Translate this page into:
Effect of short-term heat stress on life table parameters of green peach aphid [Myzus persicae (Sulzer) (Hemiptera: Aphididae)]
⁎Corresponding authors. aroosakhurshid3@gmail.com (Aroosa Khurshid), liuchzh@gsau.edu.cn (Changzhong Liu)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Background
Insect populations and their dispersal can be altered by temperature changes. As a result, evaluating the effects of these changes on insect species' developmental stages is critical for establishing effective management measures.
Methods
This study examined the developmental duration, growth, longevity, fecundity, and other population parameters in two generations of green peach aphid (Myzus persicae) exposed to four different temperatures (i.e., 27 °C, 30 °C, 33 °C, and 36 °C) for four different time intervals (i.e., 1 h, 3 h, 6 h, and 10 h).
Results
The results indicated that nymph survival rate significantly decreased with exposure to 36 °C for 10 h in both generations (F1 and F2). The survival rate of 1st to 4th instars of F1 generation decreased by 85.0 %, 88.5 %, 85.7 %, and 83.7 % respectively at 36° C for 10 h exposure compared to the control (25 °C). Similarly, survival rate of 1st to 4th instars of F2 generation decreased by 83.7 %, 86.0 %, 82.2 %, and 78.7 %, respectively when exposed to 36 °C for 10 h compared to the control. The shortest longevity of both generations was observed under 33 °C and 36 °C exposure for 10 h. The highest and the lowest net reproductive rate (R0) for both generations was observed under exposure to 27 °C for 1 h and 36 °C for 10 h, respectively. The intrinsic rate of increase (rm) of the two generations reached the highest level under 27 °C exposure for 1 h and the lowest was recorded for the individuals exposed to 36 °C for 10 h. The highest finite rate of increase (λ = F1, 1.41 and F2, 1.42 d−1) for two generations was examined for the individuals exposed to 27 °C for 1 h. The values of life table parameters of both generations were significantly affected under exposure to 36 °C for 10 h.
Conclusion
The combined effect of different temperatures and time intervals on biological and life table parameters of M. persicae studied first time in this study. Therefore, our research will help in estimating the modifications that a population of M. persicae may experience in response to heat stress.
Keywords
M. persicae
Population parameters
Heat stress
Fecundity
Reproduction
- R0
-
Net reproductive rate
- rm
-
Intrinsic rate of increase
- T
-
Mean generation time
- GRR
-
Gross reproductive rate
- DT
-
Population doubling time
Abbreviations
1 Introduction
Aphids are heterotrophs; thus, temperature has a significant impact on their biological mechanisms. The increase in temperature increases their digestion rate and a reduces population doubling times. Aphids can be found all over the world in temperate climates (Hodek et al., 2012). The longevity of adults and oviposition of insects can be influenced by high temperatures (Mamay et al., 2022). The population dynamics and dispersal of insect species can be affected by temperature fluctuations. When insects are exposed to different temperatures, they react in several ways. Insect behavior, or physiological and morphological variation within same population might be the response to different temperatures (Khadioli et al., 2014).
In addition to increasing the average temperature, climate change will also increase the fluctuation of temperature regimes (Jalali et al., 2018; Zhao et al., 2017). It is predicted variability in temperature fluctuations will accelerate in the future (Ganguly et al., 2009). The mean temperature is rarely expected to approach ectotherms' optimum ranges in temperate climates. Extreme temperatures are highly affected by even small shifts in mean temperature. Temperature mostly affects growth rate, weight, size, fecundity, and metabolism of insects (Mamay et al., 2022; Wonglersak et al., 2020). Exothermic organisms such as insects and reptiles (arachnids, plants) have three types of temperature regimes, i.e., low, suitable, and the high. There are different suitable temperature ranges for different insect species. The developmental rate and lifespan of the bird cherry-oat aphid Rhopalosiphum padi were increased and shortened within 15–30 °C temperature range (Park et al., 2016). Studies on changes in life-history parameters show how aphids react to different climatic circumstances. Denaturation of proteins and enzymes causes apoptosis in cells exposed to high temperatures, resulting in changes in membrane structure and function.
Numerous insect species such as butterfly, drosophila, beetles (Klockmann et al., 2017), Diptera, Coleoptera, Odonata, and Blattaria differ ontogenetically in their behavior (Berger et al., 2011), thermal tolerance (Pincebourde and Casas, 2015), biological parameters (Mamay et al., 2022), growth rate, and metabolism rates (Folguera et al., 2010). Higher temperatures are expected to have stage-specific impacts on insects. The impacts of high temperatures can also differ throughout generation in the insects with very basic life cycles, such as true bugs and scale insects (Zhao et al., 2017). Additionally, high-temperature effects observed in earlier life span can be transmitted to subsequent phases and between generations. Temperature prevailing during the embryonic period could affect larvae growth, adult survivability and total weight, whereas the temperature during the initial development stages affects adults’ activities (MacLean et al., 2017), growth or even fertility (Zhao et al., 2017).
Myzus persicae Sulzer (Hemiptera: Aphididae) is an economically important pest specie that affects more than 50 plant species, particularly those belonging to Cruciferae, Leguminosae, and Solanaceae plant families. Moreover, it transmits >100 plant viruses (Blackman and Eastop, 2000).
Life tables are critical in population and community-based research. This table offers detailed information about a population's survival, development, and fecundity. The life table is the oldest and most significant tool in population ecology. Several researchers used life tables to investigate the impact of climatic conditions, pesticides, host plant species, secondary metabolites, cannibalism, and mother age on insects (Huang and Chi, 2012). Other factors, in addition to these, can affect the development rates, survival rates, and fertility of insect species. The most comprehensive and exhaustive explanation of the influence of various factors on certain biological traits is provided by life tables (Mamay et al., 2022). Despite the widespread acceptance of age-stage, two-sex life tables, some academics use female-specific life tables (Wei et al., 2020). It is useful to describe stage differentiation and the impact of male on population parameters in the age-stage, two-sex life table. Age-stage two-sex life tables are more useful than female age-specific life tables. Both sexes must be considered in a life table since they have an impact on population dynamics (Mamay et al., 2022). Traditional life tables are based on age and exclusively include data from women, hence the results may be erroneous. The age-stage, two-sex life table, on the other hand, accounts for the variation in development rates between individuals and the overall population, yielding more trustworthy results. Many studies have looked at insect development rates at different temperatures using temperature-dependent growth models. Biophysical mechanisms and quantitative analysis have been used to investigate the association between temperature and developmental performance. Life table experiments are more thorough than temperature-dependent production models.
