis
The 176 bp PCR product of the PON1 gene was further purified from the Sanger sequencing analysis. This reaction was carried out using the Sanger dideoxy chain-termination process in bidirectional high-throughput multi capillary sequencing. In this study, 10 % of the PCR samples were sequenced to validate the RFLP analysis, as well as to reconfirm the results. The Sanger sequencing analysis was carried out through the use of genetic analyzer of an Applied Biosystems. The data was obtained in the form of chromatograms, and this chromatogram peaks were analyzed utilizing blast of the rs662 polymorphism using Bioedit tools. According to the studies of Alharbi et al (Alharbi et al., 2021), DNA sequencing was conducted utilizing both sense and antisense oligonucleotides.
2.7 Quality control
DNA extraction, PCR, RFLP, and agarose gel electrophoresis were performed at the KSU male campus of the Department of Clinical Laboratory Sciences, College of Applied Medical Sciences, in this study (Genetics laboratory: G141/1). To validate the accuracy of our PCR-RFLP results, Sanger sequencing analysis was conducted outside of G141/1 laboratory, and DNA sequencing analysis verified the accuracy of our PCR-RFLP results.
2.8 Clinical and Q192R data analysis
Data was compiled into Excel and then converted to statistical packages for the social sciences (25th version). The genotype frequencies in controls were determined using the χ2 test. Hardy-Weinberg equilibrium (HWE) for the rs662 polymorphism in the control group was determined. The mean and standard deviation are shown as continuous variables, and percentages are verified as categorical variables. Chi-squared analysis (χ2) on both observations and predicted data collected in this study was conducted. Table 2 lists the clinical characteristics of the participants in this study. The genotype/allele frequencies in female infertility cases and normal controls were calculated using 95 % CIs and odds ratios. Statistical analysis was performed on the cases and controls using dominant, recessive, and co-dominant forms, and the results were imported into Table 3. Table 4 shows the one-way analysis of variance (ANOVA) (Khan, 2019) that was performed to assess the relationship between the rs662 polymorphism and BMI in female infertility cases. Fig. 3 shows a scatter plot of genotyping frequencies in female infertility cases and healthy controls, and a box plot presents the relationship between rs662 polymorphism genotypes and BMI in female infertility cases (Fig. 4). The significance value of p < 0.05 was considered to be the cut-off point.
Variables
Infertile cases (n = 122)
Fertile cases (n = 100)
P value
Age (Years)
32.7 ± 5.9
28.3 ± 6.8
0.0001
Weight (kgs)
78.6 ± 12.5
73.8 ± 13.3
0.006
Height (cms)
157.8 ± 5.4
158.1 ± 6.4
0.70
BMI (kg/m2)
31.4 ± 4.3
28.8 ± 4.4
0.00001
Women effected with infertility
122 (100 %)
0 (0 %)
<0.0001
Family History
71 (58.2 %)
0 (0 %)
<0.0001
Gene
Polymorphism
Allele
Frequency
OR
Sample size (Case/control)
HWE
P value
PON1
rs662
R
32 %
1.9
122/100
9.76
0.001
Allele and Genotype frequencies
Cases (n = 122)
Controls (n = 100)
ORs/95 % CIs
P-value
Q allele
165 (0.68)
160 (0.80)
Reference
Reference
R allele
79 (0.32)
40 (0.20)
1.91 (1.23–2.96)
0.003
QQ genotype
58 (47.5 %)
69 (69 %)
Reference
Reference
QR genotype
49 (40.2 %)
22 (22 %)
2.65 (1.43–4.88)
0.001
RR genotype
15 (12.3 %)
9 (9 %)
1.98 (0.80–4.86)
0.13
Dominant (QR + RR vs QQ)
107 (87.7 %)
91 (91 %)
2.38 (1.31–4.31)
0.003
Recessive (RR vs QQ + QR)
15 (12.3 %)
9 (9 %)
0.70 (0.29–1.68)
0.43
Co-dominant (QR vs QQ + RR)
49 (40.2 %)
22 (22 %)
1.41 (1.31–4.31)
0.003
Anthropometric measurements
QQ (n = 58)
QR (n = 49)
RR (n = 15)
P-value
Age (Years)
32.3 ± 5.6
33.7 ± 5.9
31.1 ± 7.1
0.25
Weight (kgs)
80.6 ± 12.3
78.4 ± 12.6
71.8 ± 10.8
0.04
Height (cms)
158.1 ± 5.8
158.1 ± 4.9
155.3 ± 4.8
0.16
BMI (kg/m2)
32.1 ± 4.3
31.04 ± 4.3
29.6 ± 3.9
0.10
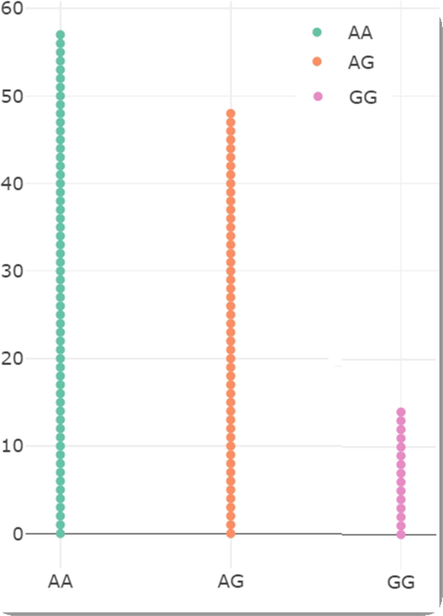
Scatterplot represents the genotypes in female infertility cases in Q192R polymorphism.
