Translate this page into:
Effect of host preference and micro habitats on the survival of Tetranychus urticae Koch (Acari: Tetranychidae) in Saudi Arabia
⁎Corresponding author. Kholod.Alshammery@gmail.com (Kholoud A. Al-Shammery)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Objective
Control of pests is essential to save the loss of economically important plants. Biological and environmental control became the most important interventions of choice. The two-spotted spider mite Tetranychus urticae is a herbivorous mite which is considered to be a harmful pest. The objective of the study was to investigate the effect of host preference on the survival and distribution of Tetranychus urticae.
Methods
Specimens of T. urtica were collected from Solanum melongena plants grown at Al Qaseem University. Eggs were transferred individually to the bottom surface of leaves of certain plants namely: Phasoelus vulgaris (beans), Solanum melongena (eggplant), Fragaria ananasa (Strawberry), Citrus sinensis (Orange), and Cucumis sativus (Cucumber) and reared on those plants under optimum conditions. The effect of climatic conditions and the type of the plant host on the survival of mature and immature stages of T. urtica has been observed.
Results
The total growth period length of the immature stage of the male mites was found to be shorter than the growth period of the female immature stages. The peak of this period was recorded in Citrus sinensis followed by Cucumis sativus then Solanum melongena, Phasoelus vulgaris, and Fragaria ananasa.
Conclusion
These results may lead to the screening process of comparatively resistant herbal species to be used in the management of T. urticae.
Keywords
Tetranychus urticae
Phasoelus vulgaris
Solanum melongena
Fragaria ananasa
Citrus sinensis
Cucumis sativus
Survival
1 Introduction
Tetranychus urticae, the two-spotted spider mites, are cosmopolitan pests of a wide range of crops such as cucumber, strawberry, orange, eggplant, and beans (Eniko Gyuris et al., 2018). They are some of the most threatening economic pests and often require selective control interventions which might not be easy due to the rising resistance to the most recommended insecticides. These mites often inhabit the below side of leaves, where they feed and induce straps that coat specific parts of the leaf, this phenomenon decreases the efficacy of insecticidal control (Numa et al., 2015; Sarwar and Sattar, 2013).
The extent of deterioration of leaves caused by T. urticae is obvious in the ply and length of the leaves (Bianchi et al., 2006; Estrella et al., 2020). The diet of T. urticae is usually chloroplasts of cells especially the bottom of the leaf, whereas the upper surface of the leaf develops creamy-colored punctuations, which may alter to darker brown color upon the feeding of mites. Park and Lee (2002) studied the impact of damage due to the attack of T. urticae on Cucumis sativus and reported that on the abaxial leaf surface, adult T. urticae damaged the spongy parenchyma as well as part of the palisade parenchyma, whereas immature T. urticae fed only the spongy parenchyma. Infestation with T. urticae will significantly affect the net photosynthetic rate, chlorophyll content, and thereby the greenness of the leaves. Besides the loss in the number of leaves their ability to alter photosynthetic rate after the mite infestation was estimated to be a hundred each minute (Silvere et al., 2017; Hajar and Annie, 2012).
The fast rate of development, high fecundity, and arrhenotoky in T. urticae let them quickly reach a harmful population level under favorable conditions that subsequently lead to a rapid decline in host plant quality. (Kanika Tehri, 2014). There are some good reports on the within-plant distribution of T. urticae on different plant hosts (Tollerup et al., 2013; Choi et al., 2022). Choi et al (2022) have reported that on strawberry plants, optimal sampling units of T. urticae were the fifth, fifth, or sixth, and sixth oldest leaves for adult and immature motiles as well as eggs.
Because of difficulties associated with their control and huge economic losses, there is much interest in the search for alternative control measures, especially biological control. Hence, this study aimed to investigate the effect of host preference of Tetranychus urticae on their survival and distribution.
2 Materials and methods
2.1 Samples collection and rearing
Adults and eggs of T. urtica were collected from Solanum melongena plants grown in greenhouses in the research center of Faculty of Agriculture and Veterinary Sciences, Al Qaseem University. These insects were reared on bean plants under laboratory conditions (26 °C, 70% Relative humidity (RH) for three weeks.