The use of life tables is essential to analyze and evaluate an insect's overall biological performance under various environmental conditions. Temperature and aphid population growth and life cycle have been studied extensively, with a focus on taxonomy, morphology, regional distribution, and identifying traits. Previous work has been done on the effect of high and fluctuating temperatures on M. persicae (Davis et al., 2006) and the effect of temperature on its performance in different host plants (Abdel-Salam et al., 2009). In our previous work, the physiological and molecular responses of M. persicae were studied under short-term heat stress (Khurshid et al., 2021). However, our present study reports the impact of short-term heat stress and exposure times upon all life stages (first instar nymphs to adults) of two generations of M. persicae. This study aimed to determine the effect the life table parameters of M. persicae under different heat-stress levels in two generations. The purpose of this research was to develop a scientific foundation for improved knowledge and management of M. persicae under heat stress. This study will present the biological foundation for pest control methods based on short-term heat stress.
2 Materials and methods
2.1 Host plant and insect species
The green peach aphid (Myzus persicae) individuals were obtained from the potato research field Gansu Agricultural University, Lanzhou, China (36°05′22.9″N 103°42′01.0″E) and maintained in climate chambers under controlled conditions (25 °C with 16: 8 h light: dark photoperiod and 60 ± 5 % humidity). The research was carried out in Plant Protection laboratory, College of Plant Protection, Gansu Agricultural University Lanzhou, China. The M. persicae were reared for three successive generations on potato seedlings in growth chambers. Gansu Seeds Research Laboratory, China provided the host plant potato (Solanum tuberosum L.) (Solanaceae) tubers, watered on a regular basis, and placed in pots.
2.2 Insect rearing
The stock population, i.e., F0 generation of M. persicae female adults were selected randomly on potato leaf disks and exposed to different temperatures (27, 30, 33, and 36 °C) for nymphal production of F1 generation. Freshly hatched nymphs were put in 9-cm Petri dishes with wet filter sheets and placed to the bottom ends of detached potato leaves. Each temperature treatment consisted of 30 nymphs in a Petri dish (30 Petri dishes for each treatment). The same process was repeated for F2 generation.
2.3 Heat stress exposure
Temperatures from 27 to 36 °C and time intervals from 1 to 10 h were regarded as short-term heat stress. Since M. persicae has high level of resistance, we selected different temperatures above the ambient temperature 25 °C (control) to study the life history traits of M. persicae under short-term heat stress. Ma and Ma (2012) created a new parameter, drop-off temperature (DOT), to define the critical temperature at which an aphid leaves its host plant when the ambient temperature rises and discovered that adults starved for 12 h had higher DOT values than un starved or starved for 6 h. As a result, in this study heat stress exposure times were 1, 3, 6, and 10 h. In growth chambers, newly born nymphs in Petri dishes were independently subjected to 27, 30, 33, and 36 °C for 1, 3, 6, and 10 h (Khurshid et al., 2021). The treatments were carried out in four growth chambers, with temperatures controlled by a temperature controller that was programmed. The nymphs in the control group were kept at 25 °C.
2.4 Measurement of developmental duration, survival rate, adult longevity, and fecundity
The 1st instar nymphs were reared on potato leaves in Petri dishes till 4th instar. Nymph survival was observed on daily basis and the nymph developmental duration from 1st to 4th instar nymph was observed. The developmental progress of M. persicae at each temperature (27, 30, 33, and 36 °C) and each exposure time (1, 3, 6, and 10 h) was monitored two times daily (at 8:00 in the morning and 8:00 in the evening). For 4th instar nymph, we recorded the number of emerging adults. The numbers of 1st, 2nd, 3rd, and 4th instars nymph emerged into adults were recorded until the developmental stages were completed. The survival of nymphs and adults, pre-reproduction period (the interval between adult emergence and the production of first off-spring), fecundity (the number of off-springs produced by each female), and adult longevity were recorded. Every day, the filter papers in culture dishes were moistened with water and after every 2–3 days fresh potato leaves were supplied for nymphs. The same process was repeated for F2 generation (nymphs and adults) of M. persicae.
2.5 Data analysis
The age-stage, two-sex life table was used to analyze the life history data of M. persicae. The individual biological parameters, i.e., survival, nymph duration, adult aphid reproductive period, period of total pre-reproduction (from birth to producing the first nymph), fecundity (number of nymphs per female) and period of longevity (from birth to death), and population parameters were calculated by Chi and Liu (1985) using the method explained by Chi (1988) and using the computer program TWO SEX-MSChart (Chi, 2015).
Age-specific net reproductive is obtained from the product of lx and mx. A net reproductive rate (R0) is equal to lxmx divided by all ages. The average number of offspring an individual produce during their lifetime is defined as the R0. Net reproduction rate was calculated as.
R0 = ∑ lx mx
Intrinsic rate of increase (rm) is determined using the Euler–Lotka formula, and the age is indexed from the day “0″.
rm = ln R0 / T;
The daily rate of increase at which a stable age-stage distribution is reached is known as the finite rate (λ) and calculated as follows:
Finite rate of increase: λ = er.
The length of time that takes a population to reach R0-fold after reaching steady growth rates r and is known as the mean generation time (T). i.e., erT = R0 or T = R0, and it was calculated as:
Mean generation time (T) =∑ x lx mx / ∑ lx mx
Gross reproductive rate (GRR) = ∑ mx.
Population doubling time (DT) = ln2 / rm
where x is the number of days between days, lx denotes the probability of an adult survive during the period of x, and mx represents the average number of nymphs produced during the period of x. The bootstrap approach was used to calculate the means and standard errors of the developmental duration, longevity, fecundity, and population parameters. In this investigation, we performed 100,000 bootstrap replications to lessen the variability of the data. The statistical differences between treatments were analyzed using a paired bootstrap test with a significance level of 5 %.
