Translate this page into:
Entomopathogenic nematodes for the control of oriental fruit fly Bacterocera dorsalis (Diptera: Tephritidae)
⁎Corresponding authors. atif_entomologist@giabr.gd.cn (Atif Idrees), junl@giabr.gd.cn (Jun Li)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Background
Fruit fly species are most damaging pests around the globe which reduced the commercial value of fruits at maturity. Entomopathogenic nematodes (EPNs) from genera Heterorhabditis and Steinernema cause death by inducing septicimia in insect pests in the soil, moreover, endemic nearly all soils. Current study was planned to manage the oriental fruit fly, Bacterocera dorsalis (Hendel.) (Diptera: Tephritidae) hazards by using indigenous EPNs (Heterorhabditis bacteriophora, H. indica, Steinernema asiticum, S. corpocapsae and S. glasseri) as they have host finding ability and recognise as potential eco-friendly biocontrol agent over synthetic chemicals.
Methods
Study for the assessment of EPNs concentrations, various temperatures, soil type and soil moisture levels against mortality (%) of fruit fly (B. dorsalis) larvae was conducted in completely randomized design (CRD) under factorial arrangements.
Results
EPNs concentrations (70 IJs/ml, 110 IJs/ml, 150 IJs/ml) showed that S. asiaticum (150 IJs/ml) showed maximum mortality (94.97 %) of B. dorsalis as compared to other used EPNs along with their respective concentrations. Impact of various temperatures (20 °C, 24 °C, 28 °C, 32 °C, 36 °C) with respect to used EPNs exhibited that H. indica (36 °C) showed maximum mortality (94.33 %) of B. dorsalis as compared to all other treatment. Various soil types (Sandy, Sandy loam, Loam, Clay) impacted the infectivity of EPNs against fruit fly, In case of sandy loam soil, S. asiaticum showed maximum mortality (98.05 %) of B. dorsalis followed by all used treatments. Soil moisture level (12 %, 18 %, 24 %, 30 %) also significantly influenced the infectivity of EPNs against mortality of fruit fly. In case of 12% moisture level, S. asiaticum showed maximum mortality (99.06 %) of B. dorsalis over all the applied treatments along with respective moisture levels.
Conclusion
In crux, Steinernema asiaticum higher concentration exhibited efficient control of fruit fly larvae in sandy loam soil with 12 % moisture level at 36 °C over the used EPN species. While application of EPNs against fruit fly is most suitable strategy to manage the fruit fly hazards and it should be included as a part of integrated pest management control programme.
Keywords
Biological control
Fruit fly
Mortality
Soil moisture
Soil temperature
Soil type
1 Introduction
Fruit flies are damaging pests around the globe which reduced the commercial value of fruits at maturity (Grechi et al. 2021). It is declared as quarantine pest among all fly-free regimes due to larval infestation in commercial fruit packages and international trading (Mwando et al. 2021). Numerous genera and species of invasive fruit flies (Tephritidae) are known to be problems in orchards (Diller et al. 2022). The majority of the fruit flies complete their life cycle between 21 and 40 days under similar climatic conditions. In tropical and subtropical conditions, fruit flies multiply and spread quickly to distant fields or locations (Saeed et al. 2022). Khan and Naveed (2017) conclude that fruit fly population correlated positively with temperature (optimum 26 to 35 °C) and negatively with relative humidity (60 %).
When it comes to controlling arthropods, scientists are developing biopesticides like entompathogenic fungus that won't harm the environment or the insects' natural predators (Idrees et al., 2022a, 2022b, Ahmad et al., 2022, Idrees et al., 2021, Qadir et al., 2021, Idrees et al., 2016). The management of tephritidae fruit flies, particularly for the genus Bactrocera, has recently been effective thanks to the application of biocontrol agents (Luo et al., 2018, Gu et al., 2018, Cai et al., 2017) and certain molecular approaches (Zhang et al., 2019a, Zhang et al., 2019b).
Entomopathogenic nematodes (EPNs) from genera Heterorhabditis and Steinernema cause death by inducing infection in insect pest in the soil, moreover, endemic nearly all soils (Campos et al. 2016). Insect pests which show susceptible behavior towards EPNs attack failed to induce immunity (Peña et al. 2015; Filgueiras and Willett 2021). In laboratory conditions, mortality of fruit flies increased with increasing number of nematode infective juveniles (IJs) per fruit fly until maximum mortality was reached, beyond which mortality declined (Minas et al. 2016). This was attributed to the fact that intraspecific interference between infective juveniles of the same species can occur between actively moving IJs at EPN concentrations higher than the optimum IJ dose and may negatively affect efficacy of EPNs. In all reports, pupae of fruit flies largely proved to be more resistant than larvae to EPN infections (Aatif et al. 2019). However, some reports have shown that reasonable efficacy can be achieved if EPNs are allowed to attack all fruit-to-soil stages of fruit flies (Torrini et al. 2017). This suggests that fruit fly abundance can be reduced by virulent EPNs in the field. The infectivity of EPNs decreases with time-length of storage although stored infective juveniles can survive longer at temperatures between 4 and 15 °C. At temperatures near freezing, the infective juveniles become dormant (the dauer) and few may break dormancy after extended cold conditions. Between 20 and 30 °C, nematodes are more infective and reproduce faster. Reproduction and infection of the majority of EPN species are inhibited at temperatures above 32 °C. However, S. riobrave can reproduce at 32 °C and infect an insect at 39 °C (Aryal et al. 2022; Grewal et al. 2005). Also, EPNs respond to moisture regimes. The majority of EPN species are more active in moderate moisture regimes to field capacity of soils whereas others are desiccation tolerant (Aryal et al. 2022; Shapiro-Ilan et al. 2014).
