Translate this page into:
Biological activities of novel 2-pyrazolin-5-one derivatives and their toxicity on certain pests infesting field crops in laboratory conditions
⁎Corresponding author. ahmed.abeed76@aun.edu.eg (Ahmed Abdou O. Abeed)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
A variety of highly potent derivatives of 2-pyrazoline-5-one, including chromene-3-carbohydrazide 2, 2-thioxothiazole-5-carbohydrazide 3, 2-oxopyridine-3-carbonitriles (4–6), 3-aryl-2-cyanoacrylohydrazide (7, 8), and 3-amino-5-arylpyrazole-4-carbohydrazide (9, 10), were synthesized as pesticide agents using acetohydrazide 1, a versatile and easily obtainable compound. The compounds structures were entirely ascertained utilizing different spectroscopic methods, including Fourier-transform infrared (FTIR), nuclear magnetic resonance spectroscopy, and analysis of elements. In the laboratory, we assessed the impact of nine 2-pyrazoline-5-one derivatives on the fourth larval stage of the cotton leaf worm (Spodoptera littorals), Monacha obstructa, and Tetranychus urticae adults. We used abamectin as the reference compound. The LC50 values of compound 3 for S. littorals, M. obstructa, and T. urticae are 0.90, 0.70, and 0.52 mg a.i./L, respectively. The LC50 values for the same compound for S. littorals, M. obstructa, and T. urticae are 0.80, 0.60, and 0.45 mg a.i./L, respectively. Therefore, it is a remarkably potent compound. Compound 6 exhibited lower levels of danger, as indicated by LC50 values of 1.94, 1.90, and 1.83 mg a.i./L against the three tested pests. Ten days after treatment, all three pests were moderately toxic to the remaining 2-pyrazolin-5-one derivatives, 1, 2, 4, 5, and 7–10. Conversely, the mortality rate of the tested compounds increased when the treated individuals were exposed to high concentrations. These 2-pyrazolin-5-one derivatives are suggested as suitable alternatives and foundational structures for developing novel insecticides.
Keywords
Bioactivity
2-Pyrazolin-5-one
Cyanoacetohydrazide
Spodoptera littorals
Monacha obstructa
Tetranychus urticae
1 Introduction
Some of the most dangerous pests harming Egypt's crops are the leafworm of cotton (Spodoptera littoralis, the land snail (Monacha obstructa), and the twospotted spider mite (Tetranychus urticae). Therefore, there is an urgent demand for innovative and environmentally friendly pesticides that employ various methods. 2-Pyrazoline derivatives, used as pesticides, have a significant effect on agriculture (Hassn et al.,1994; Silver and Soderlund, 2006). 2-pyrazolin-5-one analogues, one of these heterocycles, are extensively employed in biological and synthetic chemistry (Abeed 2015; and Omaiya et al., 2020).
Conversely, cyanoacetohydrzide was used as a precursor for the formation of heterocycles and the study of their biological functions (Refat and Fadda, 2013; Mahmoud et al., 2020; Cheng et al., 2021; Ji et al., 2021; Salah et al., 2021; and Monier et al., 2020).
Motivated by the previously mentioned aspects and as a progression of our endeavours to create novel bioactive heterocyclic compounds (Abeed 2015; Abeed et al., 2019a,b, 2022, 2023), that we decided to produce a novel series of heterocycles with the 2-pyrazolin-5-one motif to investigate their biological actions in the laboratory against the three pests observed. The reference compound used was abamectin, which exhibits high efficacy against the pests being studied (Ali 2021). Abamectin has demonstrated efficacy as a molluscicide against various species of terrestrial snails (Abdallah et al., 2015 and Gaber et al., 1994), which currently pose an important threat to sustainable crop production in Egypt (Heiba et al., 2018; Shahawy et al., 2018). Abamectin is primarily used to control mites. The two-spotted spider mite infests various crops, causing immediate harm to the greenhouse and crops (Abdel-Halim and Kalmosh, 2019). Abamectin exhibits the highest level of toxicity towards T. urticae and serves a distinct purpose in integrated mite management, as stated by Ismail et al. (2006), Salem et al. (2022), and Sheasha et al. (2023). An investigation was conducted to assess the potential compatibility of chemical control methods to develop an improved integrated pest management (IPM) strategy for cotton leaf worms, land snails, and mites.
2 Results and discussion
2.1 Chemistry
The interaction of 4-acetyl-2-pyrazolin-5-one with cyanoacetic acid hydrazide yielded acetohydrazide 1 (Abdelall 2009). Employing compound 1 as a starting point, new heterocycles of 2-pyrazoline-5-one derivatives were created. The compound 2-oxo-2H-chromene-3-carbohydrazide 2 was produced by heating it with salicylaldehyde in dioxane. On the other hand, when the nitrile derivative 1 was heated with an equimolar amount of sulfur element and phenyl isothiocyanate in dioxane with a small quantity of triethyl amine (TEA), 4-aminothioxothiazole 3 was obtained (Scheme 1). Compounds 2 and 3 were structurally characterized using FTIR, NMR, MS, and analysis of elements techniques.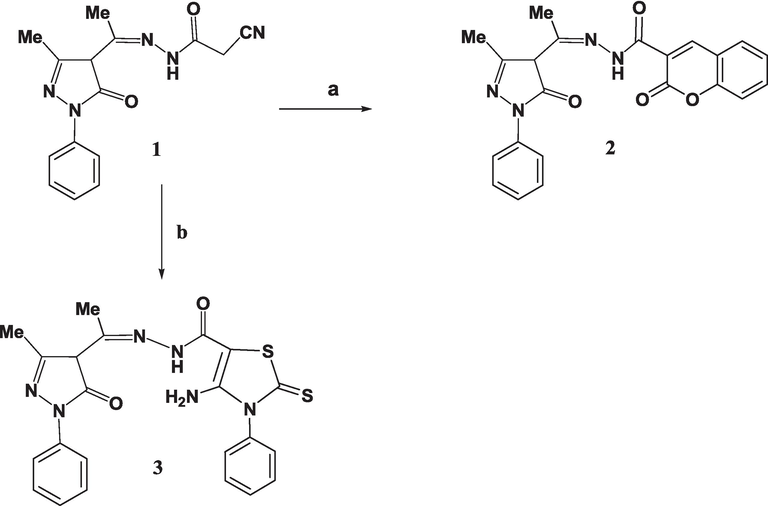
(a) Salicylaldehyde, dioxane, Pip., 5 h; (b) Phenyl isothiocyanate, S, TEA, EtOH, 5 h.
The FTIR of 4-aminothioxothiazole 3 proved an absence of the carbonitrile group band and the emergence of new peaks at v = 3430, 3312, and 3230 cm−1, which were assigned to the NH and NH2 groups. Moreover, a band at v = 1282 cm−1 was observed, confirming the existence of the C═S functional group. The 1H NMR analysis provides additional evidence of a signal from an amino group and aromatic protons within a chemical shift range of δ = 6.98–8.20 ppm. In addition, the 13C NMR spectroscopic study verifies the existence of a C=S group at a chemical shift of δ = 174.1 ppm.