Thirty eggs were separated using a zero-size brush under a binocular microscope (X100 lens magnification). Eggs were transferred individually to the bottom surface of leaves (1 inch2 area) of the following plants:
-
Phaseolus vulgaris (Beans)
-
Solanum melongena (Eggplant)
-
Fragaria ananasa (Strawberry)
-
Citrus sinensis (Orange)
-
Cucumis sativus (Cucumber)
Replicates were placed on wet cotton pieces on petri dishes (10 cm diameter). Drops of water were added to the dishes daily to keep the cotton moist. Plant leaves were changed weekly for transferring the hatching mite individuals gently using brushes.
2.2 Recording time tables data
The number of hatched eggs was recorded and the hatching proportion was calculated. The incubation period until hatching, larval stage, and pupal stages (including protonymphal and deutonymphalstages) were also registered once a day.
2.3 Sex ratio estimation
After emergence from the pupal stages number of adult males and females stage was recorded to calculate the sex ratio using the following formula:
The obtained parentage was analyzed using the life table parameter to forecast the females ratio in the next generation. (Charly et al., 2020; Sayed et al., 2012).
2.4 Adult's life span
The life span of adult males was estimated. Each newly emerged female was located with a male (obtained from the mite colony) for her entire life then the pri-oviposition, the oviposition, and the post- oviposition periods were estimated. Methods of (Birch, 1948; Andrewartha and Birch, 1954; Abou-Setta et al., 1986; Ismail, 2004) were adopted for calculating the longevity of mites as well as estimating the total number of laid eggs and the daily oviposition rate in order to calculate the life table parameters using the following formula:
Where:
L = Number of viable females per day.
X = The actual age of the female (/day).
Mx = Number of females per generation or special fertility rate per day (x).
Lx = Survival rate per day (x).
R0 = Replication rate per generation duration (∑LxMx).
T = Average generation duration.
Rm = Average normal growth.
Erm = Average daily replication.
3 Results
3.1 Effect of type of plant hosts on the longevity of eggs of T. urticae
Results exhibited that variation of the plants' surfaces on which T. urticae were reared had no significant impact on the incubation period of the eggs hatching which ranged between 3.2 and 4.1 days for males while it was 3.8–4.9 days for females (Fig. 1). On the other hand, the type of the surface of plants hosts, the leaves' texture, bristles, and glands on the leaves have an impact on both mature and immature stages of T. urticae (Figs. 2 and 3).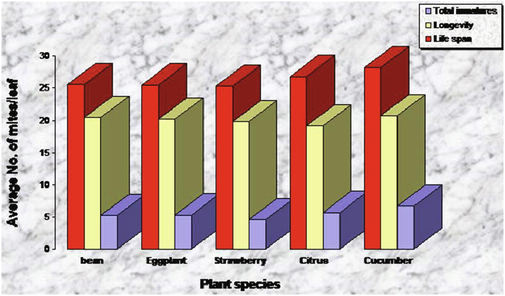
Effect of different textures on duration of developmental stages, (longevity) of Tetranychus urticae adult females.
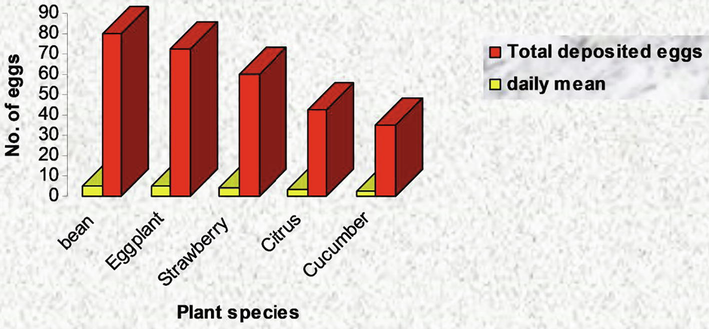
Effect of different textures on total and daily mean of deposited eggs of T. urticae adult females.
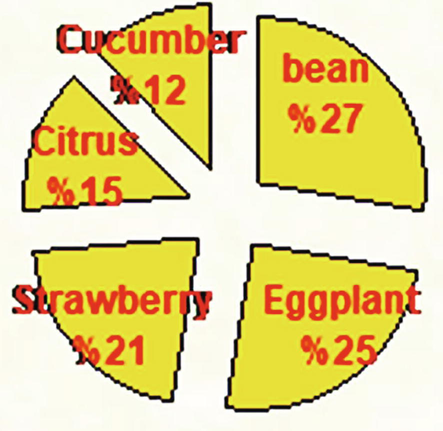
Proportion of total deposited eggs of T. urticae on different plant leaves (%).