3 Results
3.1 Survival rate of nymphal stages
The nymph survival rate decreased under exposure to 36 °C for 10 h in both generations. The nymph survival rate in F1 generation decreased by 85.0 %, 88.5 %, 85.7 %, and 83.7 %, for 1st to 4th instar, respectively compared to control. Similarly, the decrease in the survival rate of F2 generation was 83.7 %, 86.0 %, 82.2 %, and 78.7 %, respectively compared to control (Fig. 1).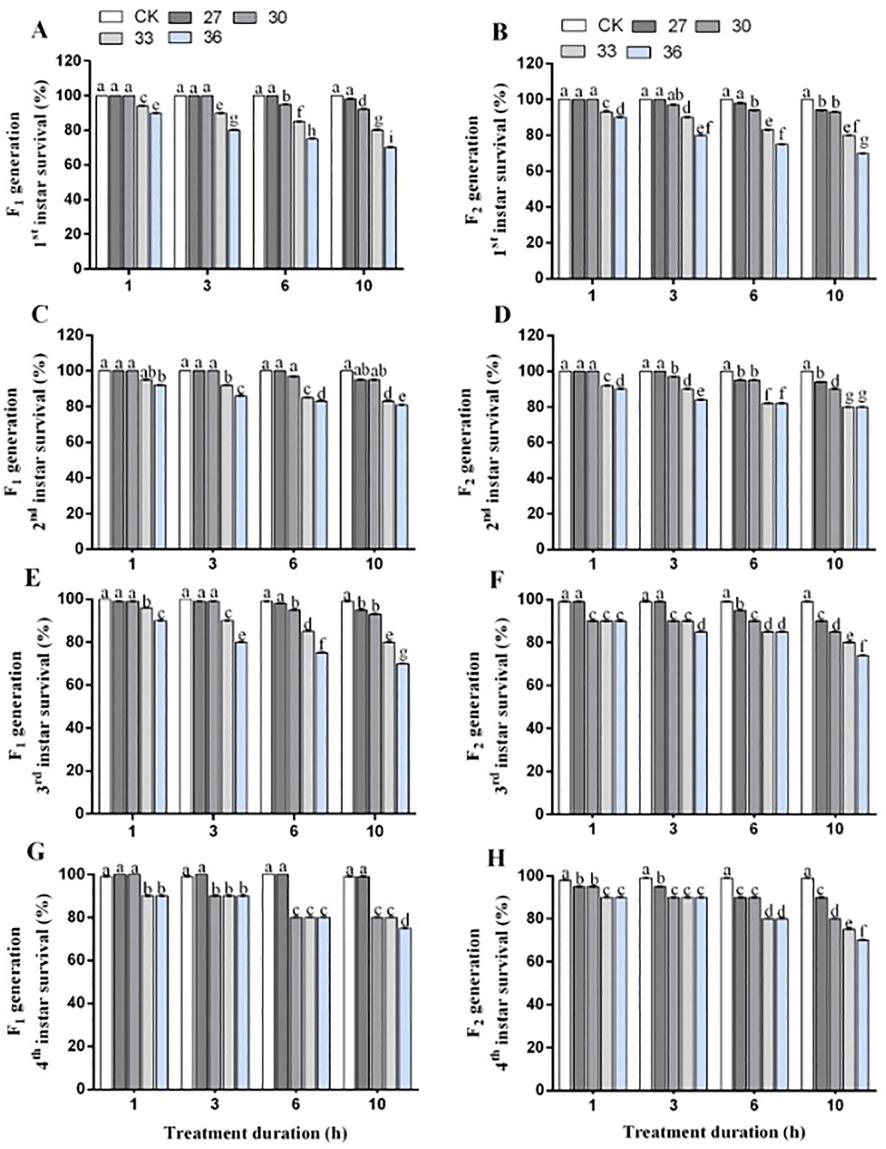
Survival rate of 1st (A-B), 2nd (C-D), 3rd (E-F), and 4th instar (G-H) nymphs of two generations of M. persicae exposed to different temperatures for different treatment durations. Different lowercase letters indicate that the means significantly differ at 95% probability determined by the paired bootstrap.
3.2 Survival rate of adults
The adult survival rate of two generations significantly decreased under 6 h and 10 h exposure to increasing temperatures, i.e., 27, 30, 33 and 36 °C. The 6 h exposure to 27, 30, 33 and 36 °C decreased adult survival of F1 generation by 99 %, 96 %, 87 %, 75 %, whereas 10 h exposure decreased the adult survival by 99 %, 95 %, 85 %, 70 %, respectively compared to control. Likewise, the exposure to 27, 30, 33 and 36 °C for 6 h decreased the adult survival in F2 generation by 98 %, 95 %, 83 % and 75 %, while the reduction under 10 h exposure was 98 %, 95 %, 81 % and 70 %, respectively. Meanwhile, there were no significant variations in the adult survival rate of two generations under exposure to same temperature for same time interval (Fig. 2).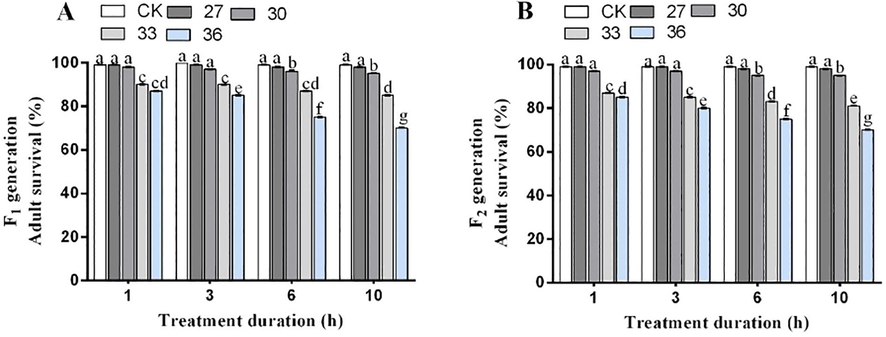
Survival rate of the adults in F1 (A) and F2 (B) generations of M. persicae exposed to different temperatures for differenttreatment durations. Different lowercase letters indicate significant differences among means at 95% probability level determined by the paired bootstrap.