Based on the reactivity of acetohydrazide 1, it interacted with active methylenes as malononitrile, ethyl cyanoacetate, and acetylacetone, resulting in the formation of 2-oxopyridine-3-carbonitrile analogues 4, 5, and 6, respectively. The analytical data for the products obtained agreed with the assigned structures (Scheme 2).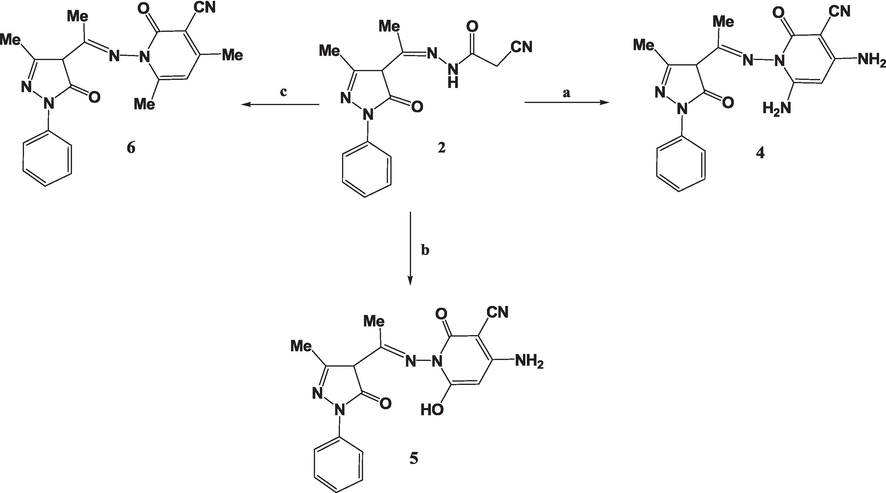
(a) Malononitrile, dioxane, TEA, 5 h; (b) Ethyl cyanoacetate, dioxane, TEA, 5 h; (c) Acetyl acetone, dioxane, TEA, 5 h.
The FTIR spectra of compounds 4 and 5 exhibited the emergence of new bands in the range of υ = 3187 to 3387 cm−1, indicating the presence of NH2 groups. Additionally, the FTIR spectrum of compound 6 displayed a new peak at v= 3422 cm−1, which can be assigned to the absorption band of an OH group. The 1H NMR analysis of the synthesized compounds 4–6 confirmed the presence of an amino group in either compound 4 or 5 and a hydroxyl group in compound 7.
The 13C NMR spectra corroborate the findings of the FTIR and 1H NMR spectroscopy. As an illustration, the spectrum of 4 exhibited distinct signals at δ = 12.7, 20.6, 115.8, 160.4, and 166.0 ppm, which corresponded to (pyrazolone-CH3), (CH3), (CN), (pyrazolone-CO), and (CO), respectively. The reactivity of 1 with electrophilic reagents was investigated. Consequently, the treating 1 with various aromatic aldehydes within a hot dioxane, augmented with just a little TEA, formed the respective arylidenes 7 and 8. The NMR spectroscopy of arylidenes 7 and 8 indicated that the CH2 signal of compound 2 was no longer present. Compounds 7 and 8 underwent hydrazinolysis in hot ethanol to produce new pyrazole derivatives 9 and 10 (Scheme 3). The FTIR analysis of compounds 9 and 10 indicated the lack of the stretching absorption peak related to the nitrile functional group.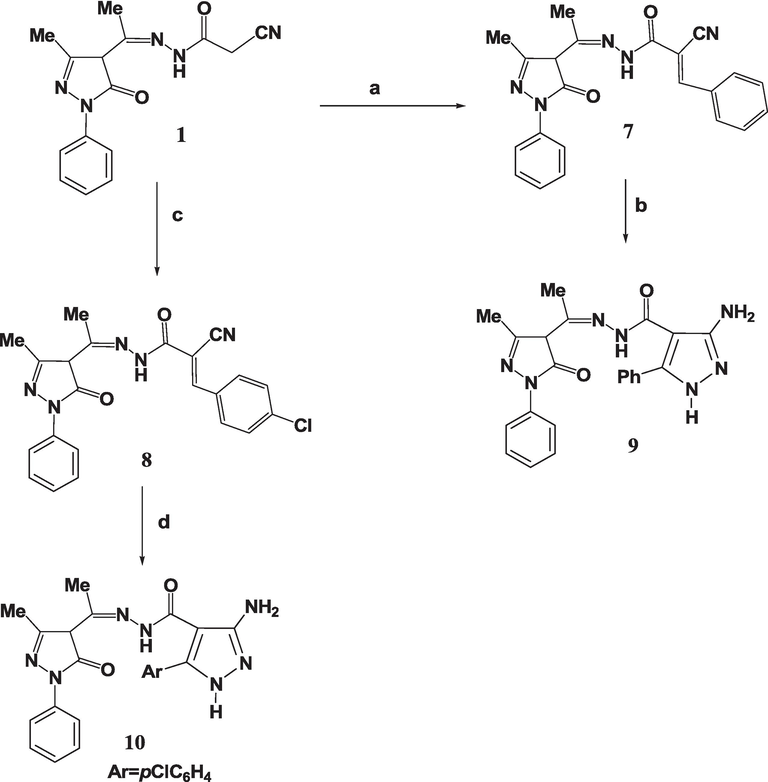
(a) Benzaldehyde, dioxane, TEA, 5 h; (b) Hydrazine hydrate, EtOH, 6 h; (c) p-Chlorobenzaldehyde, dioxane, TEA, 5 h.; (d) Hydrazine hydrate, EtOH, 6 h.
The formation mechanism of compounds 9 and 10 was proposed to occur through the reaction of Michael addition. The nitrogen atoms for hydrazine initiate an attack on the β-carbon of the α,β-unsaturated nitrile structure, resulting in a 1,5-exo-dig cyclization (Scheme 4).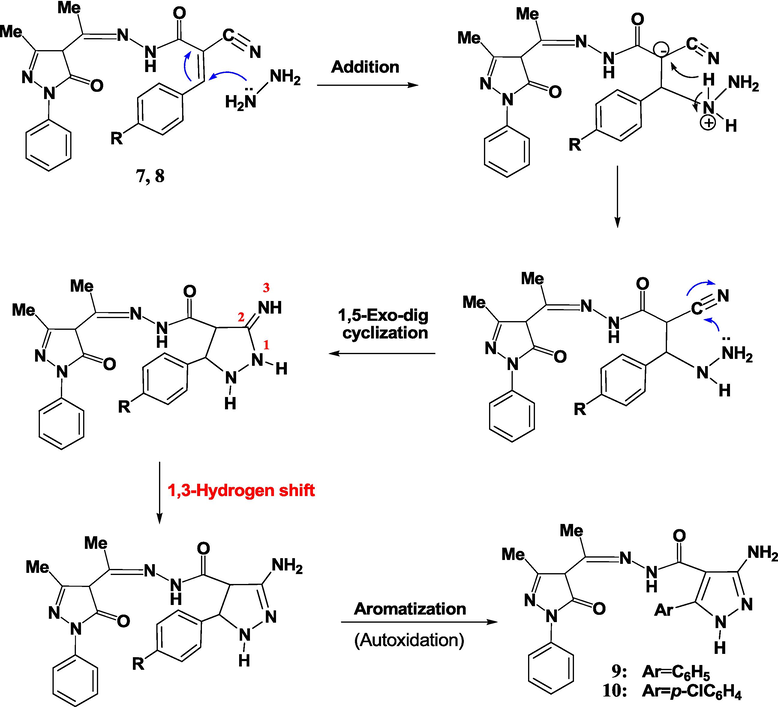
The proposed mechanism for synthesizing pyrazoles 9 and 10.