Key objective of current trial was to evaluate most efficient entomopathogenic specie among H. bacteriophora, H. indica, S. asiticum, S. corpocapsae for their curative effect at various IJs concentrations, temperature, soil type and moisture levels against B. dorsalis, a step towards introduction of novel management strategy.
2 Methods
In vitro study of five different indigenous species of EPNs (H. bacteriophora, H. indica, S. asiticum, S. corpocapsae) and an exotic species (S.glasseri) taken from plant pathology laboratory UAF, Faisalabad against 3rd instar larvae of B. dorsalis was carried out in laboratory at Bahadur sub-campus Layyah. Mass production of G. mellonella were done initially on bee hives and at latter stages on artificial diet (Aatif et al. 2019). Rearing cages (size 18 × 20 × 20 in.) covered with metallic screen and filter paper were used.
Petri plates (9 cm) were taken and lined with filter paper and 5 larvae of G. mellonella were placed in each petri plate (Aatif et al. 2015). By using pipette each Petri plate was inoculated with ml of IJs suspension and sealed with covers. These Petri plates were then incubated at 24 ± 2 ⁰C. Larvae of G. melonella were died within 48 h of inoculation and were transferred for IJs extraction after regular intervals.
2.1 Effect of different concentrations of entomopathogenic Nematodes species against larvae of fruit fly (B. dorsalis)
The study's soil was oven-sterilized before being added to the Petri dishes (90 mm diameter). 20 g of soil with 10 % (v/w) moisture was added to each Petri plate. For each species of EPNs, separate Petri plates were filled with various concentrations (70, 110, and 150 IJs/ mL of the distilled water). Each Petri dish included fifteen B. dorsalis larvae in their third instar, arranged individually; the control treatment contained merely 250 l of distilled water (without EPNs). For each treatment, seven replications were created. Petri dishes were covered with plastic film during the experiment. These Petri plates were kept at 25 5 °C, 85 5 % relative humidity, and a 12-hour photoperiod in a well controlled environment (Minas et al. 2016). The experiment was carried out until all adult emergence in the control treatment had taken place (upto 15 days). By examining the deceased larvae, it was determined that EPNs killed the larvae.
2.2 Impact of different temperatures on effectiveness of EPNs against larvae of fruit fly (B. dorsalis)
Petri dishes (90 mm in diameter) lined with Whatman No. 1 filter paper were used for the experiment, and the temperatures used were 20, 24, 28, 32, and 36 °C. Each treatment was conducted in petri dishes containing 175 IJs. In each culture plate covered with Parafilm, fifteen B. dorsalis third-instar larvae were introduced. While just 250 l of distilled water is used in the control treatment (without EPNs). Mortality (%) data was obtained everyday for 10 days by simply counting the numbers that molted into adult stage across five replicates for each treatment. Dissecting the larvae that had been killed by EPNs provided conclusive evidence of their demise.
2.3 Efficacy of different soil types on the infectivity of EPNs against larvae of fruit fly (B. dorsalis)
Impact of different soil types (Sandy soil, Sandy loam soil, Loam soil, Clay soil) on the infectivity of EPNs against larvae of B. dorsalis were studied by using Petri dishes (90 mm diameter) in separate setts. Moisture level was maintained at 10 % (v/w) and 25 g of each type of sterilized soil was added to the Petri dishes separately. 200 IJs of each species of EPNs (H. bacteriophora, H. indica, S. asiaticum, S. corpocapsae and S. glasseri) were applied. Calculating the IJs in each Petri dish allowed for the concentrations to be produced under the microscope. All EPN species treatments included pipetting 1 mL of each dosage into the soil before 20 fruit fly larvae were put to Petri plates. In contrast, the control treatment merely received 250 l of distilled water (without EPNs). The Petri plates were covered with Parafilm and kept in climate-controlled chambers at 25 2 °C, RH = 70 5 %, and photo phase lasted 12 h. Five replications were taken for each treatment (Minas et al. 2016). Data was gathered as previously mentioned.