3.3 Developmental duration of nymphal instars
The developmental duration for 1st nymphal instar of both generations significantly decreased under exposure to each temperature and time duration compared to control. The shortest developmental duration for 1st nymphal instar of both generations was 1.23 days (F1) and 1.0 days (F2) under exposure to 36 °C for 10 h compared to control (F1, 2.3 days, and F2, 2.2 days). Similar effect was observed on 2nd nymphal instars of both generations where the shortest developmental duration was recorded under exposure to 36 °C for 10 h (F1, 1.21 days and F2, 0.8 days) (Fig. 3). The developmental durations of 2nd, 3rd, and 4th nymphal instars in F1 generation were longer than F2 generation under exposure to 30 and 33 °C for 1, 3, 6, and 10 h (p < 0.05). The 3rd nymphal instar duration was the shortest among all developmental stages for both generations (F1, 1 day and F2, 0.7 days) and significantly decreased under exposure to 36 °C for 10 h compared to control (Fig. 3).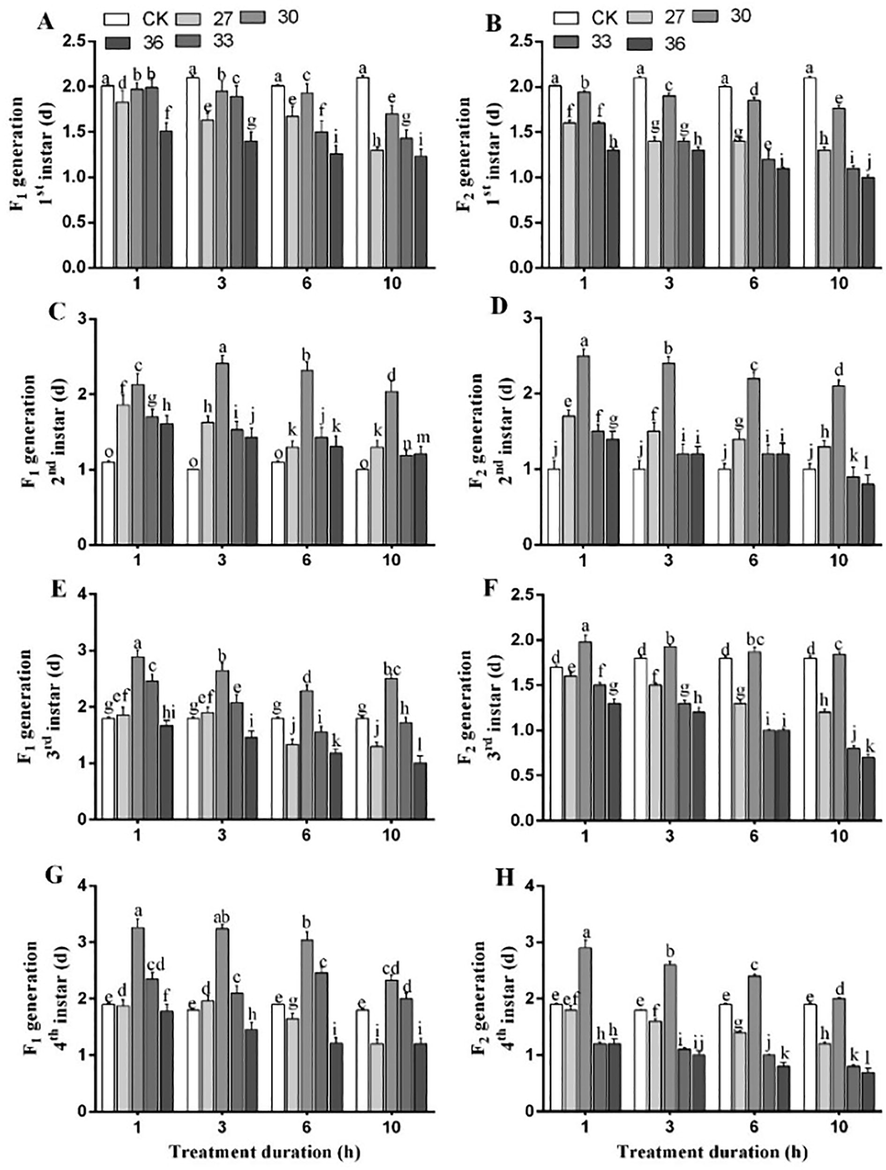
Developmental durations of 1st (A-B), 2nd (C-D), 3rd (E-F) and 4th (G-H) nymphal instars of two generations of M. persicae exposed to different temperatures for different treatment durations. Different lowercase letters indicate statistically significant differences among means at 95% probability level determined by the paired bootstrap.
3.4 Pre-adult duration
The pre-adult duration was significantly increased under 30 °C exposure for different time intervals (1, 3, 6, and 10 h). The pre-adult duration in F1 generation increased by 11.7, 11.0, 10.0, and 9.0 days, respectively, under exposure for 1, 3, 6, and 10 h compared to control (8.1 days). Similarly, the pre-adult duration in F2 generation also increased by 10.6, 10.1, 9.7, and 9.2 days, respectively as compared to the control (7.8 days). However, exposure to 36 °C for each time interval (i.e., 1, 3, 6, and 10 h) decreased pre-adult duration in F1 generation by 5.7, 4.2, 3.9, and 3.6 days respectively as compared to the control (8.1 days). In addition, the pre-adult duration of F2 generation was decreased by 5.2, 4.7, 4.2, and 3.2 days, respectively as compared to the control (7.8 days) (Fig. 4).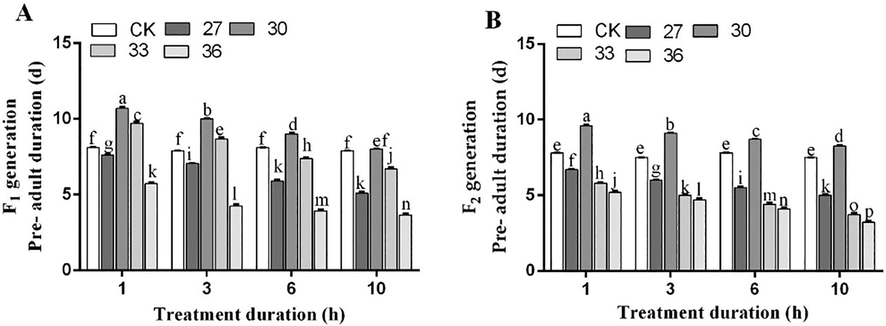
Pre-adult duration of the F1 (A) and F2 generations (B) of M. persicae under exposure to different temperatures for various treatment durations. Different lowercase letters indicate significant difference among means at 95% probability determined by the paired bootstrap.