2.2 Biological activity
2.2.1 The effect of 2-pyrazolin-5-one derivatives on Spodoptera littoralis, Monacha obstructa, and Tetranychus urticae
As shown in Tables 1-3, the efficacy of synthetic compounds (1–10) and abamectin against three pests, namely Spodoptera littoralis, Monacha obstructa, and Tetranychus urticae, was evaluated in a laboratory setting using the leaf dipping method. The results were obtained after a treatment period of 72 h. Table 1 shows that abamectin had the highest bioactivity against S. littoralis, with an LC50 of 0.80 mg a.i./L and a 100 % toxicity index. The new compounds 2-thioxothiazole 3 and 2-oxochromene 2 showed comparable effectiveness to ambactin, with corresponding LC50 levels of 0.90 and 0.92 mg a.i./L. Their toxicity indices were determined to be 86.53 % and 84.90 %. Their toxicity indexes were calculated to be 86.53 % and 84.90 %. The toxicity index for the reference pesticide, compound 3, and compound 2 against M. obstructa were 100.0 %, 85.71 %, and 73.17 % respectively. The LC50 levels for these compounds were 0.60, 0.70, and 0.82 mg a.i./L, respectively, as shown in Table 2. Furthermore, the effectiveness of these substances against T. urticae was demonstrated by LC50 values of 0.45, 0.52, and 0.53 mg a.i/L, resulting in toxicity levels of 100.0 %, 86.53 %, and 84.90 %, respectively, as indicated in Table 3. Nevertheless, upon assessment of the remaining compounds (4–10) against each of the three pests, they exhibited lower efficacy. CL.L.U. confidence limit (lower- upper). CL.L.U. confidence limit (lower- upper)- Ti. Toxicity index. CL.L.U.: Confidence limit (lower- upper). Ti: Toxicity index.
Compound
No.
LC50
Slope ± SE
Ti
X2
Abamectin
0.80
(0.63–1.02)1.11 ± 0.22
100.0
0.42
1
0.94
(0.79–1.95)1.51 ± 0.66
85.10
0.46
2
0.92
(0.59–2.11)1.31 ± 0.43
86.96
0.53
3
0.90
(0.73–0.94)1.22 ± 0.61
88.88
0.55
4
0.96
(0.72–1.11)1.71 ± 0.71
83.33
0.60
5
0.93
(0.81–1.98)1.41 ± 0.59
86.02
0.44
6
1.94
(1.11–2.52)1.91 ± 0.63
41.23
0.84
7
1.81
(1.23–2.09)1.83 ± 0.72
44.19
0.69
8
0.95
(0.71–0.95)1.69 ± 0.81
84.21
0.51
9
1.62
(1.02–2.53)1.81 ± 0.72
49.38
0.67
10
1.92
(0.97–1.94)1.90 ± 0.52
41.66
0.57
Compound
No.
LC50
Slope ± SE
Ti
X2
Abamectin
0.60
(0.31–0.82)1.21 ± 0.62
100.0
0.58
1
0.91
(0.89–1.11)1.55 ± 0.61
65.93
0.45
2
0.82
(0.71–0.89)1.42 ± 0.51
73.17
0.62
3
0.70
(0.61–0.98)1.41 ± 0.44
85.71
0.46
4
1.42
(1.02–2.24)1.71 ± 0.51
42.25
0.62
5
0.90
(0.82–1.24)1.51 ± 0.55
66.66
0.63
6
1.90
(1.73–2.11)1.81 ± 0.40
31.57
0.68
7
1.43
(1.21–1.80)1.63 ± 0.66
41.95
0.70
8
0.98
(0.81–1.01)1.61 ± 0.43
61.22
0.45
9
1.21
(1.00–1.43)1.45 ± 0.62
49.58
0.72
10
1.71
(1.31–1.99)1.80 ± 0.42
35.08
0.56
Compound
No.
LC50
Slope ± SE
Ti
X2
Abamectin
0.45
(0.32–0.59)1.21 ± 0.42
100.0
0.68
1
0.62
(0.33–0.92)1.55 ± 0.47
72.58
0.44
2
0.53
(0.24–0.82)1.31 ± 0.59
84.90
0.31
3
0.52
(0.30–0.81)1.25 ± 0.82
86.53
0.63
4
0.74
(0.42–0.93)1.66 ± 0.72
60.81
0.52
5
0.58
(0.32–0.62)1.54 ± 0.31
77.58
0.44
6
1.83
(1.00–2.52)1.91 ± 0.91
24.59
0.72
7
1.72
(1.21–2.23)1.81 ± 0.58
26.16
0.48
8
0.71
(0.43–0.99)1.61 ± 0.81
63.38
0.52
9
1.31
(0.99–2.11)1.76 ± 0.67
34.35
0.63
10
1.51
(1.01–2.24)1.78 ± 0.71
29.80
0.78
The impact of the highest concentration of the tested compounds on S. littoralis, M. obstructa, and T. urticae after 3, 7, and 10 days of treatment is indicated in Tables 4 and 5. Table 4 provides a detailed analysis showing that the pests evaluated were exposed to Abamectin and two other tested compounds, 3 and 2. Over time, their ability to survive decreased gradually, reaching the lowest level after 10 days of treatment. Specifically, the surviving individuals for S. littoralis, M. obstructa, and T. urticae were 2, 0, and 1, respectively. The survival rate of individuals increased when they were exposed to different substances.
Compound
No.
Conc
(mg a.i/ L)
Post-treated count (days)
3 days
7 days
10 days
S.
littoralis
M. obstructa
T.
urticae
S.
littoralis
M.
obstructa
T.
Urticae
S. littoralis
M.
obstructa
T.
urticae
Abamectin
0.45
4 a
3 a
1 a
2 a
1 a
1 a
2 a
0 a
0 a
1
0.35
7b
8b
8c
5b
5b
7c
3 a
2 a
4b
2
0.65
6 a
7b
7c
4 a
4b
6b
1 a
1 a
1 a
3
0.50
5 a
6b
4b
3 a
2 a
1 a
0 d
0 a
0 a
4
0.85
8b
9c
10 d
7b
7c
8c
1 a
5b
6b
5
0.40
6 a
7b
7c
4 a
5b
7c
1 a
2 a
2 a
6
0.58
9b
12 d
14 e
9c
10 d
13 e
4b
9c
11 d
7
0.30
9b
10c
11 d
8c
10 d
11 d
1 a
6b
5b
8
0.60
8b
9c
9c
6b
6b
8c
1 a
4b
5b
9
0.65
9b
10c
11 d
3 a
7c
10 d
1 a
5b
6b
10
0.55
9b
10c
13 d
8c
10 d
11 d
2 a
6b
10 d
Control
−
15c
15 e
15 e
15 d
15 e
15 e
15c
15 d
15c
Compound
No.