2.4 Effect of soil moisture levels on the virulence of EPNs against larvae of B. dorsalis
To assess the effectiveness of EPNs against fruit fly at different soil moisture levels, an experiment was conducted. In this experiment 190 IJs each species of EPNs (H. bacteriophora, H. indica, S. asiaticum, S. corpocapsae and S. glasseri) were applied separately. Moisture level was maintained at 10 % (v/w) and 25 g of sterilized soil was added to the Petri dishes separately. Four different moisture level (12 %, 18 %, 24 % and 30 %) were adjusted separately. Treatments were prepared under the microscope as described in earlier experiments. The larvae of fruit fly were applied by using fork. The Petri plates were closed by using plastic film, whereas, control treatment receives only 250 µl distilled water (without EPNs). Five replications of each treatment were maintained. These Petri plates were then placed in control chamber to provide controlled conditions (moisture and temperature). Data for insect mortality were recorded regularly for 10 days. Mortality of larvae by EPNs was confirmed by dissecting the dead larvae.
The corrected mortality (%) data was recorded by using Abbott’s formula (1925) given below
Mo = Mortality observed.
Mc = Mortality in control.
2.5 Statistical analysis
All experiments used a factorial configuration with five replicates and were done using a Completely Randomized Design (CRD). Statistical analysis was performed using Statistix 8.01 software. Analysis of variance (ANOVA) was used to compare means at P 0.05, and the least-significant-differences test was used to assess whether or not the differences were statistically significant (LSD).
3 Results
3.1 Effect of different concentrations of Entomopathogenic Nematodes species against larvae of fruit fly Bactrocera dorsalis
All EPNs viz. H. bacteriophora, H. indica, S. asiaticum, S. carpocapsae and S. glasseri along with their used concentrations (70 IJs/ml, 110 IJs/ml, 150 IJs/ml) significantly (p ≤ 0.05) impacted the mortality (%) of B. dorsalis.
Higher mortality (%) of B. dorsalis was assessed in case of S. carpocapsae (79.43 %) followed by H. bacteriophora (74.18 %), S. asiaticum (73.81 %) and S. glasseri (64.65 %), whereas, H. indica (64.58 %) exhibited minimum mortality of B. dorsalis (Table 1). Any two means followed by different letters are statistically significant at 95% probability level.
Factors
Mortality (%)
Treatment
Heterorhabditis bacteriophora
74.18B
Heterorhabditis indica
64.58C
Steinernema asiaticum
73.81B
Steinernema carpocapsae
79.39 A
Steinernema glasseri
64.65C
LSD Tr (p ≤ 0.05)
2.55
Concentration
70 IJs/ml
52.69C
110 IJs/ml
72.41B
150 IJs/ml
88.86 A
LSD (p ≤ 0.05)
1.97
Tr × C
4.42
Applied EPNs concentrations significantly (p ≤ 0.05) influenced the mortality (%) of B. dorsalis. Higher concentration (150 IJs/ml) exhibited maximum mortality (88.86 %) followed by 110 IJs/ml (72.41 %), whereas, mortality of B. dorsalis was minimum (52.69 %) in case of 70 IJs/ml applied concentration (Table 1).
Regarding interaction between used EPNs and concentrations significantly (p ≤ 0.05) impacted the mortality (%) of B. dorsalis. S. asiaticum (150 IJs/ml) showed maximum mortality of B. dorsalis followed by S. carpocapsae, H. bacteriophora, S. glasseri over H. indica, whereas, decreasing trend of mortality (%) of B. dorsalis viz. H. bacteriophora < S. carpocapsae < S. asiaticum < H. indica < S. glasseri was observed where 70 IJs/ml concentration was applied (Fig. 1).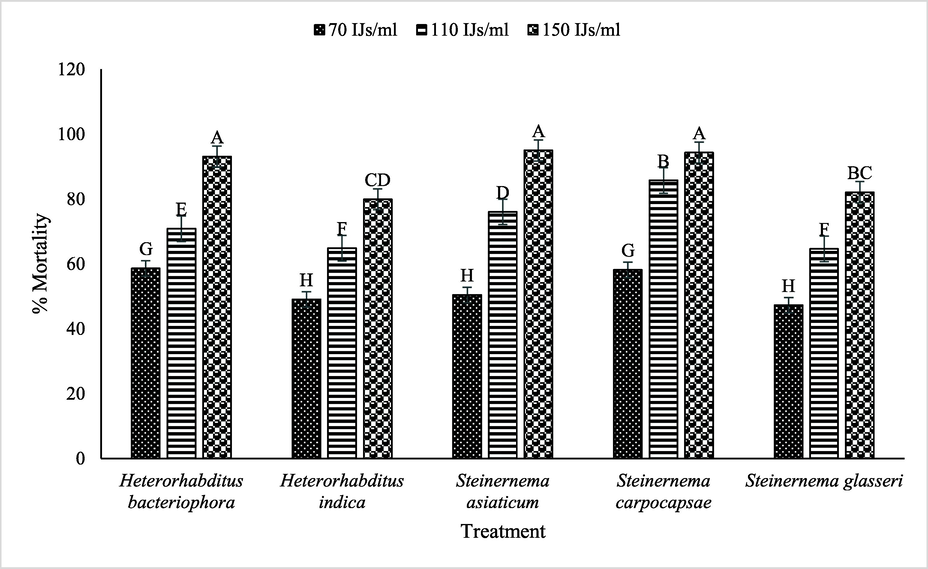
Interactive effect of various EPNs and concentrations on mortality of B. dorsalis.