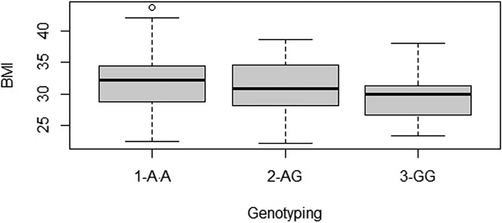
Boxplots depict the relationship between Q192R genotyping and BMI in female infertility women.
3 Results
3.1 Clinical details
This case-control study in local Saudi women was conducted with 222 participants recruited after the inclusion and exclusion requirements were verified, consisting of 122 infertile women and 100 fertile women. Table 1 illustrated the anthropometric and clinical characteristics of both cases and controls. The mean age of fertile (28.3 ± 6.8) and infertile cases (32.7 ± 5.9) was equivalent (p = 0.0001). However, there was variation between weight (p = 0.006) and BMI in both the cases (31.4 ± 4.3) and in controls (28.8 ± 4.4) (p = 0.00001). All of the cases in this study were caused by female infertility (women who have trouble conceiving due to infertility), and the control group had no infertility problems (p < 0.0001). The family history of 58.2 % was documented in the cases and controls was recruited without any family history which was one of the inclusion criteria (p < 0.0001). The details were showed in Table 1.
3.2 HWE analysis of rs662 polymorphism
Based on the HWE principle, the control genotypes were calculated using excel and genotype frequencies for Q192R polymorphism in the PON1 gene was found to be in accordance with HWE with the obtained p value (p = 0.001). Table 2 describes the details of HWE analysis in detail.
3.3 Genotyping analysis of Q192 polymorphism
Table 3 shows the allele and genotype frequencies found in female infertility cases and control women in the Saudi population (Fig. 3). The genotyping distribution in the control population was QQ (69 %), QR (22 %), and RR (9 %), while in the infertile cases it was QQ (47.5 %), QR (40.2 %), and RR (12.3 %). Frequencies of alleles in a control sample indicated that the Q allele (80 %) was more common than the R allele (20 %) and that the R allele (70 %) was more frequent than the Q allele (68 %) in the female infertile group (32 %). When comparing cases and controls, the allele frequency was found to be associated (OR-1.91; 95 % CI [1.23–2.96]; p = 0.003). Heterozygous genotype was associated when compared with Homozygous QQ genotype (QR vs QQ- OR-2.65; 95 % CI [1.43–4.88]; p = 0.001). Among the genetic models, both dominant (OR-2.38; 95 %CI [1.31–4.31]; p = 0.003). There was an association between and co-dominant models (OR-1.41; 95 %CI [1.31–4.31]; p = 0.003). When comparing infertile and fertile women, the QR genotype (p = 0.13) and recessive model (p = 0.43) were not associated.
3.4 Anova analysis
Anova analysis was performed between anthropometric characteristics (documented in Table 4) Age (p = 0.25), weight (p = 0.04), height (p = 0.16), and BMI (p = 0.10), as well as QQ (n = 58), QR (n = 49), and RR (n = 15) genotypes in female infertility cases. This study results demonstrates Weight was associated with QQ genotypes was elevated (80.6 ± 12.3) among 3 the modes of genotyping and when compared with QR (78.4 ± 12.6) and RR (71.8 ± 10.8) genotypes.