Conc.(mg a.i/ L)
Average reduction percentage
%R Grand mean
3 days
7 days
10 days
S.
littoralis
M. obstructa
T. urticae
S.
littoralis
M. obstructa
T. urticae
S.
Littoralis
M. obstructa
T. urticae
Abamectin
0.45
73.33
80.00
93.33
86.66
93.33
93.33
86.66
100.00
100.00
88.14
1
0.35
53.33
46.66
46.66
66.66
66.66
53.33
80.00
86.66
73.33
64.43
2
0.65
60.00
53.33
53.33
73.33
73.33
60.00
93.33
93.33
93.33
72.59
3
0.50
66.66
60.00
73.33
80.00
86.66
93.33
100.00
100.00
100.00
85.18
4
0.85
46.66
40.00
33.33
53.33
53.33
46.66
93.33
66.66
60.00
54.81
5
0.40
60.00
53.33
53.33
73.33
66.66
53.33
93.33
86.66
86.66
69.62
6
0.58
40.00
20.00
6.66
40.00
33.33
13.33
73.33
40.00
26.66
32.59
7
0.30
40.00
33.33
26.66
46.66
33.33
26.66
93.33
60.00
66.66
48.88
8
0.60
46.66
40,00
40.00
60.00
60.00
46.66
93.33
73.33
66.66
58.51
9
0.65
40.00
33.33
26.66
50.00
53.33
33.33
93.33
66.66
60.00
50.73
10
0.55
40.00
33.33
13.33
46.66
33.33
26.66
86.66
60.00
33.33
41.47
Table 5 shows that the decrease in writing ability becomes more significant as the treatment duration increases, reaching its highest level after 7 and 10 days of treatment. Abamectin had the most critical effect on the three pest populations when used with the greatest concentration. Following 7 and 10 days of medication, the values for S. littorals, M. obstructa, and T. urticae were 86.66 %, 93.33 %, and 93.33 %, and 86.66 %, 100.00 %, and 100.00 %, respectively. After the same amount of time, 2-thioxo-2,3-dihydrothiazole-5-carbohydrazide 3 showed reduction percentages of 80.00 %, 86.66 %, and 93.33 %, respectively, as well as 100.00 % reduction in all three trials. On the other hand, 2-oxo-2H-chromene-3-carbohydrazide 2 exhibited reduction percentages of 73.33 %, 73.33 %, 60.00 %, and 93.33 % in all three trials.
On the other hand, 2-oxopyridine 6 exhibited the slightest reduction in activity against S. littorals, M. obstructa, and T. urticae, with decreases of 73.33 %, 40.00 %, and 26.66 % respectively after 7 days and 10 days. The compounds studied had varying effects on the three pests, with abamectin having the most significant impact, followed by compounds 3, 2, 5, 1, 8, 4, 9, 7, 10, and 6. The mean values for the effects of these compounds were 88.14 %, 85.18 %, 72.59 %, 69.62 %, 64.43 %, 58.51 %, 54.81 %, 50.73 %, 48.88 %, 41.47 %, and 32.59 %, respectively.
Morphological symptoms of deceased individuals from the three pests treated with synthetic compounds are depicted in Figure 1. When cotton leafworm larvae undergo a process in which their bodies shrink and stop moving altogether, their color changes to dark brown (Figure 1. A). The clover snail M. obstructa gradually increases its secretion of mucous fluids, relaxing its soft body at the shell's opening, causing cessation of movement and, ultimately, death of the snail (Figure 1. B). The administration of the tested compounds resulted in immobility, dehydration, and mortality in T. urticae mites (Figure 1. C).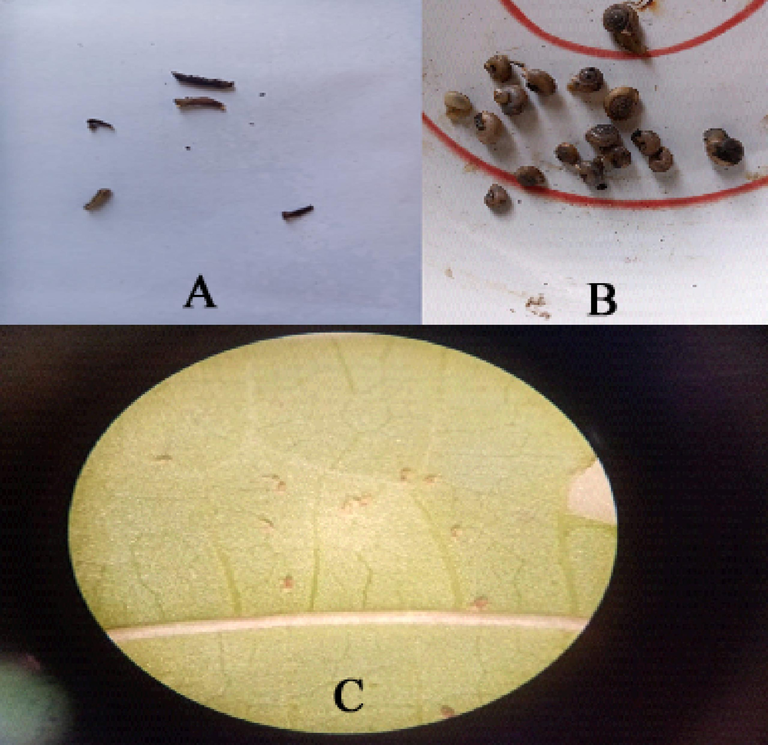
Observable signs of deceased specimens subjected to synthetic substances (A: Spodoptera littorals; B: Monacha obstructa; C: Tetranychus urticae).
The identical letters within the same column indicate their lack of significance.
2.2.2 Discussion
The current study demonstrated that the investigated compounds showed varying potency against the three pests, with abamectin serving as the standard drug. These findings align with multiple prior studies that demonstrate abamectin's robust effectiveness. The reference compound, abamectin, was most harmful to T. urticae, according to study results (Keratum et al., 2010, Salem et al., 2022, and Abd-Allah et al., 2022). In addition, multiple studies have demonstrated that abamectin exhibits superior efficacy in managing S. littorals, as reported by Abdel-Latif and Abd-Allah (2013) and Ali 2021. Abamectin was highly toxic to land snails (Abdel-Halim et al., 2021). Still, it was more effective against the glassy clover snail, M. obstructa, than methomyl, profenofos, and chlorfenapyr compared to the control (Sallam et al., 2016).