3.2 Effect of different temperatures on infectivity of EPNs against larvae of fruit fly B. dorsalis
All EPNs viz. H. bacteriophora, H. indica, S. asiaticum, S. carpocapsae and S. glasseri along with various temperature like 20 °C, 24 °C, 28 °C, 32 °C and 36 °C significantly (p ≤ 0.05) impacted the mortality (%) of B. dorsalis.
Maximum mortality (%) of B. dorsalis was assessed in case of H. bacteriophora (79.54 %), S. asiaticum (77.21 %), H. indica (74.51 %) and S. glasseri (74.01 %), whereas, S. carpocapsae (70.88 %) exhibited minimum mortality of B. dorsalis (Table 2). Any two means followed by different letters are statistically significant at 95% probability level.
Factors
Mortality (%)
Treatment
Heterorhabditis bacteriophora
79.54 A
Heterorhabditis indica
74.51C
Steinernema asiaticum
77.21B
Steinernema carpocapsae
70.88 D
Steinernema glasseri
74.07C
LSD (p ≤ 0.05)
1.75
Temperature (T)
20 °C
52.39 E
24 °C
66.34 D
28 °C
76.30C
32 °C
88.30B
36 °C
92.89 A
LSD (p ≤ 0.05)
1.74
Tr × T
3.92
Mentioned temperatures significantly (p ≤ 0.05) influenced the mortality (%) of B. dorsalis. Higher temperature (36 °C) exhibited maximum mortality (92.89 %) followed by 32 °C (88.30 %), 28 °C (76.30 %) and 24 °C (66.34 %), whereas, mortality of B. dorsalis was minimum (52.39 %) in case of 20 °C temperature (Table 2).
Regarding interaction between used EPNs and temperature significantly (p ≤ 0.05) impacted the mortality (%) of B. dorsalis. H. indica (36 °C) showed maximum mortality of B. dorsalis followed by S. asiaticum, H. bacteriophora S. carpocapsae over S. glasseri, whereas, decreasing trend of mortality viz. H. bacteriophora < H. indica < S. asiaticum < S. glasseri < S. carpocapsae was observed where temperature was 20 °C (Fig. 2).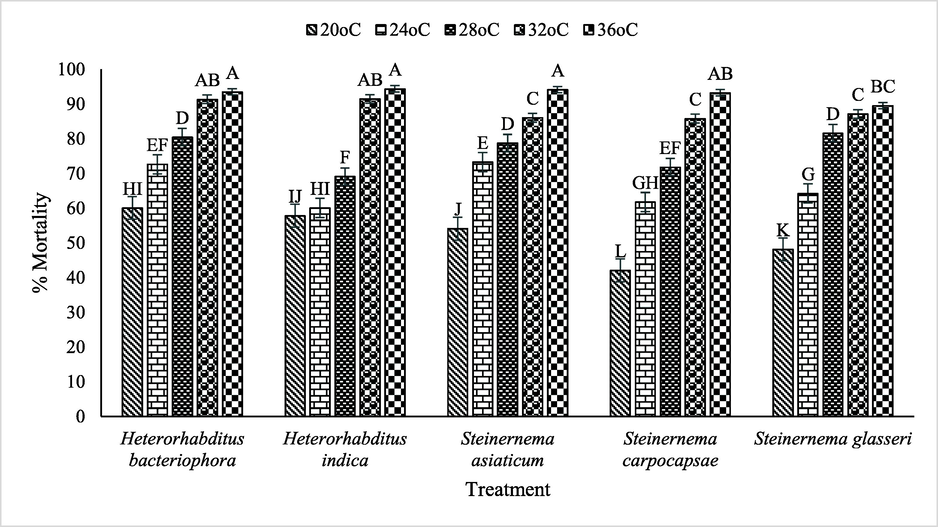
Interactive impact of various EPNs and temperature on mortality of B. dorsalis.
3.3 Effect of different soil types on infectivity of EPNs against larvae of fruit fly B. dorsalis
All EPNs viz. H. bacteriophora, H. indica, S. asiaticum, S. carpocapsae and S. glasseri along with used soils (Sandy soil, Sandy loam soil, Loam soil, Clay soil) significantly (p ≤ 0.05) impacted the mortality (%) of B. dorsalis.
Maximum mortality (%) of B. dorsalis was assessed in case of H. bacteriophora (73.44 %) followed by S. asiaticum (72.85 %), S. carpocapsae (69.15 %) and S. glasseri (65.30 %), whereas, H. indica (64.43 %) exhibited minimum mortality of B. dorsalis (Table 3). Any two means followed by different letters are statistically significant at 95% probability level.