3.5 Adult longevity and fecundity
There was no significant difference in female adult longevity and fecundity between two generations at all temperatures and time intervals. The adult longevity of both generations (F1 and F2) significantly decreased with increasing temperatures and exposure times as compared to control. The shortest longevity of the two generations was observed under exposure to 27, 30, 33, and 36 °C for 10 h (F1, 14.6, 12.5, 9.58, 5.68 days and F2, 14.4, 12.1, 9.1, 4.9 days) compared to control (F1, 19.6 and F2, 18.9 days) (Fig. 5 A, B). The fecundity of both generations under different heat stress treatments is shown in Fig. 5 C, D. The fecundity of both generations of M. persicae was significantly decreased at all temperatures and exposure intervals as compared to control. The lowest fecundity of two generations (F1, 22.3 and F2, 19.6 nymphs/female) was observed under 36 °C exposure for 10 h and the highest fecundity was recorded under exposure to 27 °C for 1 h (F1, 76.7 and F2, 76.3 nymphs/female) compared to control (F1, 78.5 and F2, 77.8 nymphs/female). The summary diagram of the effect of heat stress on survival, longevity, and fecundity is shown in Fig. 8.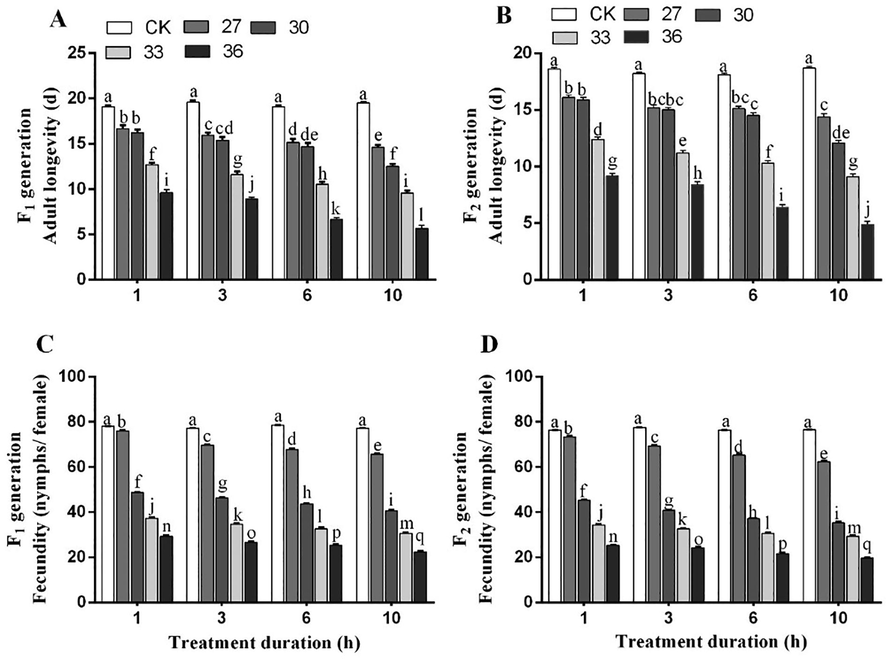
Adult longevity (A and C) and fecundity (B and D) of both generations (F1 and F2) exposed to different temperatures for various treatment durations. Different lowercase letters indicate significant differences among means at 95% probability determined by the paired bootstrap.
3.6 Population parameters
The population dynamics in both generations was studied using net reproduction rate (R0), intrinsic rate of increase (rm), finite rate of increase (λ), mean generation time (T), gross reproductive rate (GRR) and population doubling time (DT). The R0, rm, λ, T and DT were significantly altered by temperature and exposure time interaction. The highest R0 of both generations (F1, 33.26 and F2, 31.26) was observed under exposure to 27 °C for 1 h, while the lowest R0 was observed under 36 °C exposure for 10 h (F1, 8.87 and F2, 8.5). The R0 was decreased with increasing temperature (Fig. 6A, B). The rm of both generations reached the highest (F1, 0.264 and F2, 0.254) under 27 °C exposure for 1 h and the lowest (F1, 0.171 and F2, 0.161) under exposure to 36 °C for 10 h (Fig. 6C, D). The highest λ (F1, 1.41 and F2, 1.42 d−1) was noted under 27 °C exposure for 1 h. The λ in both generations dropped to 1.13 and 1.11 d−1 under exposure to 36 °C for 10 h (Fig. 6 E, F). The longest T (F1, 17.16 and F2, 16.91 d) was recorded under exposure to 27 °C for 1 h, which was two times higher (F1, 7.11 and F2, 6.9 d) when the insects were exposed to 36 °C for 10 h (Fig. 7 A, B). The DT of both generations was the longest under exposure to 27 °C for 1 h (F1, 3.94 and F2, 3.52 days), while the shortest DT (to F1, 2.01 and F2, 1.91 days) was noted for the individuals exposed to at 36 °C for 10 h (Fig. 7 C, D). The GRR of both generations significantly decreased with increasing temperature under all exposure durations. The highest values of GRR for two generations (F1, 45.02 and F2, 38.9 offspring) were noted under exposure to 27 °C for 1 h, whereas the lowest values (F1, 12.33 and F2, 11.33 offspring) were noted under exposure to 36 °C for 10 h (Fig. 7 E, F).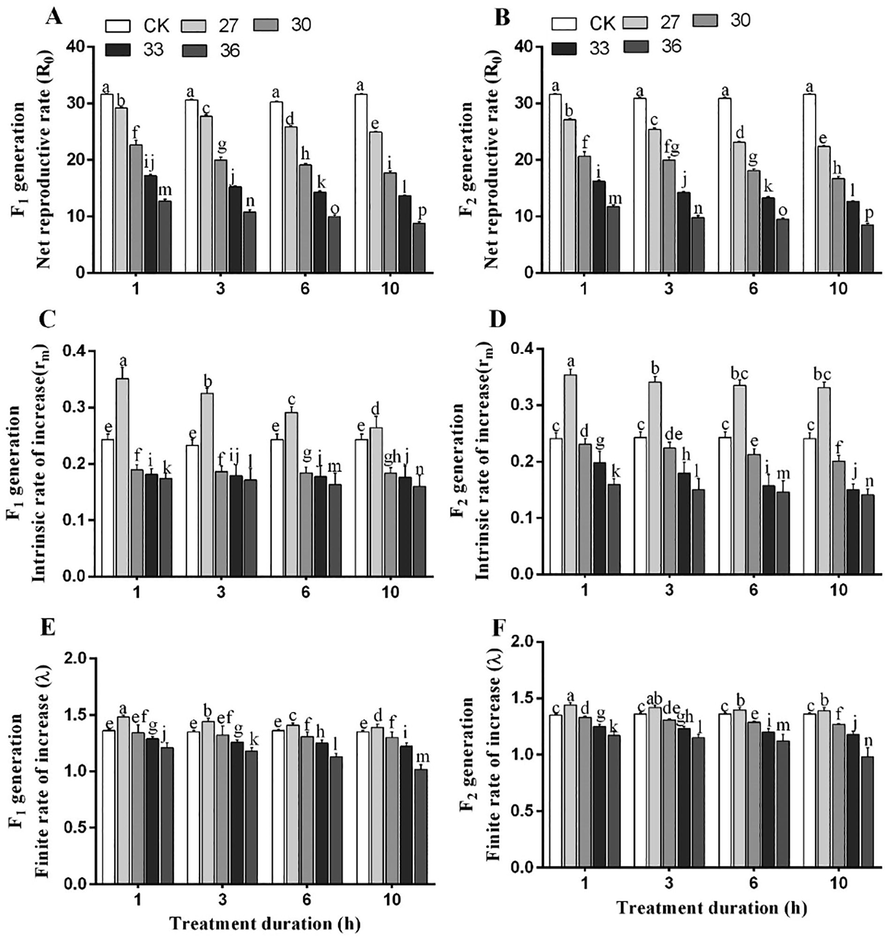
Population parameters, i.e., net reproductive rate (A, B), intrinsic rate of increase (C-D), and finite rate of increase (E-F) of two generations (F1 and F2) of M. persicae exposed to different temperatures for various treatment durations. Different lowercase letters indicate significant differences among means at 95% probability determined by the paired bootstrap.