Abd El-Lateef et al., (2023) produced specific pyridines as neonicotinoid analogues to assess their toxicity in adult and nymph stages of Aphis craccivora, a broad category of insecticides. Fadda et al., (2017) discovered that 1,3,4-thiadiazoles exhibited the highest level of toxicity towards the second instar larvae of S. littoralis following a 7-day period of contact, as assessed by the leaf dip technique. The molluscicidal capabilities of several novel compounds comprising thiophene, pyrazole and thiadiazol, versus Biomphalaria alexandrinasnails (Fadda et al., 2009). El Shehry (2010), assessed the effectiveness of novel benzofuran-containing isoxazole, pyrazole, pyridine, pyrimidine, 1,4-thiazine, and 1,3,4-thiadiazine compounds against B. alexandrina. The study results indicated that these compounds exhibited significant molluscicidal activity, showing potential for further development. Hamama et al., (2018) employed the leaf-dip method to assess the acaricidal efficacy of 1,2,4-triazines on T. urticae eggs and adult females. The compounds exhibited lethal properties, displaying varying toxicity against T. urticae.
3 Experimental
3.1 Instrumentation and chemicals
The substances used in this work were of laboratory-grade quality. The degrees of melting were measured using an APP Digital ST 15 instrument and uncorrected. The FTIR spectroscopic data were acquired using the Shimadzu company-408 infrared analyzer that are presented as cm−1 units. The nuclear magnetic resonance spectra were obtained using the JEOL ECA II 500 MHz spectrometer. Chemical shifts are measured in parts per million (ppm), using TMS as an internal reference. A Varian MAT312 apparatus in EI mode with an energy of 70 eV was used for mass spectrometry at the Faculty of Science, Assiut University, Egypt. The elements were examined using the System GmbH Vario ELV2.3 1998 CHNS Mode.
3.1.1 2-Cyano-N'-(1-(3-methyl-5-oxo-1-phenyl-2-pyrazolin-4-yl)ethylidene)acetohydrazide (1)
3.1.2 N'-(1-(3-Methyl-5-oxo-1-phenyl-2-pyrazolin-4-yl) ethylidene)-2-oxo-2H-chromene-3-carbohydrazide (2)
A solution of 1 (1.19 g, 4 mmol) and salicylaldehyde (0.49 g, 4 mmol) in dioxane (20 ml) with piperidine (5 drops) was heated for 5 h. After cooling, the reaction product was added to ice water along with a few drops of HCl, The product was collected, dried and crystallized using toluene, giving 2: White crystals (0.88 g, 71 % yield); mp 155–157 °C; IR (KBr, cm−1): 3221 (NH), 3049 (Ar-H), 2925 (aliphatic-H), 1655 (C=O), 1642 (C=O), 1626 (pyrazolone-C=O); 1H NMR (500 MHz, Acetone‑d6): δ 2.30 (s, 3H, pyrazolone-CH3), 2.38 (s, 3H, CH3), 6.94–8.09 (m, 10H, Ar-H and pyrazolone-H), 8.92 (s, 1H, CH), 11.04 (s, 1H, NH, exchanged with D2O); 13C NMR (125 MHz, Acetone‑d6): 12.9 (pyrazolone-CH3), 20.3 (CH3), 100.7, 113.1, 115.3, 118.9, 119.9, 120.8, 122.8, 123.5, 124.1, 125.9, 127.3, 129.5, 131.4, 134.2, 138.6, 148.6, 149.2, 159.4 (pyrazolone-C=O), 165.4 (C=O), 177.5 (C=O); EI. MS: m/z 402.1 (M+, 60 %). Anal. Calcd. for C22H18N4O4 (402.40): C, 65.66; H, 4.51; N, 13.92 %. Found: C, 65.56; H, 4.40; N, 13.78 %.
3.1.3 4-Amino-3-phenyl-N'-(1-(3-methyl-5-oxo-1-phenyl-2-pyrazolin-4-yl)ethylidene)-2-thioxo-2,3-dihydrothiazole-5-carbohydrazide (3)
A solution of acetohydrazide derivative 1 (1.19 g, 4 mmol), elemental sulfur (0.13 g, 4 mmol), and phenyl isothiocyanate (0.52 g, 4 mmol) in ethanol (25 ml) with triethylamine (0.40 ml) was refluxed for 5 h. On cooling and throwing into cold water, the solid result was recrystallized using ethanol, producing 3: Orange crystals (1.23 g, 66 % yield); mp 126–127 °C; IR (KBr, cm−1): 3430, 3312, 3230 (NH, NH2), 3039 (Ar-H), 1665 (C=O), 1631 (pyrazolone-C=O), 1282 (C=S); 1H NMR (500 MHz, CDCl3): δ 2.31 (s, 3H, pyrazolone-CH3), 3.40 (s, 3H, CH3), 6.98–8.20 (m, 13H, Ar-H, pyrazolone-H and NH2), 11.00 (s, 1H, NH, exchanged with D2O); 13C NMR (125 MHz, CDCl3): 12.8 (pyrazolone-CH3), 20.4 (CH3), 100.6, 115.5, 115.9, 118.5, 119.4, 120.2, 121.6, 122.4, 124.2, 125.8, 127.2, 128.4, 130.3, 134.8, 137.6, 148.5, 149.1, 159.6 (pyrazolone-C=O), 165.4 (C=O), 174.1 (C=S); EI. MS: m/z 464.3 (M+, 71 %). Anal. Calcd. for: C22H20N6O2S2 (464.56): C, 56.88; H, 4.34; N, 18.09; S, 13.80.%. Found: C, 56.52; H, 4.65; N, 17.89; S, 13.80.
3.1.4 A general approach to synthesizing pyridine derivatives (4–6)
Equal quantities of 1 (1.48 g, 5 mmol) and active methylene reagents like malononitrile, ethyl cyanoacetate, and acetylacetone (5 mmol) were heated in a solution of dioxane (20 ml) with triethylamine (a few drops) for 5 h. The resulting product was subsequently cooled and dried under vacuum conditions. The remaining residue was triturated with ethanol, and the resultant product was filtered, dried, and recrystallized using ethanol.
3.1.4.1 1-((1-(3-Methyl-5-oxo-1-phenyl-2-pyrazolin-4-yl)ethylidene)amino)-4,6-diamino-2-oxo-1,2-dihydropyridine-3-carbonitrile (4)
Red brown crystals (1.20 g, 66 % yield); mp 307–309 °C; IR (KBr, cm−1): 3387, 3293, 3273 (2 NH2), 3029 (Ar-H), 2227 (CN), 1663 (C=O), 1629 (pyrazolone-C=O); 1H NMR (500 MHz, DMSO‑d6): δ 2.32 (s, 3H, pyrazolone-CH3), 3.39 (s, 3H, CH3), 4.72 (s, 1H, pyridine-H), 5.44 (s, 2H, NH2), 6.50 (s, 2H, NH2), 7.54–8.15 (m, 6H, Ar-H and pyrazolone-H); 13C NMR (125 MHz, DMSO‑d6): 12.7 (pyrazolone-CH3), 20.6 (CH3), 99.8, 110.5, 115.8 (CN), 120.4, 122.6, 124.3, 126.0, 128.0, 128.8, 131.5, 134.9, 136.4, 137.8, 141.2, 160.4 (pyrazolone-C=O), 166.0 (C=O); EI. MS: m/z 363.1 (M+, 59 %). Anal. Calcd. for: C18H17N7O2 (363.37): C, 59.50; H, 4.72; N, 26.98 %. Found: C, 59.62; H, 4.61; N, 26.86.
