Translate this page into:
Bioerosion and encrustation of the rocky shore dwellers along the Arabian Gulf, Northeast Saudi Arabia
⁎Corresponding author. asmohamed@ksu.edu.sa (Abdelbaset S. El-Sorogy),
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The coastal area between Al-Khafji and Al-Jubail, Arabian Gulf, Saudi Arabia is characterized by natural and artificial rocky shores, which inhabited by intensive dwellers. The present work aimed to shed light on the taxonomy, distribution, and environmental factors affecting the abundance of the invertebrate borers and encrusters in the study area. A total of 614 specimens of bivalve, gastropod, coral, and lithified rocky shores were collected from 13 sites. Eighteen ichnospecies of 8 ichnogenera were identified and illustrated. These traces were produced by clionid sponges (31.75%), endolithic bivalves (26.19%), naticid gastropods (24.60%), polychaete annelids (15.08%), acrothoracican barnacles (1.85%), and vermitid gastropods (0.53). The rocky shore dwellers act as hard substrate for colonization by serpulids, barnacles, bryozoans, and other cemented invertebrates. Most of the thick invertebrates and lithified rocky grounds were bioeroded by endolithic bivalves, clionaid sponges, polychaete annelids, and acrothoracican barnacles, while the thin walled invertebrate dwellers were bioeroded by naticid gastropods and clionaid sponges. Barnacles, serpulid worms and some molluscs were intensively covered the rocky shore blocks and solid rubbish in intertidal area facing wave action to comb microscopic food from the water.
Keywords
Bioerosion
Encrustation
Rocky shore dwellers
Arabian Gulf
Saudi Arabia
1 Introduction
Boring, rasping, drilling, and scraping activities were the most common bioerosion structures resulted by different organisms on hard substrates using mechanical and/or chemical processes (Buatois et al., 2002; Santos and Mayoral, 2008; Richiano et al., 2012). Lithified rocky shores and associated skeletons of the invertebrate dwellers can be used by several other organisms for shelter and/or food (Gibert et al., 2004; El-Sorogy, 2008; Demircan et al., 2023). Some invertebrates, such as bryozoans, serpulid worm tubes, balanids, corals, algae and ostreids use hard skeletons of the invertebrate rocky shore dwellers as substrates for colonization. Other organisms can destruct these substrates through bioerosion (Taylor and Wilson, 2002; Lopes, 2012). Therefore, bioerosion weak the substrates and can convert carbonate rocks into fine chips and consequently accelerates the mechanical destruction by waves and currents (Bromley, 1970; Lopes, 2012).
The bioerosion structures produced by macro- and microorganisms can be tunnels, chambers or holes, which represent the domichnia or praedichnia as ethological categories. Moreover, these structures may record rasping, scraping, and gnawing. Therefore, they can be ascribed to represent the paschichnia (Warme, 1975; Gibert et al., 2004; Verde, 2007; Lopes, 2012). The detection of the paleoecological and paleoenvironmental conditions prevailing in marine environments throughout the geological history were the application importance of studying of bioerosion (Bromley, 1970; Lopes, 2012). Works concerned with taphonomic signatures in the Aabian Gulf are very scarce (e.g. El-Gendy et al., 2015; El-Sorogy et al., 2018, 2020; Demircan et al., 2021). These articles dealt with the bioerosion and encrustation on the intertidal bivalves, gastropods, and corals. Although the coastal zone along the Arabian Gulf have been subjected to a lot of works up on environmental studies (e.g. El-Sorogy et al., 2016, 2019; Youssef et al., 2016; Al-Kahtany et al., 2015, 2018; Alharbi et al., 2017; Alharbi and El-Sorogy, 2019). Therefore, the objectives of the present work are: (i) to document the distribution of the invertebrate rocky shore dweller along the coastal area between Al-Jubail and Al-Khafji, (ii) to identify the bioeroders and encrusters affecting these rocky shore dwellers, and (iii) to analyze the bioerosive structures from an ethological standpoint.