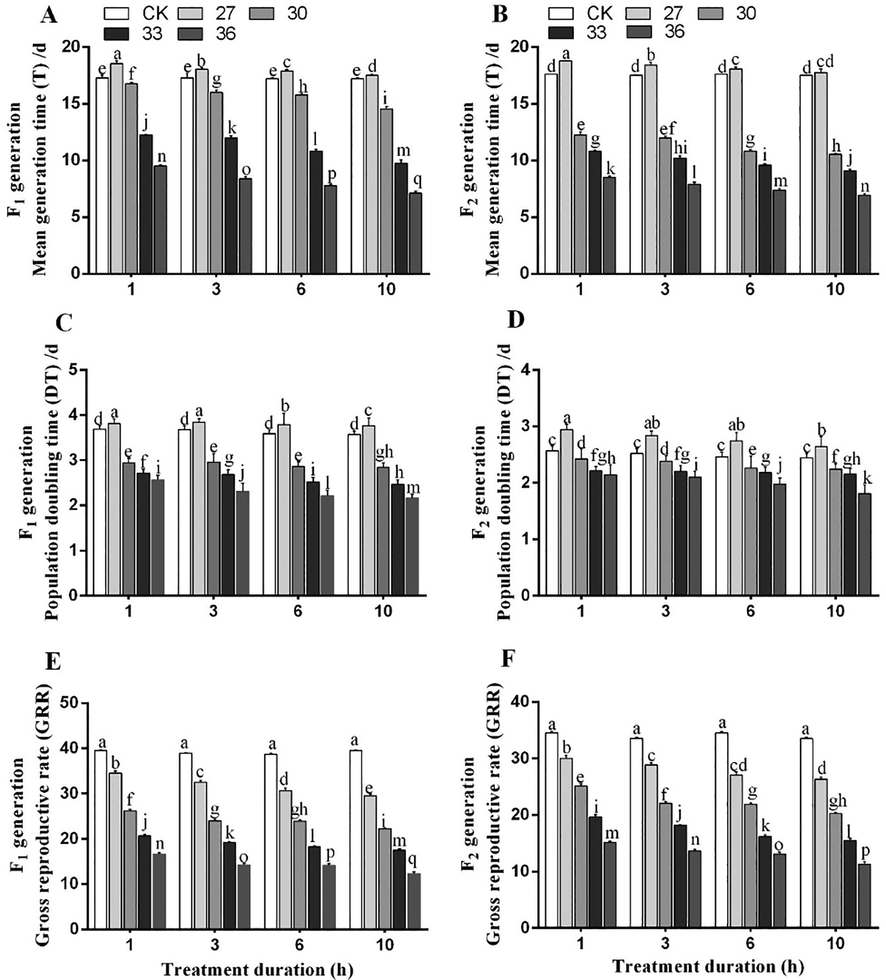
Population parameters, i.e., mean generation time (A, B), population doubling time (C, D), and gross reproductive rate (E, F) of two generations (F1 and F2) of M. persicae exposed to different temperatures for various treatment durations. Different lowercase letters indicate significant differences among means at 95% probability level determined by the paired bootstrap.
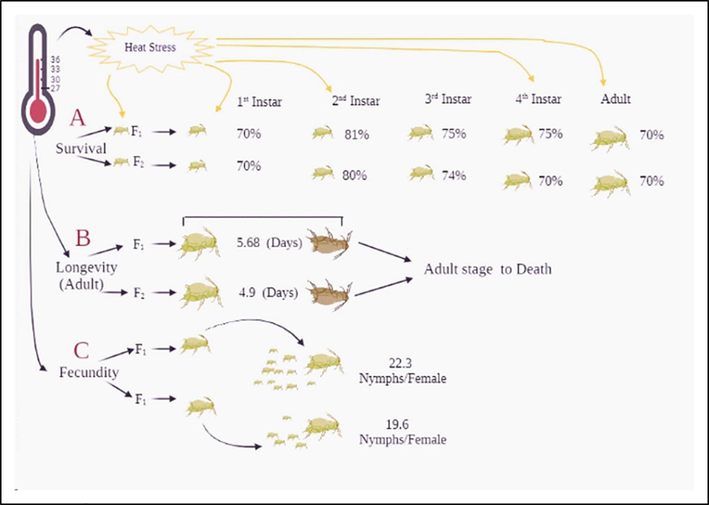
Summary diagram of biological parameters of two generations of M. persicae under exposure to different temperatures for various time intervals.