3.1.4.2 1-((1-(3-Methyl-5-oxo-1-phenyl-2-pyrazolin-4-yl)ethylidene)amino)-4-amino-6-hydroxy-2-oxo-1,2-dihydropyridine-3-carbonitrile (5)
Pale orange crystals (1.13 g, 62 % yield); mp 219–221 °C; IR (KBr, cm−1): 3457 (OH), 3282, 3187 (NH2), 3031 (Ar-H), 2212 (CN), 1659 (C=O), 1632 (pyrazolone-C=O); 1H NMR (500 MHz, DMSO‑d6): δ 2.30 (s, 3H, pyrazolone-CH3), 3.28 (s, 3H, CH3), 4.70 (s, 1H, pyridine-H), 5.46 (s, 2H, NH2), 7.49–8.19 (m, 6H, Ar-H and pyrazolone-H), 11.22 (s, 1H, OH); 13C NMR (125 MHz, DMSO‑d6): 13.0 (pyrazolone-CH3), 20.8 (CH3), 99.6, 111.4, 116.2 (CN), 121.2, 123.1, 124.7, 125.2, 128.2, 130.2, 131.6, 135.0, 136.2, 137.6, 140.9, 161.0 (pyrazolone-C=O), 166.6 (C=O); EI.MS: m/z 364.1 (M+, 70 %). Anal. Calcd. for: C18H16N6O3 (364.36): C, 59.34; H, 4.43; N, 23.07 %. Found: C, 59.45; H, 4.32; N, 22.96 %.
3.1.4.3 1-((1-(3-Methyl-5-oxo-1-phenyl-2-pyrazolin-4-yl)ethylidene)amino)-4,6-dimethyl-2-oxo-1,2-dihydropyridine-3-carbonitrile (6)
Orange crystals (0.26 g, yield 66 %); mp 203–205 °C; IR (KBr, cm−1): 3422 (OH), 3059 (Ar-H), 2930 (aliphatic-H), 2208 (CN), 1637 (pyrazolone-C=O); 1H NMR (500 MHz, DMSO‑d6): δ 2.31 (s, 3H, pyrazolone-CH3), 2.60 (s, 3H, pyridine-CH3), 2.74 (s, 3H, pyridine-CH3), 3.27 (s, 3H, CH3), 4.80 (s, 1H, pyridine-H), 7.40–8.10 (m, 6H, Ar-H and pyrazolone-H); EI.MS: m/z 361.2 (M+, 59 %). Anal. Calcd. for: C20H19N5O2 (361.40): C, 66.47; H, 5.30; N,19.38 %. Found: C, 66.56; H, 5.20; N, 19.26 %.
3.1.5 General route for synthesizing 3-aryl-2-cyanoacrylohydrazide (7, 8)
The acetohydrazide solution 1 (1.19 g, 4 mmol) was refluxed for 5 h together with appropriate aromatic aldehydes (4 mmol) in dioxane (20 ml) and triethylamine (a few drops). The precipitate underwent filtration, drying, and purification using ethanol, resulting in a yield of 7 and 8.
3.1.5.1 2-Cyano-N'-(1-(3-methyl-5-oxo-1-phenyl-2-pyrazolin-4-yl)ethylidene)-3-phenyl-acrylo-hydrazide (7)
White crystals (1.22 g, 64 % yield); mp 203–204 °C; IR (KBr, cm−1): υ 3227 (NH), 3077 (Ar-H), 2212 (CN), 1653 (C=O); 1H NMR (500 MHz, pyridine‑d6) δ (ppm): 2.34 (s, 3H, pyrazolone-CH3), 2.38 (s, 3H, CH3), 7.10–8.23 (m, 12H, Ar-H, pyrazolone-H and = CH–), 11.10 (s, 1H, NH); 13C NMR (125 MHz, pyridine‑d6) δ (ppm): 13.4 (pyrazolone-CH3), 21.2 (CH3), 99.8, 112.2, 114.4, 116.2 (CN), 117.7, 118.8, 121.0, 122.4, 124.2, 125.7, 126.6, 128.5, 130.4, 132.2, 134.0, 135.5, 137.6, 140.8, 150.3, 163.4 (pyrazolone-CO), 167.2 (C=O); EI.MS: m/z 385.2 (M+, 66 %). Anal. Calcd. for: C22H19N5O2 (385.42): C, 68.56; H, 4.97; N, 18.17 %. Found: C, 68.45; H, 4.86; N, 18.28.
3.1.5.2 2-Cyano-N'-(1-(3-methyl-5-oxo-1-phenyl-2-pyrazolin-4-yl)ethylidene)-3-(4-chlorophenyl) acrylohydrazide (8)
White crystals (1.34 g, 68 % yield); mp 199–201 °C; IR (KBr, cm−1): υ 3231 (NH), 2925 (aliphatic-H), 2227 (CN), 1655 (C=O); 1H NMR (500 MHz, pyridine‑d6) δ (ppm): 2.31 (s, 3H, pyrazolone-CH3), 2.39 (s, 3H, CH3), 7.11–8.20 (m, 11H, Ar-H, pyrazolone-H and = CH–), 11.09 (s, 1H, NH); EI. MS: m/z 419.7 (M+, 72 %), 421.6 (M++2, 24 %). Anal. Calcd. for: C22H18N5O2Cl (419.86): C, 62.93; H, 4.32; N, 16.68; Cl, 8.44 %. Found: C, 62.83; H, 4.21; N, 16.57; Cl, 8.56 %.
3.1.6 General process synthesizing 5-aryl-3-aminopyrazole-4-carbohydrazide (9, 10)
A solution of arylidene 7 or 8 (4 mmol) in 20 ml ethanol was prepared. Hydrazine hydrate (0.13 ml, 4 mmol) was added to this solution, and the mixture was refluxed for 6 h. Upon cooling, the product were obtained by collecting and recrystallizing the solid product in a suitable solvent.
3.1.6.1 3-Amino-N'-(1-(3-methyl-5-oxo-1-phenyl-2-pyrazolin-4-yl)ethylidene)-5-phenyl-1H-pyraz-ole-4-carbohydrazide (9)
White crystals (g, 60 % yield); mp 233–235 °C; IR (KBr, cm−1): υ 3229 (NH), 3098 (Ar-H), 2211 (CN), 1626 (C=O); 1H NMR (500 MHz, CDCl3) δ (ppm): 2.32 (s, 3H, pyrazolone-CH3), 2.41 (s, 3H, CH3), 6.38 (s 2H, NH2, exchanged with D2O), 7.10–8.08 (m, 11H, Ar-H, pyrazolone-H), 9.00 (s, 1H, NH, exchanged with D2O); 9.87 (s, 1H, NH, exchanged with D2O); 13C NMR (125 MHz, CDCl3) δ (ppm): 13.6 (pyrazolone-CH3), 21.0 (CH3), 99.2, 111.9, 114.8, 116.0, 119.2, 121.4, 122.6, 123.8, 124.8, 126.4, 127.6, 130.4, 132.0, 134.1, 135.4, 136.2, 137.2, 142.0, 162.4 (pyrazolone-C=O), 168.0 (C=O); EI. MS: m/z 415.3 (M+, 82 %). Anal. Calcd. for: C22H21N7O2 (415.45): C, 63.60; H, 5.09; N, 23.60 %. Found: C, 63.71; H, 4.99; N, 23.49 %.