Factors
Mortality (%)
Treatments
Heterorhabditis bacteriophora
73.44 A
Heterorhabditis indica
64.43C
Steinernema asiaticum
72.85 A
Steinernema carpocapsae
69.15B
Steinernema glasseri
65.30C
LSD (p ≤ 0.05)
2.52
Soil Texture
Sandy
43.39 D
Sandy Loam
91.36 A
Loam
79.00B
Clay
62.39C
LSD (p ≤ 0.05)
2.51
Tr × ST
5.04
Mentioned soil types significantly (p ≤ 0.05) influenced the mortality (%) of B. dorsalis. Sandy loam soils exhibited maximum mortality (91.36 %) followed by loam soil (79.00 %) and clay soil (62.39 %), whereas, mortality of B. dorsalis was minimum (43.39 %) in case of sandy soils (Table 3).
Regarding interaction between used EPNs and soil types significantly (p ≤ 0.05) impacted the mortality (%) of B. dorsalis. In case of sandy loam soil, S. asiaticum showed maximum mortality of B. dorsalis followed by H. bacteriophora, S. carpocapsae, S. glasseri over H. indica, whereas, decreasing trend of mortality viz. S. asiaticum < H. bacteriophora < S. carpocapsae < S. glasseri < H. ndica was observed in case of sandy soil (Fig. 3).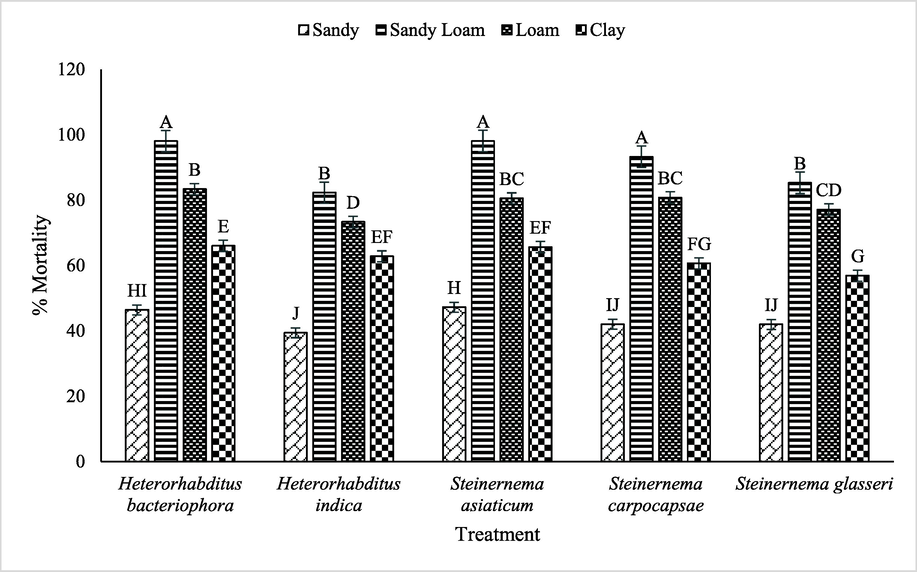
Impact of various EPNs and soil type on mortality of B. dorsalis.
3.4 Effect of different soil moisture levels on infectivity of EPNs against larvae of fruit fly B. dorsalis
All EPNs viz. H. bacteriophora, H. indica, S. asiaticum, S. carpocapsae and S. glasseri along with different moisture levels (12 %, 18 %, 24 %, 30 %) significantly (p ≤ 0.05) impacted the mortality (%) of B. dorsalis.
Maximum mortality (%) of B. dorsalis was assessed in case of S. asiaticum (77.58 %) followed by H. bacteriophora (75.82 %), S. carpocapsae (73.93 %) and H. indica (67.72 %), whereas, S. glasseri (65.66 %) exhibited minimum mortality of B. dorsalis (Table 4). Any two means followed by different letters are statistically significant at 95% probability level.
Factors
Mortality (%)
Treatment
Heterorhabditis bacteriophora
75.82 AB
Heterorhabditis indica
67.72C
Steinernema asiaticum
77.58 A
Steinernema carpocapsae
73.93B
Steinernema glasseri
65.66C
LSD (p ≤ 0.05)
2.25
Moisture Level (Ml)
12 %
89.95 A
18 %
89.35 A
24 %
59.33B
30 %
49.95C
LSD (p ≤ 0.05)
2.01
Tr × Ml
4.51
Mentioned moisture levels significantly (p ≤ 0.05) influenced the mortality (%) of B. dorsalis. Mortality (%) was higher (89.95 %) at 12 % moisture level followed by 18 % (89.35 %) and 24 % (59.33 %), whereas, mortality of B. dorsalis was minimum (49.95 %) in case of 30 % moisture level (Table 4).