2 Material and methods
The study area is located between Al-Jubail and Al-Khafji, along the Arabia Gulf coast, Saudi Arabia, between latitudes N27°00́84″ – N28°18́26″ and longitudes E49°40́00″ – E48°31́37″ (Fig. 1). The coastline is sandy-dominated shore, with artificial and natural rocky in parts. The sandy shores are characterized by fine to coarse, moderately sorted sand grains, with biogenic concentrations of gastropods, bivalves, foraminifers, and seagrass in parts. The natural and artificial rocky shores are inhabited with different invertebrate dwellers (Fig. 2). A total of 614 bivalves, gastropods, corals, and lithified rocky shore specimens were collected from 13 rocky shore sited in the area between Al-Jubail and Al-Khafji (Fig. 1). The 383 specimens which showed bioerosion and/or encrustation signatures were washed, examined and identified and differential distributions on the skeletal surfaces were evaluated. All examined specimens are housed in the Museum of the Department of Geology and Geophysics, College of Science, King Saud University, Saudi Arabia.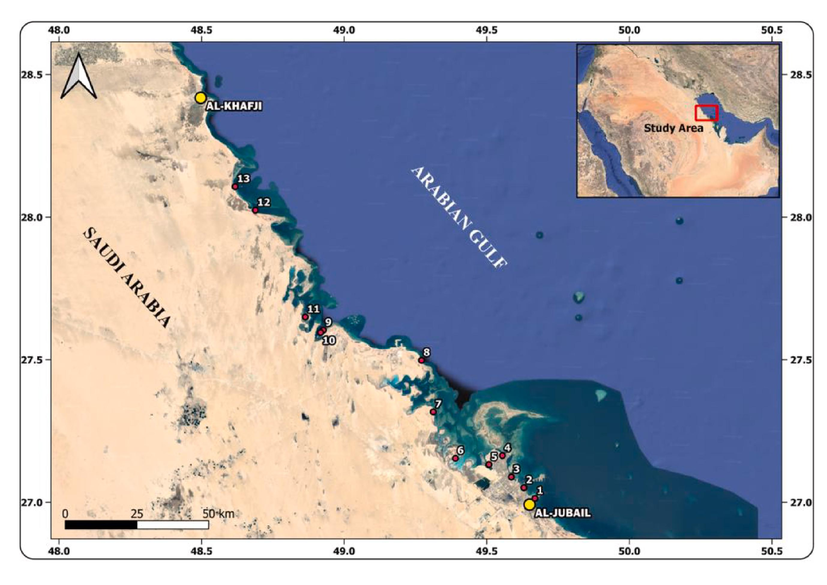
Location map of the study area and sample stations.
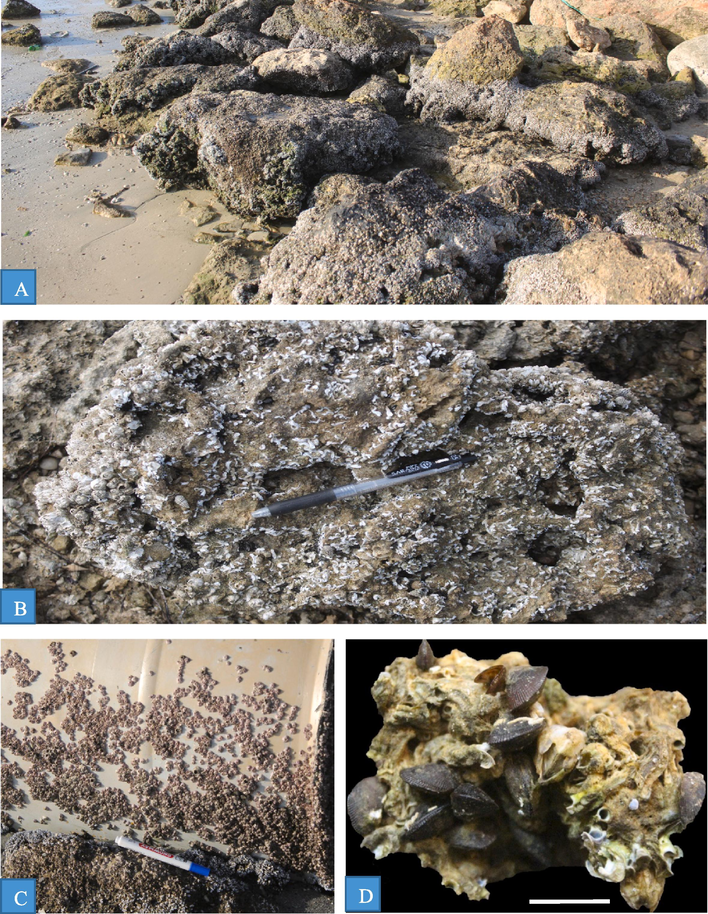
Invertebrate dwellers from the Arabian Gulf coast. A. Rocky shore blocks covered by serpulid worm tubes and blaniids in intertidal area facing sea water, site 2; B. Rocky shore block covered by serpulid worm tubes, balanids and gastropods, site 5; C. Encrusting balanids up on a barrel thrown in the intertidal area, site 2; D. Fixed byssate bivalves (Brachidontes) on intensive serpulids, site 2.