3.1.6.2 3-Amino-N'-(1-(3-methyl-5-oxo-1-phenyl-2-pyrazolin-4-yl)ethylidene)-5-(4-chlorophenyl)-1H-pyrazole-4-carbohydrazide (10)
White crystals (g, 61 % yield); mp 240–242 °C;; IR (KBr, cm−1): υ 3313 (NH), 3056 (Ar-H), 2213 (CN), 1668 (C=O); 1H NMR (500 MHz, CDCl3) δ (ppm): 2.33 (s, 3H, pyrazolone-CH3), 2.49 (s, 3H, CH3), 6.35 (s, 2H, NH2, exchanged with D2O), 7.06–7.99 (m, 10H, Ar-H, pyrazolone-H), 9.04 (s, 1H, NH, exchanged with D2O); 10.10 (s, 1H, NH, exchanged with D2O); 13C NMR (125 MHz, CDCl3) δ (ppm): 13.4 (pyrazolone-CH3), 20.9 (CH3), 100.0, 112.2, 113.8, 115.9, 119.4, 121.6, 122.8, 123.9, 124.9, 125.8, 127.8, 129.8, 132.2, 134.4, 135.6, 136.8, 137.4, 142.4, 160.9 (pyrazolone-C=O), 168.8 (C=O); EI.MS: m/z 449.82 (M+, 71 %), EI.MS: m/z 451.86 (M++2, 23 %). Anal. Calcd. for: C22H20N7O2Cl (449.89): C, 58.73; H, 4.48; N, 21.79; Cl, 7.88 %. Found: C, 58.64; H, 4.38; N, 21.68; Cl, 7.99 %.
3.2 Biological activity
3.2.1 Abamectin 1.8 %
The reference compound was provided by Merck Company, Inc., located in Rahway, New Jersey, U.S.A.
3.2.1.1 Tested pests
The pests used in the toxicity experiments were obtained from a standard laboratory culture at the department of Cotton Pesticide Evaluation, Plant Protection Research Institute, Agricultural Research Center, Egypt. We gathered specimens of S. littorals, M. obstructa, and T. urticae from agricultural areas in Sidi Salem, Kafr El-Sheikh Governorate. We then transferred them to the laboratory, where they were kept at a temperature of 26 ± 2°C and a relative humidity of 70 ± 5 %. The S. littorals reference strain was cultivated on castor leaves for three generations until the larvae reached the fourth instar, which was subsequently utilized in the present investigation. Monacha obstructa snails in the adult stage were raised in plastic bags with wet, sterilized sandy-textured soil. The participants were given recently picked lettuce leaves (Lactuca sativa L.) to induce relaxation in the laboratory. Furthermore, mature T. urticae mites were raised by breeding.
3.2.1.2 Toxicity tests
Using the leaf dipping method, the toxicity of ten derivatives of 2-pyrazolin-5-one and abamectin as a reference drug was assessed against the three pests, S. littoralis, M. obstructa, and T. urticae. The compounds under investigation were prepared in three different quantities using distilled water and a control treatment that did not include any drug. The experiment utilized three duplicates for each concentration. Each replication of all examined bugs involved the use of five individuals. The dilutions were used to immerse fresh cotton and lettuce leaves for 20 s, after which they were allowed to air dry.
Consequently, the processed cotton leaves are delivered individually in a plastic container with a diameter of 10 cm. Each box was populated with five recently moulted fourth-instar S. littoral larvae. These larvae were provided with food by applying a delicate camel hair brush onto the treated cotton leaves for eating. Five mature glassy clover snails (M. obstructa) were placed singly in plastic containers filled with clay soil at 3–5 cm depth. The utilization of processed lettuce leaves facilitated feeding. Each container was wrapped with muslin cloth and fastened by bands of rubber, whereas the control group consisted of untreated lettuce discs. The efficacy of the new compounds against the twospotted spider mite and T. urticae was evaluated using the cotton leaf disc dip method. Cotton leaf discs with a diameter of 9 cm were positioned in Petri dishes, with a piece of filter paper placed beneath each disc. A total of five T. urticae specimens were placed in each dish on prepared discs using a delicate camel hair brush (Abo-Elghar et al., 1994). Throughout 10 days, each element of the therapy was meticulously observed daily.
3.2.1.3 Statistical analysis
The mortality percentage was determined at 3, 7, and 10 days and then corrected using Abbott's method (Abbott 1925). The LC50 concentrations were calculated via probit evaluation, as described by Finney (1971). The toxicity index of the examined substances was calculated utilizing the sun's method (Sun 1950). The toxicity index is determined by dividing the LC50 of the most potent chemical by the LC50 of the tested substance and subsequently dividing the quotient by 100. The data underwent analysis of variance (ANOVA) with a significance threshold of p < 0.05 following a normality test. The data were examined utilizing SPSS version 23.
4 Conclusion
The 2-pyrazolin-5-one nucleus was used for generating novel heterocycles. The structures were determined utilizing spectroscopic data like FTIR and NMR spectroscopy. The efficacy of the acquired compounds was assessed against specific pests, including the fourth larval stage of the cotton leaf worm, Spodoptera littorals, Monacha obstructa, and adult Tetranychus urticae. Our investigation showed that two drugs produced remarkable results.
Funding
This work was funded by the Researchers Supporting Project (RSP2024R120), King Saud University, Riyadh, Saudi Arabia.
CRediT authorship contribution statement
Ahmed Abdou O. Abeed: Writing – review & editing, Writing – original draft, Methodology, Investigation, Formal analysis, Data curation, Conceptualization. Emad A. El Shamy: . Sahar I.A. Abd El-Wahed: . Mohamed Mohany: . Marija Milošević: . Salim S. Al-Rejaie: . Hesham A.M. Ibrahim: . Abd El-Salam A. Farag: .
Acknowledgments
The authors would like to express their gratitude to the Researchers Supporting Project (RSP2024R120), King Saud University, Riyadh, Saudi Arabia.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- A method of computing the effectiveness of an insecticide. J. Econ. Eco. Entomol.. 1925;18(2):265-267.
- [Google Scholar]
- Functionali zed pyridines: Synthesis and toxicity evaluation of potential insecticidal agents against Aphis craccivora. ACS Omega. 2023;8(32):29685-29692.
- [Google Scholar]
- Abdallah, E.A., Abdelgalil, G.M., Kassem, F.A., Asran, A.A., Abou-Elnasser, H.S., 2015. Comparative molluscicidal activity of abamectin and methomyl against Eobania vermiculata (Müller) and Theba pisana (Müller). J. Plant Prot. Path. 6, 1671-1683. Abdel-Halim, K.Y.; Kalmosh, F.Sh., 2019. Acaricidal Activity of Nano-. (Acai: Tetranychidae). Acad J. life sci. 5(10): 81-86.