4 Discussion
The biological and life table parameters of two generations of M. persicae were examined under exposure to different temperatures for various intervals. All observed parameters were significantly influenced by different heat stress treatments in F1 and F2 generations compared to control.
Heat stress exerts significant positive/negative effects on the life history traits of insects (Clusella-Trullas et al., 2011). Aphids are heterotrophs; thus, temperature changes would have significant impact on their life history traits. Rising temperature increases digestion rate and reduces population doubling times. The longevity of adults and the oviposition can be influenced by high temperature. Temperature is one of the most significant abiotic stresses and insects being ectothermic, must regulate population growth, survival, fecundity, or periodic occurrences under temperature fluctuations (Ahn et al., 2019; Park et al., 2016). Several studies reported that determining the impact of temperature life history traits of aphids is of great significance as it is generally known they can withstand temperature changes (Ahn and Choi, 2022; Fernandes et al., 2021; Mamay et al., 2022). The impacts of climate change on aphids are expected to be complex (Crossley et al., 2021). The abundance of green peach aphids strongly depends on temperature. Crop aphids may become serious pests under rising CO2 emissions and temperatures. The impacts of environmental variables may be studied and analyzed using life tables (Stathas et al., 2011).
Our results showed that increasing temperature and exposure durations decreased the pre-adult duration. Our findings are consistent with a previous study in which heat treatment interval has a significant impact on overall lifespan (Zhao et al., 2019). Moreover, temperature increase decreased reproduction of nymphs and longevity of adults in both generations. Several insects, including other aphids have shown the same results. Nymphs and adults’ survival decreased under exposure to 36 °C for 10 h in both generations significantly. Adult aphids in both generations were successful in spawning new nymphs under 30 °C exposure for 1 to 10 h (Ahn et al., 2020).
Temperature has a considerable impact on the population parameters of green peach aphids. The highest R0 in both generations was observed under 27 °C exposure for 1 h, while the smallest R0 was noted for the individuals exposed to 36 °C for 10 h. Barlow, (1962) reported that population of M. persicae and M. euphorbiae decreased with increased temperature. The intrinsic rate of increase and the finite rate of increase of the two generations in current study reached the highest values under 27 °C exposure for 1 h and the lowest under 36 °C exposure for 10 h. According to our findings, the intrinsic rate and finite rate of increase in both generations decreased at 36 °C exposure (1 to 10 h) compared to the control. Like our findings, Barlow (1962) reported that temperatures > 20 °C caused early instar mortality of M. persicae more than that of adult stages, resulting in a reduction in the finite rate of increase, although the developmental time was decreased. When insects are exposed to different temperatures, they react in several ways. Insect behavior, physiological or morphological variation might be the response to these different temperatures (Khadioli et al., 2014). The rm represents the impact of the age of initial reproduction, the timing of the peak of reproduction, the duration of the reproductive period, and the survival rate on the population growth rate under various environmental conditions and is critical in determining the potential for insect population increase under various environmental conditions. Andrewartha and Birch (1984) described physiological characteristics of herbivores about increasing capacity, as summarized by rm. In current study, rm of two generations decreased as the temperature was raised from 27 to 36 °C for all exposure durations (1 to 10 h) compared to control. Similarly, finite rate of increase of two generations increased at 27 °C (1 to 10 h); however, sharply decreased at 36 °C (1 to 10 h) compared to control. Zhou et al. (2014) found rm of M. persicae and M. euphorbiae reduced with the increasing temperature. Similarly, the population of Acyrthosiphon pisum demonstrated that when temperature increased from 27 to 39 °C, the intrinsic and finite rates of increase dropped (Mastoi et al., 2020). Previous research has shown that the higher temperatures are expected to have stage-specific impacts on insects. In insects with very basic life cycles, such as True bugs and Scale insects (Zhao et al., 2017), the impacts of high temperatures can also differ throughout generations.
The impact of climate conditions on aphids can be determined using changes in life-history parameters and various techniques of rearing could have changed the development. Zhao et al. (2019) reported that the stress exposure at one stage has fitness consequences later. Implying that fitness component selection at the adult stage may be influenced by heat exposures at a previous stage in an insect's life cycle. This shows that difficulty in physiological reactions must be taken into consideration when evaluating the effects of heat stress on populations. Computer programs, such as life table analysis, have been designed to examine a lot of information from ecological and biological research (Akca et al., 2015).
In our study, the duration of the heat stress application has generated an interesting knowledge on the control operation where the high temperature exposure, i.e., 36 °C for 10 h can affect longevity, fecundity, rm, R0, and finite rate of increase of M. persicae. We found that an increase in the length of stress frequently increases the negative effects on M. persicae. The findings in our research showed the life history data and population factors of M. persicae were affected by the temperature. Biological properties of two generations of M. persicae in response to different temperatures and exposure durations provide a basis for their monitoring and management.
5 Conclusions
All life stages and population parameters of the two generations of M. persicae were significantly affected by exposure to 36 °C for 10 h. The population parameters and biological parameters based on life tables might be useful for estimating population changes in the field for developing appropriate management methods for M. persicae. Increased temperature and exposure duration in greenhouse and house crops, might provide a method for integrated pest management and improve scientific information for managing the population dynamics of M. persicae. More research is needed to understand the population dynamics of M. persicae, as well as their behavioral responses to various heat stresses.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (No. 31960351). This work was supported by the King Khalid University through a grant KKU/RCAMS/22 under the Research Center for Advance Materials (RCAMS) at King Khalid University, Saudi Arabia.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Biological and life table parameters of Myzus persicae (Salz.)(Hemiptera: Aphididae) in relation to host plants and thermal requirements. J. Plant Prot. Pathol.. 2009;34(7):8251-8262.
- [Google Scholar]
- Thermal effects on the population parameters and growth of Acyrthosiphon pisum (Harris)(Hemiptera: Aphididae) Insects. 2020;11:481.
- [Google Scholar]
- Population parameters and growth of Riptortus pedestris (Fabricius) (Hemiptera: Alydidae) under fluctuating temperature. Insects. 2022;13(2):113.
- [Google Scholar]
- Effects of temperature on the development, fecundity, and life table parameters of Riptortus pedestris (Hemiptera: Alydidae) Appl. Entomol. Zool.. 2019;54(1):63-74.
- [Google Scholar]
- Demography and population projection of Aphis fabae (Hemiptera: Aphididae): with additional comments on life table research criteria. J. Econ. Entomol.. 2015;108(4):1466-1478.