4 Discussion
The current study was designed to assess the relationship between Q192R polymorphism and female infertility in Saudi women. The current study results confirmed the strong genetic association with allele (p = 0.003), QR genotype (p = 0.001), and genetic model of inheritance as dominant (p = 0.003) models. In this study, the family history of female infertility was documented with 58.2 % and 100 % of participated infertile women was affected with infertility (p < 0.001). Anova analysis revealed weight was significantly associated within the various forms of genotypes (p = 0.04).
Female infertility is a consequence of fertile aged women undergoing premature menopause, as well as the increasing incidence of intentionally delayed pregnancy, at least in Western countries (Zorrilla and Yatsenko, 2013). Three-quarters of infertility is caused by female factors, and one-quarter is caused by male factors. Infertility, on the other hand, may be described by both male and female causes. Female infertility is difficult to diagnose and there are many treatment choices for infertility, depending on the root cause. It is well understood that infertile couples would return to parenthood in the absence of medical care. After couple of years, nearly 95 % of couples are effective in getting pregnant. A multifactorial heritage trend has been established for female infertility and genetic factors play a decisive role in disease predisposition (Turner, 2020). The prevalence of female infertility varies within the ethnicity.
Globally, of infertility is present in a very large percentage of Southern-East Asian regions and Sub-Saharan countries of Africa (Elhussein, 2019). Al-Turki et al (Al-Turki, 2015) studies confirmed the prevalence of obesity was documented to be 18.9 % in the Saudi women, while China has 15.5 %, Canada is about 11.5 %-15.7 %, and the UK is 12.5 % ADDIN EN.CITE < EndNote><Cite><Author > Alamri1</Author><RecNum > 1593</RecNum><DisplayText>(Alamri, n.d.)</DisplayText><record><rec-number > 1593</rec-number><foreign-keys><key app=“EN” db-id=“9vtstdtxxa0asge2w5fv0awrp0drxerraspa” timestamp=“1617923235″>1593</key></foreign-keys><ref-type name=”Journal Article“>17</ref-type><contributors><authors><author > Alamri1, Aladin Alhadi</author><author > Tarifi1, Ayman Kh</author><author > Alanazi, Sarah Musharraf</author><author > Alshammari, Nujud Muteb</author><author > Alenezi, Bdoor Attaulla Fahes</author><author > Mater, Reem Farhan</author><author > Alanzi, Maha Thiab Abdalaziz Alshbiny</author><author > Alshaleikhi, Etizaz Ayed Saud</author></authors></contributors><titles><title > Causes and risk factors of infertility among women of Arar city, Northern Saudi Arabia: a hospital-based study</title><secondary-title > Age</secondary-title></titles><periodical><full-title > Age</full-title></periodical><pages > 1.1</pages><volume > 20</volume><number > 6</number><dates></dates><urls></urls></record></Cite></EndNote>(Alamri, n.d.). Obesity is especially common in infertile women, and is related to infertility. Females who are overweight or obese have a much higher risk of having an unintended pregnancy. This raises the likelihood of subfecundity and sterility, as well as the risk of conception, miscarriage, and pregnancy complications (Dağ and Dilbaz, 2015). Previous studies has found a relation between obesity and female infertility. Obese women consume pregnancy for a longer period of time, according to two large cohort studies of Danish women, which revealed a decline in fecundability ratios with increasing BMI and Surprisingly, obese women remain infertile even in the absence of ovulatory dysfunction (Ramlau-Hansen, 2007; Wise, 2010). Obese women undergoing in vitro fertilization have atypically atrophied oocytes, which reduce oocyte fertility. Adipose tissue (obesity) also seems to influence the results of assisted reproductive technology, suggesting that the pathology extends beyond an ovulatory disorder. It seems that as shown in numerous studies to have an adverse effect on live birth rate and may be dependent on weight, and/is correlated with higher BMI (Broughton and Moley, 2017). In our study, the mean of the infertile was documented to be 31.4 kg/m2 when compared within the fertile women. When Anova analysis were conducted between the Q192R polymorphism genotypes and BMI in infertile women, negative associations were achieved (p = 0.10), despite the fact that weight was positively reported in these genotypes (p = 0.04).