- Abd-Allah, G.E., Habashy, M. G., Shalaby, M.M., 2022. Efficacy of mint derivatives, Memtha Spicata L. against two species of Tetranychus Spp. (Acari: Tetranychidae) and the predator, Neoseiulus sp. Egypt Acad. J. Biolog. Sci. 15(1), 63.
- Comparative toxicity of abamectin and nano-derived from on land snail, Helix aspersa in attributing to cytotoxicity and biochemical alterations. World J. Adv. Res. Rev.. 2021;10(1):296-311.
- [Google Scholar]
- Abdel-Latif, G., Abd-Allah, M., 2013. Ovilarvicidal Activity of Abamectin and Spinosad against the cotton leafworm Spodoptera littorals (Boisd) (Lepidoptera: Noctuidea) under Laboratory Conditions the First Assiut Intarnational Conference of Horticulture, 24-25 th February Hort. Dep., Fas. Agric. Assiut Univ. Assiut, Egypt.
- Utilities of active methylene compounds and heterocycles bearing active methyl or having an active methine in the synthesis of triazoles, pyridazines, triazines and diazepines. Synth. Commun.. 2021;51:971-996.
- [Google Scholar]
- 2-Pyrazolin-5-one-based heterocycles: synthesis and characterization. J. Heterocycl. Chem.. 2015;52:1175.
- [Google Scholar]
- A Facile synthesis and reactions of some novel pyrazole-based heterocycles. Curr. Org. Synth.. 2019;16(3):405-412.
- [Google Scholar]
- Novel heterocyclic hybrids based on 2-pyrazoline: synthesis and assessment of anti-inflammatory and analgesic activities. Curr. Org. Synth.. 2019;16(6):921-930.
- [Google Scholar]
- Efficient synthetic access to novel indolo[2,3-b] quinoxaline-based heterocycles. Curr. Org. Synth.. 2022;19:177.
- [Google Scholar]
- Synthesis and reactions of fused pyrazolo[3,4-b]pyridine derivatives: insecticidal activity and digestive dysfunction against mosquito larvae. Curr. Org. Chem.. 2023;27:852-859.
- [Google Scholar]
- Histopathological effects of abamectin, thuringiensis and diflubenzuron on the midgut of Spodoptera littoralis (Lepidoptera Noctuidae) larvae. Bull. Ent. Soc. Egypt. 1994;21:41-52.
- [Google Scholar]
- Efficacy and residual effect of abamectin and chlorpyrifosmethyl against the second larval instar of the cotton leafworm Spodoptera littorals (Boisd) Under Field and Semi-Field Condition. J. Plant Prot. Path., Mansoura Univ.. 2021;12(1):5-9.
- [Google Scholar]
- Synthesis and inhibitory activity of novel 2, 3-dihydroquinolin-4(1H)-one derivatives as potential succinate dehydrogenase inhibitors. Eur. J. Med. Chem.. 2021;214:113246
- [Google Scholar]
- Synthesis and molluscicidal evaluation of some new pyrazole, isoxazole, pyridine, pyrimidine, 1,4-thiazine and 1,3,4-thiadiazine derivatives incorporating benzofuran moiety. Eur. J. Med. Chem.. 2010;45(11):4783-4787.
- [Google Scholar]
- Synthesis and molluscicidal activity of some new thiophene, thiadiazole and pyrazole derivatives. Eur. J. Med. Chem.. 2009;44(3):1250-1256.
- [Google Scholar]
- Synthesis and insecticidal assessment of some innovative heterocycles incorporating a thiadiazole moiety against the cotton leafworm. Spodoptera Littoralis. RSC Advances. 2017;7(63):39773-39785.
- [Google Scholar]
- Probit analysis (3rd ed.). London: Cambridge University Press; 1971. p. :318.
- Quantitative structure-activity relationships of insecticidal pyrazolines. Pestic. Sci.. 1994;42:291.
- [Google Scholar]
- Synthesis and acaricidal activity of some new 1,2,4-triazine derivatives. J. Heterocycl. Chem.. 2018;56(1):239-250.
- [Google Scholar]
- Terrestraial gastropods: survey and relationship between land snail assemblage and soil properties. J. Plant Prot. Path. Mansoura Univ.. 2018;9(3):219-224.
- [Google Scholar]
- Toxicological and biological studies of some compounds against the two-spotted spider mite, Tetranychus urticae and its predatory mite. Sci: Amplyseis gossipi on different host plants. T. Pest Contr. Environ; 2006. p. :227.
- Synthesis and anticancer activity of new coumarin-3-carboxylic acid derivatives as potential lactate transport inhibitors. Bioorg. Med. Chem.. 2021;29:115870
- [Google Scholar]
- Novel 2-indolinone thiazole hybrids as sunitinib analogues: Design, synthesis, and potent VEGFR-2 inhibition with potential anti-renal cancer activity. Eur. J. Med. Chem.. 2020;208:112752
- [Google Scholar]
- Heterocyclic steroids: Efficient routes for annulation of pentacyclic steroidal pyrimidines. Steroids. 2020;154:108548
- [Google Scholar]
- Synthesis and antimicrobial activity of some novel hydrazide, benzochromenone, dihydropyridine, pyrrole, thiazole and thiophene derivatives. Eur. J. Med Chem.. 2013;70:419-426.
- [Google Scholar]
- Pyridine-derived VEGFR-2 inhibitors: Rational design, synthesis, anticancer evaluations, in silico ADMET profile, and molecular docking. Arch. Pharm.. 2021;354:2100085.
- [Google Scholar]
- Effect of nano-abamectin in combating the two-spotted spider, Tetranychus urticae (Acai: Tetranychidae) under laboratory conditions International. J. Appl. Sci. Tech.. 2022;4(3):477-483.
- [Google Scholar]
- Toxicity of seven pesticides belonging to different chemical groups against the glassy clover snail, Moncha Obstructa by using three method of application under laboratory conditions. Inter. J. Res. Stud. Zool.. 2016;2(1):17-23.
- [Google Scholar]
- Population Density, food consumption and Damage Caused by the Land Snail Monacha cantiana to Some Vegatable Crops at Kafr El-Shiekh Governorate. J. Plant Prot. Path., Mansoura Univ.. 2018;9(9):601-604.
- [Google Scholar]
- Efficacy of some plant essential oils against two spotted spider mite Tetranychus urticae under laboratory condition. Pol. J. Environ. Stud.. 2023;32(4):3291-3298.
- [Google Scholar]
- Differential sensitivity of rat voltage-sensitive sodium channel isoforms to pyrazoline-type insecticides. Toxicol. Appl. Pharmacol.. 2006;214(2):209-217.
- [Google Scholar]
- Toxicity index, an improved method of comparing the relative toxicity of insecticides. J. Econ. Entomol.. 1950;43:45-53.
- [Google Scholar]