3.2 Effect of type of plant hosts on the longevity of larvae of T. urticae
Coinciding with the impact on the egg hatching, the type of plant's surface did not affect the longevity of the larval stage which was 2.7–2.8 days when the mites reared on the leaves of Citrus sinensis and Cucumis sativus. In contrast to this, the longevity of the larval stage diminished significantly when female larvae reared on Solanum melongena, Fragaria ananasa, and Phasoelus vulgaris respectively (Figs. 4–7; Table 1). Contrary to this, larval periods of the male larvae did not affect significantly when reared on the three plants (Table 1). For the female protonymphs results showed that leaves of Citrus sinensis insignificantly affected the growth period of these females when reared on them while the effect was significant when they reared on Solanum melongena, Phasoelus vulgaris, and Fragaria ananasa (Table 1). Data were subjected to the Micro-Computer Program COSTAT, one way ANOVA using Student-Newman Keuls Test, where means with the same letter aren't significantly different (P < 0.05).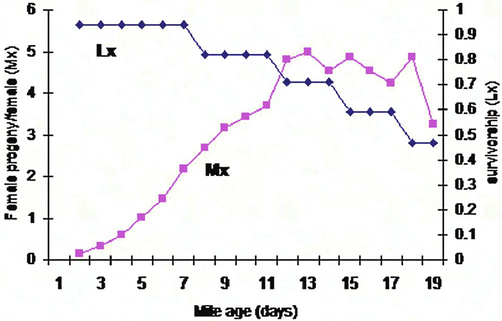
Development duration of T. urticae reared on bean leaves.
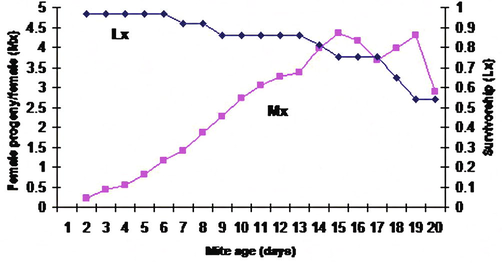
Development duration of T. urticae reared on eggplant leaves.
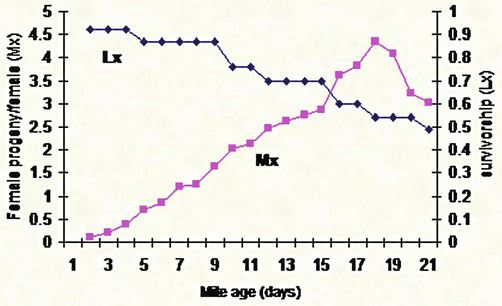
Development duration of T. urticae reared on strawberry leaves.
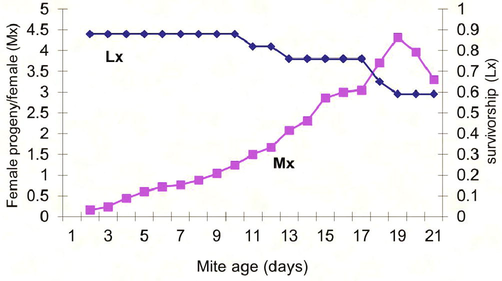
Development duration of T. urticae reared on Citrus leaves.
Plant species
No. Sex
Incubation period
Developmental stages
Total
Sex ratio %
Larva
Protonymph
Deutonymph
♀
♂
♀
♂
♀
♂
♀
♂
♀
♂
♀
♂
Bean
16
14
3.8 a ± 0.06
3.2 ab ± 0.03
1.8b ± 0.02
1.6 a ± 0.02
1.5b ± 0.04
1.3b ± 0.04
1.9b ± 0.07
1.8b ± 0.07
5.2b ± 1.04
4.7b ± 1.02
74
Eggplant
15
13
3.6 a ± 0.07
3.3 ab ± 0.04
1.9b ± 0.07
1.5 a ± 0.06
1.6b ± 0.03
1.4b ± 0.06
1.8b ± 0.06
1.6b ± 0.04
5.3b ± 1.05
4.5b ± 1.03
72
Strawberry
14
14
4.2 a ± 0.09
3.6 a ± 0.03
1.6b ± 0.04
1.6 a ± 0.06
1.6b ± 0.04
1.3b ± 0.06
1.8b ± 0.06
1.7b ± 0.06
5.0b ± 1.02
4.6 a ± 1.16
69
Citrus
14
13
4.5 a ± 0.04
3.8 a 0.06
2.7 a ± 0.04
1.6 a ± 0.03
2.3 a ± 0.06
1.8 ab ± 0.03
2.6 a ± 0.10
2.3 a ± 0.09
7.6 a ± 1.02
5.7b ± 1.46
72
Cucumber
15
14
4.9 a ± 0.06
4.1 a ± 0.06
2.8 a ± 0.04
1.9 a ± 0.07
2.4 a ± 0.04
2.2 a ± 0.07
2.8 a ± 0.09
2.6 a ± 0.07
8.0 a ± 1.40
6.7 a 1.32
71
L.S.D.(P < 0.05)
0.82
0.67
0.06
0.12
0.46
0.69
0.42
0.56
0.98
0.86
–
3.3 Effect of type of plant hosts on the longevity of pupae of T. urticae
Results showed an insignificant difference between the growth period of the second pupal stage (deutonymph) compared to the growth period of the first pupal stage (protonymph) when reared on the same plant. In contrast to this, the growth period of deutonymph significantly extended to 2.6–2.8 days when grown on Citrus sinensis and Cucumis sativus respectively while the growth period was 1.8, 1.9, and 1.8 days when the pupae grown on Solanum melongena, Phasoelus vulgaris, and Fragaria ananasa plants respectively (Table 1).