PON1 (human serum paraoxonase-1) is a calcium-dependent hydrolytic enzyme found in many mammalian organisms that belongs to the triplet serum paraoxonase family (PON1, PON2 and PON3). With 354 amino acids and a molecular mass of 43 kDa, PON1 is the most well-known member of this family. These polymorphisms can also influence disease development risk and severity. A couple of common polymorphisms were established with PON1 activity and concentration between codons 55 and 192. The PON1 isoform Q192 was found to hydrolyze paraoxon and oxidize LDL more efficiently than the R192 isoform, suggesting that the polymorphism of glutamine/arginine at position 192 affects PON1 function. It was shown that this isoform had an effect. QQ patients are more likely to prevent LDL oxidation than RR patients, and this is thought to be due to the Q192R polymorphism, which is the key oxidant-based biomarker (Shunmoogam et al., 2018). Genetic variants in the PON1 gene can influence oxidative stress and therefore female infertility susceptibility. Oxidative stress, also known as a pro-oxidant-antioxidant mismatch, is important for the proper functioning of the female reproductive system as well as the development of female reproductive diseases. The influence of oxidative stress on male infertility is well defined, but the results are largely unknown on female reproductive disorders (Motovali-Bashi et al., 2015). Based on the function of PON in oxidative stress and the impact of genetic variants of PON that may be effective on female germ cells and fertility, furthermore, the relationship between Q192R polymorphism and susceptibility to female infertility was investigated in this study. (Mashayekhi, 2015). Limited studies was documented with the combination of Q192R and L55M polymorphisms in female infertility (Motovali-Bashi et al., 2015; Mashayekhi, 2015), recurrent miscarriages (Pandey, 2020), recurrent pregnancy loss (Öztürk, 2019), obesity (Veiga, 2011) and meta-analysis studies performed in PCOS women (Jiang, 2020) in the global population. The genotype frequencies between Mashayekhi et al (Mashayekhi, 2015) and current study results varies as Mashayekhi et al (Mashayekhi, 2015) confirms 38.6 % as QQ, 53.4 % as QR and 8 % as RR genotypes and coming to the present study results QQ genotype was documented to be 47.5 %, 40.2 % for QR and 12.3 % for RR. The RR genotype was documented to be high prevalence in this present study when compared with the previous study.
This study certain strengths and limitations. The strength of this present study was carried out in the Saudi women and Q192R polymorphism showed relation with female infertility. This might also be connected with obesity which was documented in the infertile women. The combination of female infertility and obesity might have associated with Q192R polymorphism in the Saudi women. This study was performed with single SNP, skipping the protein and serum studies. Finally, couldn’t record the patient’s data in detail which was one of the limitations of this study.
5 Conclusion
Finally, this study concludes as Q192R polymorphism was associated with female infertility in the Saudi women. However, this study was agreement with the previous studies implemented in other global population with various kinds of infertility issues in the women. Based on the availability of patient involvement and recruitment of samples during COVID-19, limited samples were involved in this study. This study recommends to perform the similar study in the global population to rule out the relation between female infertility, obesity and Q192R polymorphism. Large sample sized case-control studies were recommended. Additionally, it should also evaluate other potential genetic risk factors interacting with additional PON1 polymorphisms.
Acknowledgement
This Research Project was supported by a grant from the “Research Center of the Female Scientific and Medical Colleges”, Deanship of Scientific Research, King Saud University.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Alamri1, A.A., et al., Causes and risk factors of infertility among women of Arar city, Northern Saudi Arabia: a hospital-based study. Age. 20(6): p. 1.1.
- Relationship between the paraoxonase 1 gene glutamine 192 to arginine polymorphism and gestational diabetes mellitus in Saudi women. Clin. Biochem.. 2014;47(15):122-125.
- [Google Scholar]
- Q192R polymorphism in the PON1 gene and familial hypercholesterolemia in a Saudi population. Ann. Saudi Med.. 2017;37(6):425-432.
- [Google Scholar]
- Amino-acid amendment of arginine-325-tryptophan in rs13266634 genetic polymorphism studies of the SLC30A8 gene with type 2 diabetes-mellitus patients featuring a positive family history in the Saudi population. J. King Saud Univ.-Sci.. 2021;33(1):101258
- [Google Scholar]
- Interaction with angiotensin-converting enzyme-encoding gene in female infertility: insertion and deletion polymorphism studies. Saudi J. Biol. Sci.. 2018;25(8):1617-1621.
- [Google Scholar]
- Role of the Q36R polymorphism in the KISS1 gene in female infertility. J. King Saud Univ.-Sci.. 2023;35(2):102442
- [Google Scholar]
- Screening of obese offspring of first-cousin consanguineous subjects for the angiotensin-converting enzyme gene with a 287-bp Alu sequence. J. Obesity Metab. Syndrome. 2021;30(1):63.
- [Google Scholar]
- Prevalence of primary and secondary infertility from tertiary center in eastern Saudi Arabia. Middle East Fertil. Soc. J.. 2015;20(4):237-240.
- [Google Scholar]
- Infertility and pregnancy loss in euthyroid women with thyroid autoimmunity. Gynecol. Endocrinol.. 2013;29(1):36-41.