3 Results
3.1 Bioerosion
The recorded borings on the rocky shore dwellers were produced by endolithic bivalves (Gastrochaenolites), clionaid sponges (Entobia), polychaete annelids (Caulostrepsis, Maeandropolydora, and Trypanites), and naticid gastropods (Oichnus), acrothoracican barnacles (Rogerella), and vermitid gastropods (Renichnus). Table 1 illustrated the recorded ichnospecies and their trace makers, ethological category, numbers of the bioeroded seashells, and abundance throughout the studied 13 sites. The following is a summary of the identification, and description of these groups.
Ichnotaxa
Tracemaker
Ethological category
Studied localities
Total bioeroded seashells
1
2
3
4
5
6
7
8
9
10
11
12
13
Gastrochaenolites torpedo
Endolithic bivalves
Domichnia
11
6
3
3
2
25
G. lapidicus
Endolithic bivalves
Domichnia
10
4
5
3
2
2
26
G. ornatus
Endolithic bivalves
Domichnia
4
4
G. dijugus
Endolithic bivalves
Domichnia
5
5
Gastrochaenolites isp.
Endolithic bivalves
Domichnia
2
8
6
4
4
5
5
2
2
4
2
44
Maeandropolydora sulcans
Polychaete annelids
Domichnia
7
2
2
11
Caulostrepsis taeniola
Polychaete annelids
Domichnia
6
2
8
2
2
20
Trypanites isp.
Polychaete annelids
Domichnia
2
9
4
4
7
26
Entobia geometrica
Clionid sponge
Domichnia
8
5
3
5
3
5
29
E. cretacea
Clionid sponge
Domichnia
6
4
2
2
3
17
E. ovula
Clionid sponge
Domichnia
6
3
2
3
14
Entobia isp.
Clionid sponge
Domichnia
3
4
5
3
2
6
5
5
5
2
10
10
60
Oichnus paraboloides
Carnivorous gastropods
Praedichnia
7
12
2
6
2
2
31
O. simplex
Carnivorous gastropods
Praedichnia
4
6
4
6
2
22
O. ovalis
Carnivorous gastropods
Praedichnia
6
8
1
15
Oichnus isp.
Carnivorous gastropods
Praedichnia
5
5
2
3
3
2
2
3
25
Rogerella isp.
Acorn barnacles
Praedichnia
7
7
Renichnus isp.
Vermetid gastropods
Praedichnia
2
2
Total
22
97
41
11
29
17
30
20
19
24
38
30
383
3.1.1 Clionaid sponge borings
Traces of clionaid sponges are ethologically classified as domichnia, and are represented by a series of small swollen chambers connected by thin canals to other chambers and multiple apertures, 0.5 and 2 mm in diameter (Wilson and Palmer, 1992). Such borings cause dramatic increase in rate of bioerosion and sediment production, as well as destroy countless carbonate hardgrounds. This group of traces includes the ichnogenus Entobia Bronn, 1837 (ichnofamily Entobiaidae Wisshak et al., 2019), which is strongly developed on external and internal surfaces of bivalve and gastropod shells, as well as some corals and hard grounds. In the study area, clionaid sponges accounted 31.75 % of total bioerosion traces, and represented by Entobia geometrica Bromley and D’Alessandro, 1984, E. ovula Bromley and D’Alessandro, 1984, and they are E. cretacea Portlock, 1843, and Entobia isp. (Table 1). Twenty-nine specimens of Entobia geometrica were recorded on the external and internal surfaces of Plicatula, Glycymeris, Pinctada, Barbatia, Conus, and some recorded on the lithified rocky shore grounds. E. geometrica shows networks of chambers interconnected by irregularly distributed cylindrical galleries (Fig. 3A-C, F-H). The chambers with circular apertures of different sizes, 2.5–3 mm in diameter for larger apertures, and 1–2 mm in diameter for the smaller ones (Demircan et al., 2021). Borings of E. geometrica are closely similar in morphology and size to borings made by Cliona celata Grant (Bromley and D’Alessandro, 1984).
Clionaid, polychaete, and vermetid borings. A-I. Entobia geometrica (1), sites 1, 2, 6, and 13; Entobia cretacea (2), site 2; Entobia ovula (3), sites 2 and10; Entobia isp (4), sites 2, 4, and 6; Caulostrepsis taeniola (5), site 10; Renichnus isp (6), site 4. Traces developed on the external and some internal surfaces of Plicatula, Protapes, Fulvia, and Amiantis. Scale bar = 10 mm, except of C, E, H, and I = 2 mm.