3.4 Effect of type of plant hosts on the longevity of both mature and immature stages of T. urticae
Results showed insignificant effect of the type and texture of Citrus sinensis and Cucumis sativus on the growth of the immature stages of T. urticae (8 and 7.6 days respectively) while these two factors had a significant effect on the growth of the immature stages of these mites when grown on Solanum melongena, Phasoelus vulgaris, and Fragaria ananasa (5.2, 5.3, and 5 days respectively) (Fig. 8, Table 2). Data were subjected to the Micro-Computer Program COSTAT, one-way ANOVA using Student-Newman Keuls Test, where means with the same letter aren't significantly different (P < 0.05).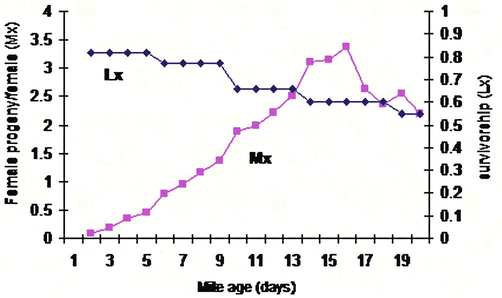
Development duration of T. urticae reared on cucumber leaves (A, B, C, D, E): Life table parameters (Mx, Lx) of Tetranychus urticae reared on five different plant leaves in laboratory.
Plant species
Adults
Longevity
Life span
Preoviposition
Oviposition
Postoviposition
♀
♂
♀
♂
♀
♂
♀
♂
♀
♂
Bean
1.3b ± 0.64
–
16.4 a ± 1.86
–
2.7b ± 0.62
–
20.4 a ± 1.67
17.6 a ± 1.28
25.6c ± 1.48
22.3 ab ± 1.86
Eggplant
1.4 b ± 0.82
–
15.7 a ± 1.72
–
3.1 b ± 0.74
–
20.2 ab ± 1.48
16.8 a ± 1.42
25.2 c ± 1.62
21.3 b ± 1.46
Strawberry
1.6 b ± 0.74
–
14.3b ± 1.36
–
3.9 ab ± 0.46
–
19.8 ab ± 1.69
17.3 a ± 1.23
24.8 c ± 1.41
21.9 b ± 1.37
Citrus
2.2 ab ± 0.68
–
12.7 c ± 1.42
–
4.2 a ± 0.81
–
19.1b ± 1.56
15.4 a ± 1.08
26.77 b ± 1.21
21.1 b ± 1.28
Cucumber
2.8 a ± 0.87
–
13.2 bc ± 1.26
–
4.7 a ± 0.92
–
20.7 a ± 1.86
16.5 a ± 1.13
28.7 a ± 2.16
23.2 a ± 1.84
L.S.D.
(P < 0.05)0.48
–
1.02
–
0.62
–
1.04
1.08
1.42
1.16
Results also showed that the total growth period length of the immature stage of the male mites was shorter than the growth period of the female immature stages. The peak of this period was recorded in Citrus sinensis followed by Cucumis sativus then Solanum melongena, Phasoelus vulgaris, and Fragaria ananasa (Table 1, Fig. 8).