Regarding interaction between used EPNs and moisture level significantly (p ≤ 0.05) impacted the mortality (%) of B. dorsalis. In case of 12 % moisture level, S. asiaticum showed maximum mortality of B. dorsalis followed by H. bacteriophora, S. carpocapsae, S. glasseri over H. indica, whereas, decreasing trend of mortality viz. H. indica < S. asiaticum < S. carpocapsae < H. bacteriophora < S. glasseri was observed in case of 30 % moisture level (Fig. 4).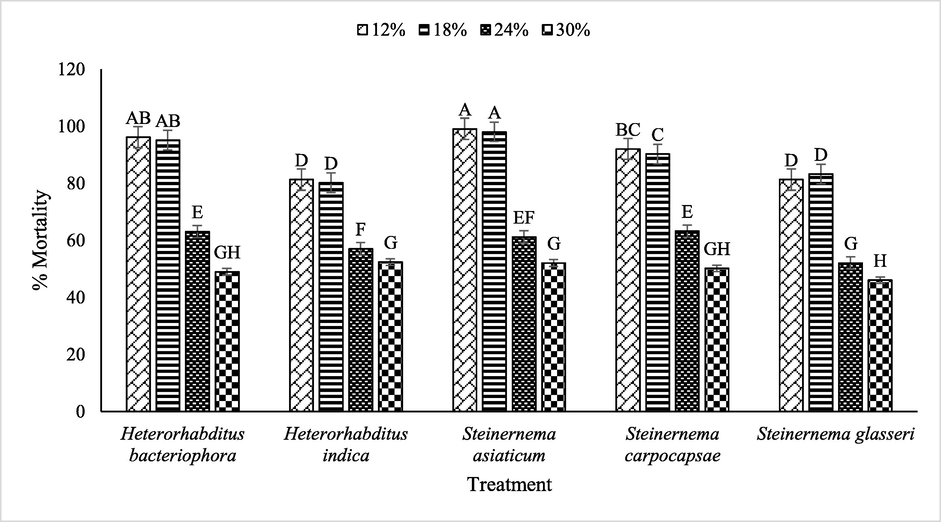
Interactive impact of various EPNs and moisture levels on mortality of B. dorsalis.
4 Discussion
Current study was planned to identify the most conducive climatic conditions required to evaluate the effectiveness of Entomopathogenic nematodes (EPNs). EPNs modulate their level of resistance in response to changing climatic conditions i.e. concentrations, temperature, moisture, soil texture etc. Results revealed that increase in IJs concentrations have significant impact on larval mortality of Bactrocera dorsalis. Some EPN strains may lack the virulence necessary to generate severe infections in the larval stage of some fruit fly species. Increasing their concentrations or applying them at the ideal temperature may boost their efficacy (Aatif et al. 2019; Langford et al. 2014). Impact of various temperatures with respect to pathogenicity of EPNs species, it was found that infectivity of EPNs increases with increase in temperature whereas negative impact was observed as the moisture increases. Application of S. feltiae considerably enhanced the mortality level of insects after exposure to EPNs at 20–25 °C temperature as compared to 15 °C temperature (Langford et al. 2013). H. bacteriophora significantly increased the mortality rate in maggots of B. dorsalis at above 30 °C temperature. Similarly, indiginious species of EPNs showed maximum infection level against R. cerasi fruit fly maggots by boosting temperature whereas, S. feltiae proved highly infectious EPN species at different IJs concentration and temperatures (Kepenekci et al. 2015). Heterorhabditis species have considerably high ability to indicate maximum level of virulence at 30 °C temperature (El Khoury et al. 2018). The EPN strains showed maximum infection at moderate temperature whereas, their level of pathogenicity greatly decreased below 20 °C. Heterorhabditis species are different as compared to Steinernema in their level of virulence due to their searching capacity. The S. carpocapsae are slower, whereas most of the Heterorhabditis species are fast moving (Foelkel et al. 2016). Thus temperature could be another factors, subsequently Steinernema species are considered highly virulent at 25 °C temperature, while at 30 °C temperature Heterorhabditis species showed high infectivity (Rohde et al. 2010).
Various soil types (Lankin et al. 2020; Khan et al. 2020) as well as different moisture levels (Mokrini et al. 2020) have strong influence on the infectivity of EPNs. In case of sandy loam soil, maximum mortality (%) of B. dorsalis were observed by all used treatments. Sandy loam soil might be favourable in nematode movement at 10–15 % moisture level (Mokrini et al. 2020). Similar results was observed by (Kamali et al. 2013) H. bacteriophora and S. carpocapsi exhibited different levels of mortality in Cucurbit fly, Dacus ciliates (Diptera: Tephritidae) for different soil types by changing the different environmental conditions. Soil moisture level also significantly influenced the infectivity of EPNs against mortality of fruit fly. In case of 12 % moisture level, all the tested species of EPNs exhibited the maximum mortality of B. dorsalis over all respective moisture levels.