Seventeen specimens of E. cretacea are developed on the external surfaces of Protapes (Fig. 3D, G). The boring is branched, resembling a well-developed camerate or string-of-beads form. E. cretacea differs from E. geometrica in that the apertures of the former were smaller, as well as its chambers were connected by single intercameral canals. Fourteen specimens of E. ovula have been observed on external surfaces of Protapes and Fulvia (Fig. 3D, G, 5I). Borings in four stages graduated from narrow and branched tunnels to curved rows of elongated, and oval chambers. E. ovula was previously recorded from the Early Eocene from India, among others (Gurav and Kulkarni, 2018). Sixty specimens of Entobia isp. were reported as networks of linear chambers, with apertures 0.3–1.5 mm in diameter on the surfaces of Glycymeris, Conus, and Lopha (Fig. 3H, I, 4A, 6E, 7A, C).
3.1.2 Endolithic bivalve borings
The borings of endolithic bivalves belong to Gastrochaenolites Leymerie, 1842, which is produced mostly by the lithophagids, gastrochaenids, and pholadids as a domichnion. In the study area, the bivalve tracemakers are preserved within the borings in some cases (Fig. 4). The borings are usually oriented perpendicular to the thick substrate of coral and bivalve skeletons, and hard rocky grounds. They are less than 1.0 cm to more than 10 cm deep and up to 5 cm wide. In some hard shore rocky grounds, Gastrochaenolites may intersect and subsequently be bored with clionaid sponges, encrusted and nested by a variety of organisms inside, such as serpulids and bryozoans. In the study area four ichnospecies belongs Gastrochaenolites (ichnofamily Gastrochaenolitidae Wisshak et al., 2019) were recorded and accounted 26.19 % of the total bioerosion traces. They include G. lapidicus, G. torpedo, G. dijugus, G. ornatus Kelly and Bromley, 1984, and Gastrochaenolites isp. (Table 1).
Bivalve, gastropod, clionaid, and polychaete borings. A-H. Gastrochaenolites torpedo (1), site 2; G. lapidicus (2), sites 2; G. ornatus (3), site 2; and Oichnus isp. (4), sites 2, 13; Entobia isp. (5), sites 2, 5, 13; Trypanites isp. (6), site 2. Traces developed on the external shell surfaces of Protapes, Pinctada, Conus, Chama, corals, and lithified rocky shore grounds. Some of the trace makers still present within the drill holes. Scale bar = 10 mm, except of G = 2 mm.
Twenty-four Gastrochaenolites torpedo were recorded on the external surfaces of Protapes, Pinctada, Chama, thick corals, hard-grounds (Fig. 4B, D, H). These borings are smooth with elongated chamber, 0.5–1.4 cm in diameter and up to 1.5 cm in depth. Presence of G. torpedo perpendicular to steep substrates might be to avoid sedimentary deposition (Bromley, 2004; Uchman et al., 2017; Demircan et al., 2021).
Twenty-two specimens of Gastrochaenolites lapidicus were recorded on the external surfaces of Pictada, Acrosterigma, thick corals, lithified hard grounds (Fig. 4C, F, 5 J, 6G). G. lapidicus is produced recently by the bivalves Lithophaga and Hiatella (Kelly and Bromely, 1984; Uchman et al., 2002). The borings are smooth, with circular to ovate chambers and circular or elliptical neck, 6–13 mm wide, and 7–20 mm long. Five specimens of G. dijugus are oriented normal to the shell surfaces of Acrosterigma, Fulvia, and Glycymeris (Fig. 6A-C). The borings are circular to subcircular in transverse section, 2.5–4 mm in diameter. Gastrochaenolites ornatus is recorded in site 2 (4 borings) in the form of ovoid to elongate chambers 10–35 mm in length and 5–12 mm in width, normal to the shell surface and ornamented with concentric lines. 44 specimens of Gastrochaenolites isp. occur as small, subcircular borings without distinct neck, 4–9 mm in diameter, and up to 7 mm deep on lithified rocky grounds, and surfaces of Conus, Plicatula, Chama, Pinctada, Lopha, and Barbatia (Fig. 4A, D-F, 6G, 7C).