- [Google Scholar]
- The effects of PON1 gene Q192R variant on the development of uterine leiomyoma in Turkish patients. vivo. 2015;29(2):243-246.
- [Google Scholar]
- Barbieri, R.L., Female infertility, in Yen and Jaffe's Reproductive Endocrinology. 2019, Elsevier. p. 556-581. e7.
- Obesity and female infertility: potential mediators of obesity's impact. Fertil. Steril.. 2017;107(4):840-847.
- [Google Scholar]
- Impact of obesity on infertility in women. J. Turk. German Gynecol. Assoc.. 2015;16(2):111.
- [Google Scholar]
- Epidemiology of infertility and characteristics of infertile couples requesting assisted reproduction in a low-resource setting in Africa, Sudan. Fertil. Res. Pract.. 2019;5(1):1-5.
- [Google Scholar]
- Single nucleotide polymorphisms in Renalase and KCNQ1 genes and female infertility: a cross-sectional study in Pakistan. Andrologia. 2019;51(10):e13434.
- [Google Scholar]
- Association of Paraoxonase 1 gene polymorphisms and polycystic ovarian syndrome susceptibility: a systematic review and meta-analysis. Gynecol. Obstetric Investig.. 2020;85(2):167-177.
- [Google Scholar]
- Genetic confirmation of T2DM meta-analysis variants studied in gestational diabetes mellitus in an Indian population. Diabetes Metab. Syndr.. 2019;13(1):688-694.
- [Google Scholar]
- A new molecular variant of luteinizing hormone associated with female infertility. Fertil. Steril.. 1998;69(1):102-106.
- [Google Scholar]
- The association of PON1 192 Q/R polymorphism and the risk of female infertility. Cell. Mol. Biol.. 2015;61(2):74-77.
- [Google Scholar]
- Coronary artery disease and the frequencies of MTHFR and PON1 gene polymorphism studies in a varied population of Hyderabad, Telangana region in south India. J. King Saud Univ.-Sci.. 2015;27(2):143-150.
- [Google Scholar]
- Exposure to environmental toxins in males seeking infertility treatment: a case-controlled study. Reprod. Biomed. Online. 2008;16(6):842-850.
- [Google Scholar]
- Association between serum paraoxonase 1 activities (PONase/AREase) and L55M polymorphism in risk of female infertility. Avicenna J. Med. Biotechnol.. 2015;7(4):173.
- [Google Scholar]
- Paraoxonase-1 polymorphisms (L55M/Q192R) and activities (PONase/AREase) in patients with idiopathic recurrent early pregnancy loss: a preliminary study. Genet. Test. Mol. Biomarkers. 2019;23(7):501-505.
- [Google Scholar]
- Link between PON 1 gene mutation and recurrent miscarriage among women exposed to pesticides in North India is insignificant. Bioinformation. 2020;16(10):779-785.
- [Google Scholar]
- Paraoxonase (PON)-1: a brief overview on genetics, structure, polymorphisms and clinical relevance. Vasc. Health Risk Manag.. 2018;14:137.
- [Google Scholar]
- KISS1R polymorphism rs587777844 (Tyr313His) is linked to female infertility. Br. J. Biomed. Sci.. 2021;78(2):98-100.
- [Google Scholar]
- A study on the role of estrogen receptor gene polymorphisms in female infertility. Genet. Test. Mol. Biomarkers. 2016;20(11):692-695.
- [Google Scholar]
- Tantengco, O.A.G., M. de Castro Silva, and C.L. Velayo, The Role of genital Mycoplasma Infection in female infertility: A systematic Review and meta‐analysis. American Journal of Reproductive Immunology: p. e13390.
- Microbiota and Human reproduction: the case of female infertility. High-Throughput. 2020;9(2):12.
- [Google Scholar]
- Male infertility is a women’s health issue—research and clinical evaluation of male infertility is needed. Cells. 2020;9(4):990.
- [Google Scholar]
- Endocrine disorders & female infertility. Best Pract. Res. Clin. Endocrinol. Metab.. 2011;25(6):861-873.
- [Google Scholar]
- Q192R polymorphism of the paraoxonase-1 gene as a risk factor for obesity in Portuguese women. Eur. J. Endocrinol.. 2011;164(2):213-218.
- [Google Scholar]
- An internet-based prospective study of body size and time-to-pregnancy. Hum. Reprod.. 2010;25(1):253-264.
- [Google Scholar]
- The genetics of infertility: current status of the field. Curr. Genet. Med. Rep.. 2013;1(4):247-260.
- [Google Scholar]