3.5 Effect of type of plant on life table parameters of Tetranychus urticae reared on different plant leaf surfaces and kept at 26 °C and 70% RH
Results showed that mites have net reproductive rates (R0) of 38.07, 37.55, 28.45, and 27.28 times during generation periods (T) 14.48, 15.75, 17.18, and 18.16 days when reared on eggplant followed by Phasoelus vulgaris, Fragaria ananasa, and Citrus sinensis respectively while rearing on Cucumis sativus induced the least duplication rate where R0 was 21 time during a life span of 18.63 days (Table 3).
Plant surface
Mean generation time T (days)
Net reproductive rate (Ro)
Intrinsic rate of increase (rm)
Finite rate of increase (erm)
Bean
14.480
38.072
0.251
1.285
Eggplant
15.757
37.558
0.230
1.258
Strawberry
17.181
28.452
0.194
1.215
Citrus
18.616
27.280
0.173
1.102
Cucumber
18.632
21.211
0.167
1.103
The intrinsic rate of natural increase (rm) was 0.25, 0.19, 0.17, 0.16 individual (female)/ day when reared on Fragaria ananasa, Phasoelus vulgaris, and Citrus sinensis respectively.
4 Discussion
Determination of factors affecting the survival of T. urticae is crucial to deciding the best biological and environmental control of this pest. Besides, discovering the exact microhabitats that T. urticae prefer to inhabit, it is useful to exact certain parts of plants with insecticides instead of spraying the whole vegetation when chemical control is found to be the intervention of choice (Wekesa et al., 2011).
Results of this study revealed that the type and texture of plants have variance effects on the growth of mature (both sexes) and immature stages of T. urticae where the longest growth period of the spider was recorded when reared on Cucumis sativus hosts. This may be due to the rough texture of Cucumis sativus leaves which may retard the movement and activity of the immature stages. Diminished activity may therefore affect the ability of these stages to absorb the herbal succulents. An inadequate amount of carbohydrates will affect the viable activities of the mites such as molting. This may be the cause of the prolonged growth periods (Lewis, 1995).
These findings coincide with the results of another study which recorded that the total immature stages and life cycle were shortest when spider mite was fed on mulberry leaves. The greatest values of reproduction rate and hatchability were on mulberry leaves than on other crops and followed by castor bean (Nizam Uddin et al., 2015). The developmental and reproduction rate of T. urticae is influenced by tissue nutrient content for the host plant whereas Elsadany (2018) also reported that the sex ratio of T. urticae was highest on castor bean leaves. The results also showed that larval periods of the male larvae did not affect significantly when reared on the three plants while for the female protonymphs, results showed that leaves of Citrus sinensis insignificantly affected the growth period of these females when reared on them. In contrast to this, the effect was significant when they reared on Solanum melongena, Phasoelus vulgaris, and Fragaria ananasa.
Results also showed that mites achieved the peak of net reproductive rate (R0) when reared on an eggplant while rearing on Cucumis sativus induced the least duplication rate. The mean qualitative increase (rm) was the most when the mites reared on Fragaria ananasa. Coinciding with this, a previous study indicated that the choice of host plant species will affect how fast spider mite populations reach damaging levels in a culture where the slowest population growth was observed on the bean species with rm = 0.214 (Jabraeil et al., 2009). Depending on these findings, it is recommended to grow plants that are frequently infested by parasitic mites around crops as an herb- prophylaxis to prevent them from being attacked.
Finally, it can be inferred that host plants play a considerable role in the susceptibility of T. urticae. This is agreed with the findings of another study which recorded that spider mites are susceptible to acaricides and abamectin could be used as a puissant chemical for controlling spider mites (Tarikul, 2018).
5 Conclusion
The present work discussed the feeding preference of pests as a way to evaluate their population ecology. Tetranychus urticae is an agriculture pest mite with limited knowledge about its ecology and feeding preference. Such information is very important to its control and hence the effect of the feeding environment on its population was evaluated. The type of food shows a great effect on the number of laid eggs and their quality. Beans appear as the best host for the number of egg-laying. On the other hand, there was no clear significant impact of food type on longevity, although the rate of development shows great variation. Male larval stage developed faster than female, especially on citrus. More work is needed to get a complete understanding of the ecology of Tetranychus urticae and the present work forms a small step in this direction.
Acknowledgement
The authors acknowledge Princess Nourah bint Abdulrahman University Researchers supporting Project number (PNURSP2022R37), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- A computer program in basic for determining probit and log-probit or logit correlation for toxicology and biology. Bull. Environ. Contam. Toxicol.. 1986;36:242-249.
- [CrossRef] [Google Scholar]
- Andrewartha, H. G. and Birch, L. C. The distribution and abundance of animals. 1954 No.Edn 1 pp.xv + 782 pp. University of Chicago Press.