Infesting fruit fly maggots move into the soil in the majority of orchards during the winter or late summer, when temperatures are too cool for EPNs application. EPNs may be placed beneath the canopy of fruit trees in early spring, when adult flies emerge from the soil at moderate temperatures (Shaurub et al., 2015), however this fact deserves additional investigation since temperature changes might alter the efficacy of EPNs and other biological control agents (Laznik and Trdan 2015). Because no single measure sufficiently mitigates fruit fly damage, combination of cultural management options and natural enemies may better reduce densities of fruit flies. Numerous natural enemies (parasites, parasitoids, predators, and entomopathogens) of fruit flies have been documented (Schliserman et al. 2016). Using EPNs in mango orchards infested by fruit fly should control the fruit-to-soil life cycle stages of the flies. Thus, studies are needed to establish the guiding principles for the use of EPNs in fruit fly IPM for Pakistan mango production.
5 Conclusions
In crux, study reveals that EPNs should be the part of IPM because they exhibited efficient mortality of fruit fly at various environmental conditions viz. temperature, moisture and soil texture.
Acknowledgement
This research study was funded by "GDAS Special Project of Science and Technology Development (No. 2020GDASYL-20200301003), Agricultural scientific research and technology promotion project of Guangdong Province (No. 2021KJ260), Science and Technology Planning Project of Guangdong (No. 2018B030324003). This study was funderd by Higher Education Commission Pakistan under the project (NRPU-7007). This project was supported by Researchers Supporting Project Number (RSP-2022R7) King Saud University, Riyadh, Saudi Arabia.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Effect of Temperature on the Reproductive Potential of Indigenous and Exotic Species of Entomopathogenic Nematodes inside Galleria mellonella L. Larvae. Int. J. Agric. Biol.. 2015;17:995-1000.
- [Google Scholar]
- Assessment of the entomopathogenic nematodes against maggots and pupae of the oriental fruit fly, Bactrocera dorsalis (Hendel) (Diptera: Tephritidae), under laboratory conditions, Egypt. J. Bio Pest Control. 2019;29:51.
- [CrossRef] [Google Scholar]
- A method of computing the effectiveness of an insecticide. J. Eco. Ento.. 1925;18(2):265-267.
- [Google Scholar]
- Synergized toxicity of promising plant extracts and synthetic chemicals against fall armyworm Spodoptera frugiperda (JE Smith) (Lepidoptera: Noctuidae) in Pakistan. Agronomy. 2022;12:1289.
- [Google Scholar]
- Virulence, penetration rate and reproductive potential of entomopathogenic nematodes from eastern Australia in Queensland fruit fly, Bactrocera tryoni. Biol. Control. 2022;169:104871.
- [Google Scholar]
- The optimal age and radiation dose for Bactrocera dorsalis (Hendel) (Diptera: Tephritidae) eggs as hosts for mass-reared Fopius arisanus (Sonan) (Hymenoptera: Braconidae) Bio Control. 2017;108:89-97.
- [Google Scholar]
- Entomopathogenic nematode food web assemblages in Florida natural areas. Soil Biol. Biochem.. 2016;93:105-114.
- [Google Scholar]
- A real-time remote surveillance system for fruit flies of economic importance: sensitivity and image analysis. J. Pest. Sci. 2022
- [CrossRef] [Google Scholar]
- Effect of temperature on the pathogenicity of Mediterranean native entomopathogenic nematodes (Steinernematidae and Heterorhabditidae) from natural ecosystems. Redia. 2018;101:123-127.
- [Google Scholar]
- Non-lethal effects of entomopathogenic nematode infection. Sci. Rep.. 2021;11(1):17090.
- [Google Scholar]
- Virulence of nematodes against larvae of the south American fruit fly in laboratory using soil from Porto Amazonas, Paraná, Brazil, as substrate. Cienc Rural. 2016;46:405-410.
- [Google Scholar]
- Linking mango infestation by fruit flies to fruit maturity and fly pressure: a prerequisite to improve fruit fly damage management via harvest timing optimization. Crop Prot.. 2021;146:105663.
- [Google Scholar]
- Nematodes as biological control agents. UK: CABI; 2005.
- The response of four braconid parasitoid species to methyl eugenol: optimization of a biocontrol tactic to suppress Bactrocera dorsalis. Biol. Control. 2018;122:101-108.
- [Google Scholar]
- Acaricidal potential of some botanicals against the stored grain mites, Rhizoglyphus tritici. J. Entomol. Zool. Stud.. 2016;4:611-617.
- [Google Scholar]
- Effectiveness of entomopathogenic fungi on immature stages and feeding performance of fall armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae) larvae. Insects. 2021;12:1044.
- [CrossRef] [Google Scholar]
- Bioassays of Beauveria bassiana isolates against the fall armyworm, Spodoptera frugiperda. J. Fungi. 2022;8:717.
- [CrossRef] [Google Scholar]
- Laboratory efficacy of selected synthetic insecticides against second instar invasive fall armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae) larvae. PLoS ONE. 2022;17(5):e0265265.
- [Google Scholar]
- Biocontrol potential of the entomopathogenic nematodes Heterorhabditis bacteriophora and Steinernema carpocapsae on cucurbit fly, Dacus ciliatus (Diptera: Tephritidae) Biocontrol Sci. Tech.. 2013;23(11):1307-1323.