- [Google Scholar]
- Andrewartha, H.G., Birch, L.C., 1984. The ecological web: more on the distribution and abundance of animals. Ecol. web more Distrib. abundance Anim.
- Development, survival, and fecundity of the potato aphid, Macrosiphum euphorbiae (Thomas), at constant temperatures. Can. Entomol.. 1962;94:667-671.
- [CrossRef] [Google Scholar]
- Divergence and ontogenetic coupling of larval behaviour and thermal reaction norms in three closely related butterflies. Proc. R. Soc. B Biol. Sci.. 2011;278(1703):313-320.
- [Google Scholar]
- Aphids on the World’s Crops: An Identification and Information Guide. John Wiley & Sons Ltd.; 2000.
- Life-table analysis incorporating both sexes and variable development rates among individuals. Environ. Entomol.. 1988;17:26-34.
- [Google Scholar]
- Chi, H., 2015. TWOSEX-MSChart: a computer program for the age-stage, two-sex life table analysis. Available on: http://140.120 197.
- Two new methods for the study of insect population ecology. Bull. Inst. Zool. Acad. Sin. 1985;24:225-240.
- [Google Scholar]
- Climatic predictors of temperature performance curve parameters in ectotherms imply complex responses to climate change. Am. Nat.. 2011;177(6):738-751.
- [Google Scholar]
- Complex life histories predispose aphids to recent abundance declines. Glob. Chang. Biol.. 2021;27(18):4283-4293.
- [Google Scholar]
- Effects of high and fluctuating temperatures on Myzus persicae (Hemiptera: Aphididae) Environ. Entomol.. 2006;35:1461-1468.
- [Google Scholar]
- Sub-lethal effects of a Bt-based bioinsecticide on the biological conditioning of Anticarsia gemmatalis. Ecotoxicology. 2021;30:2071-2082.
- [CrossRef] [Google Scholar]
- Ontogenetic stage-dependent effect of temperature on developmental and metabolic rates in a holometabolous insect. J. Insect Physiol.. 2010;56(11):1679-1684.
- [Google Scholar]
- Higher trends but larger uncertainty and geographic variability in 21st century temperature and heat waves. Proc. Natl. Acad. Sci.. 2009;106(37):15555-15559.
- [Google Scholar]
- Hodek, I., van Emden, H.F., Honěk, A., 2012. Ecology and Behaviour of the Ladybird Beetles (Coccinellidae), Ecology and Behaviour of the Ladybird Beetles (Coccinellidae). John Wiley & Sons, Ltd, Chichester, UK. doi: 10.1002/9781118223208.
- Age-stage, two-sex life tables of Bactrocera cucurbitae (Coquillett)(Diptera: Tephritidae) with a discussion on the problem of applying female age-specific life tables to insect populations. Insect Sci.. 2012;19:263-273.
- [Google Scholar]
- Temperature-dependent development of Oenopia conglobata (Col.: Coccinellidae) fed on Aphis gossypii (Hem.: Aphididae) Int. J. Trop. Insect Sci.. 2018;38:410-417.
- [CrossRef] [Google Scholar]
- Effect of temperature on the life history parameters of noctuid lepidopteran stem borers, Busseola fusca and Sesamia calamistis. Ann. Appl. Biol.. 2014;165(3):373-386.
- [Google Scholar]
- Khurshid, A., Inayat, R., Tamkeen, A., Haq, I.U., Li, C., Boamah, S., Zhou, J.-J., Liu, C., 2021. Antioxidant Enzymes and Heat-Shock Protein Genes of Green Peach Aphid (Myzus persicae) Under Short-Time Heat Stress. Front. Physiol. 12.
- Heat resistance throughout ontogeny: body size constrains thermal tolerance. Glob. Chang. Biol.. 2017;23(2):686-696.
- [Google Scholar]
- Climate warming may increase aphids’ dropping probabilities in response to high temperatures. J. Insect Physiol.. 2012;58(11):1456-1462.
- [Google Scholar]
- Acclimation responses to short-term temperature treatments during early life stages causes long lasting changes in spontaneous activity of adult Drosophila melanogaster. Physiol. Entomol.. 2017;42(4):404-411.
- [Google Scholar]
- Effect of maternal age on population parameters of Anthocoris minki Dohrn (Hemiptera: Anthocoridae) reared on Ephestia kuehniella Zeller (Lepidoptera: Pyralidae) Phytoparasitica 2022
- [CrossRef] [Google Scholar]
- Effect of heat stress on life history of pea aphid, Acyrthosiphon pisum (Harris)(Hemiptera: Aphididae) based on life-table. Pak. J. Agri. Sci. 2020;57:325-332.
- [Google Scholar]
- Temperature-dependent development and fecundity of Rhopalosiphum padi (L.)(Hemiptera: Aphididae) on corns. Korean J. Appl. Entomol.. 2016;55:149-160.
- [Google Scholar]
- Warming tolerance across insect ontogeny: influence of joint shifts in microclimates and thermal limits. Ecology. 2015;96(4):986-997.
- [Google Scholar]
- Thermal requirements and effect of temperature and prey on the development of the predator Harmonia axyridis. Environ. Entomol.. 2011;40(6):1541-1545.
- [Google Scholar]
- Demography of Cacopsylla chinensis (Hemiptera: Psyllidae) reared on four cultivars of Pyrus bretschneideri (Rosales: Rosaceae) and P. communis pears with estimations of confidence intervals of specific life table statistics. J. Econ. Entomol.. 2020;113(5):2343-2353.
- [Google Scholar]
- Temperature-body size responses in insects: a case study of British Odonata. Ecol. Entomol.. 2020;45(4):795-805.
- [Google Scholar]
- Life stages of an aphid living under similar thermal conditions differ in thermal performance. J. Insect Physiol.. 2017;99:1-7.
- [CrossRef] [Google Scholar]
- The importance of timing of heat events for predicting the dynamics of aphid pest populations. Pest Manag. Sci.. 2019;75:1866-1874.
- [CrossRef] [Google Scholar]
- Effects of temperature on life table parameters of the laboratory populations of Myzus persicae and Macrosiphum euphorbiae (Hemiptera: Aphididae) Acta Entomol. Sin.. 2014;57:837-843.
- [Google Scholar]