- Sustainable pest regulation in agricultural landscapes: a review on landscape composition, biodiversity and natural pest control. Proc. R. Soc. B. 2006;273(1595):1715-1727.
- [Google Scholar]
- The Intrinsic rate of natural increase of an insect population. J. Anim. Ecol.. 1948;17(1):15-26.
- [CrossRef] [Google Scholar]
- Sex-specific patterns of senescence in artificial insect populations varying in sex-ratio to manipulate reproductive effort. BMC Evol. Biol.. 2020;20(1)
- [Google Scholar]
- Within-Plant Distribution of Two-Spotted Spider Mites, Tetranychus urticae Koch (Acari: Tetranychidae), on Strawberries: Decision of an Optimal Sampling Unit. Insects. 2022;13(55)
- [CrossRef] [Google Scholar]
- Influence of host plants and some leaf contents on biological aspects of Tetranychus urticae Koch (Arachnida:Acari: Tetranychidae) JoBAZ. 2018;79(20):2-8.
- [CrossRef] [Google Scholar]
- Eniko Gyuris, E Szép, J Kontschán, A Hettyey. Efficiency against the Two-spotted spider mite Tetranychus urticae and prey age-related choice of three predatory mites. Acta Zoologica Acta Zoologica Academiae Scientiarum Hungaricae 2018. 64.1: 75-90. https://doi.org/10.31610/zsr 2018.
- Plant defenses against Tetranychus urticae: mind the gaps. Plants. 2020;9(4):464-480.
- [CrossRef] [Google Scholar]
- Effect of different temperatures on consumption of two spotted mite, Tetranychus urticae, eggs by the predatory Thrips, Scolothrips longicornis. J. Insect Sci.. 2012;12(98):98.
- [CrossRef] [Google Scholar]
- Effect of apple cultivar and of temperature on the biology and life table parameters of the two spotted spider mite Tetranych usurticae. Phytoparasitica. 2004;32(1):73-82.
- [CrossRef] [Google Scholar]
- Life history traits of T. urticae Koch on three legumes (Acari: tetranychidae) Mun. Ent. Zool. 2009;(4)1:204-211.
- [Google Scholar]
- Habitats of two-spotted spider mites (Acari: Tetranychidae) during winter and spring in a cotton-producing region of Australia. Environ. Entomol.. 1995;24(2):332-340.
- [CrossRef] [Google Scholar]
- Life table parameters of Tetranychus urticae Koch (Acari: Tetranychidae) on different bean varieties. Afr. Entomol.. 2015;23(2):418-426.
- [Google Scholar]
- Susceptibility of Tetranychus urticae Koch to an ethanol extract of Cnidoscolus aconitifolius leaves under laboratory conditions. Springerplus.. 2015;4:338-348.
- [CrossRef] [Google Scholar]
- Leaf cell and tissue damage of cucumber caused by two-spotted spider mite (Acari: Tetranychidae) J Econ. Entomol. 2002 200;95(5):952–7
- [Google Scholar]
- Varietals variability of winter rapes (Brassica napus L.) for their susceptibility to green aphid, Myzus persicae (Sulzer) (Homoptera: Aphididae). Pakistan. J. Zool.. 2013;45(4):883-888.
- [Google Scholar]
- When to estimate sex ratio in natural populations of insects? A study on sex ratio variations of gall midges within a generation. Entomol. Sci.. 2013;16(1):54-59.
- [CrossRef] [Google Scholar]
- Importance of fluctuations in light on plant photosynthetic acclimation. Plant Physiol.. 2017;173(4):2163-2179.
- [CrossRef] [Google Scholar]
- Host plant-induced susceptibility of two-spotted spider mite Tetranychus urticae (Acari: Tetranychidae) to some reduced-risk acaricides. Am. J. Agric. Biol. Sci.. 2018;14(1):11-15.
- [CrossRef] [Google Scholar]
- Binomial and enumerative sampling of Tetranychus urticae (Acari: Tetranychidae) on peppermint in California. J. Econ. Entomol.. 2013;106:1707-1715.
- [Google Scholar]
- The effect of host plants on Tetranychus evansi, Tetranychus urticae (Acari: Tetranychidae) and on their fungal pathogen Neozygites floridana (Entomophthorales: Neozygitaceae) J. Invertebr. Pathol.. 2011;107(2):139-145.
- [CrossRef] [Google Scholar]