- [Google Scholar]
- Evaluation of native entomopathogenic nematodes for the control of the European cherry fruit fly Rhagoletis cerasi L. (Diptera: Tephritidae) larvae in soil. Turk. J. Agric. For.. 2015;39:74-79.
- [Google Scholar]
- Occurrence and Seasonal Abundance of Fruit Fly, Bactrocera zonata Saunders (Diptera: Tephritidae) in Relation to Meteorological Factors. Pak J Zool. 2017;49(3):999-1003.
- [Google Scholar]
- The mortality rate of pupae and adult of fruit fly Bactrocera cucurbitae Coquillett (Diptera: Tephritidae) affected by different submerging time and soil types under the laboratory treatment. Sarhad J. Agric.. 2020;36(3):815-822.
- [Google Scholar]
- Susceptibility of Queensland fruit fly, Bactrocera tryoni (Froggatt) (Diptera: Tephritidae), to entomopathogenic nematodes. Bio Control. 2013;96:34-39.
- [Google Scholar]
- Susceptibility of Queensland fruit fly, Bactrocera tryoni (Froggatt)(Diptera: Tephritidae), to entomopathogenic nematodes. Biol. Control. 2014;69:34-39.
- [Google Scholar]
- Soil texture, infective juvenile concentration, and soil organic matter influence the efficacy of Steinernema feltiae isolate Lican Ray. J. Nematol.. 2020;52:1-11.
- [Google Scholar]
- Failure of entomopathogens to control white grubs (Coleoptera: Scarabaeidae) Acta Agri. Scand. Sect. B. 2015;65(2):95-108.
- [Google Scholar]
- Molecular identification of cultivable bacteria in the gut of adult Bactrocera tau (walker) and their trapping effect. Pest Manag. Sci.. 2018;74:2842-2850.
- [CrossRef] [Google Scholar]
- Potential of entomopathogenic nematodes (Rhabditida: Heterorhabditidae) to control Mediterranean fruit fly (Diptera: Tephritidae) soil stages. Nematoda 2016
- [CrossRef] [Google Scholar]
- Potential of Moroccan entomopathogenic nematodes for the control of the Mediterranean fruit fly Ceratitis capitata Wiedemann (Diptera: Tephritidae) Sci. Rep.. 2020;10:19204.
- [Google Scholar]
- Hot Water Treatment for Post-Harvest Disinfestation of Bactrocera dorsalis (Diptera: Tephritidae) and Its Effect on cv. Tommy Atkins Mango. Insects. 2021;12(12):1070.
- [Google Scholar]
- Variation in the susceptibility of Drosophila to different entomopathogenic nematodes. Infect. Immun.. 2015;83(3):1130-1138.
- [Google Scholar]
- Effectiveness of different soft acaricides against honey bee ectoparasitic mite Varroa destructor (Acari: Varroidae) Insects. 2021;12(11):1032.
- [Google Scholar]
- Influence of soil temperature and moisture on the infectivity of entomopathogenic nematodes (Rhabditida: Heterorhabditidae, Steinernematidae) against larvae of Ceratitis capitata (Wiedemann) (Diptera: Tephritidae) Neotrop Entomol.. 2010;39:608-611.
- [Google Scholar]
- Preference and performance of peach fruit fly (Bactrocera Zonata) and Melon fruit fly (Bactrocera Cucurbitae) under laboratory conditions. Saudi J. Biol. Sci.. 2022;29(4):2402-2408.
- [Google Scholar]
- Temporal diversity and abundance patterns of parasitoids of fruit-infesting Tephritidae (Diptera) in the Argentinean. Envi Entomol. 2016;45(5):1184-1198.
- [Google Scholar]
- Characterisation of biocontrol traits in Heterorhabditis floridensis: a species with broad temperature tolerance. J. Nematol.. 2014;46:336-345.
- [Google Scholar]
- Infectivity of four entomopathogenic nematodes in relation to environmental factors and their effects on the biochemistry of the Medfly Ceratitis capitata (Wied.) (Diptera: Tephritidae) Neotrop Entomol. 2015;44(6):610-618.
- [Google Scholar]
- Susceptibility of olive fruit fly, Bactrocera oleae (Diptera: Tephritidae) pupae to entomopathogenic nematodes. J. Plant Protect Res.. 2017;57:318-320.
- [Google Scholar]
- Molecular characterization of prophenoloxidase-1 (PPO1) and the inhibitory effect of kojic acid on phenoloxidase (PO) activity and on the development of Zeugodacus tau (Walker) (Diptera: Tephritidae) Bull. Entomol. Res.. 2019;109(2):236-247.
- [Google Scholar]
- Tyrosine hydroxylase is crucial for pupal pigmentation in Zeugodacus tau (Walker) (Diptera: Tephritidae) Compar Biochem. Physiol. Biochem. Mol. Biol. 2019;231:11-19.
- [Google Scholar]