3.1.3 Naticid gastropod borings
Praedichnial borings of carnivore gastropods belong to the ichnogenus Oichnus Bromley, 1981 of the ichnofamily Oichnidae Wisshak et al., 2019. Oichnus accounts 24.60 % of the total bioerosion in the form of small circular to subcircular and oval borings normal to the external surfaces of the mollusc shells. Most of the traces penetrate the substrate while some ones terminate within the substrate as a shallow pit (Fig. 5D, F), indicating a failed drill attempt (Chattopadhyay and Dutta, 2013). Oichnus paraboloides Bromley, 1981, O. simplex Bromley, 1981, Oichnus ovalis Bromley, 1993, and Oichnus isp. were identified in the study area (Table 1).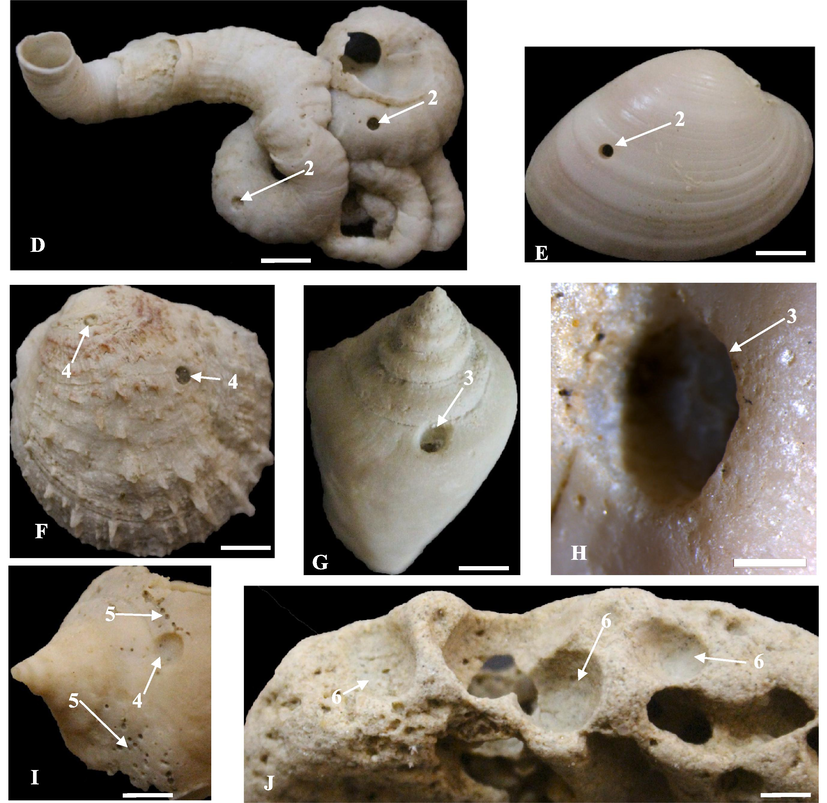
Gastropod, clionaid, and bivalve borings. A-J. Oichnus paraboloides (1), site 2; O. simplex (2) sites 2, 6, and 7; Oichnus ovalis (3), site 10; Oichnus isp. (4), site 12; Entobia ovula (5), site 13, Gastrochaenolites lapidicus (6), site 2. Traces developed on the surfaces of Callista, Spondylus, Barbatia, Conus and serpulid worm tubes. Scale bar = 10 mm, except of C and H = 2 mm.
Thirty-one borings of O. paraboloides were developed on the surfaces of Callista, Barbatia, Lopha, and Tapes shells (Fig. 5A-C). Borings are circular, paraboloid, normal to the surface in most cases, 1.4–2.8 mm in diameter. In thick shells, some borings terminate inside the shell. Twenty-two specimens of O. simplex were developed on the bivalve and gastropod shells (Barbatia, Fulvia, Circe, Spondylus, Bulla, and Conus), and serpulid worm tubes (Fig. 5D, E). Borings showed circular to subcircular shape, 1.7–2.4 mm in diameter, more or less perpendicular to the substrate. Fifteen specimens of O. ovalis developed on the gastropod Conus (Fig. 5G, H). They show oval shape, not fully penetrate the shell in most cases. Twenty-five traces of Oichnus isp. were found on shells of the bivalve Barbatia and the gastropod Conus (Fig. 4B, 5F, I). Borings are circular to slightly ovoid in shape, normal to the shell walls in most cases, 1.6–2.4 mm in diameter.
3.1.4 Polychaete annelid borings
Borings of polychaete annelids account 15.08 % of the total bioerosion traces and they are represented in the study area by Caulostrepsis taeniola Clarke, 1908, Maeandropolydora sulcans Voigt, 1965, and Trypanites isp. (Table 1). They belong to the ichnofamily Osteichnidae Hopner and Bertling, 2017. C. taeniola is a domichnion produced by spionid polychaetes traces (Bromley, 2004). Twenty specimens were recorded on the mollusc shell of Acrosterigma, Fulvia, Glycymeris, and Conus (Fig. 3G, 6E, F, I). C. taeniola consists of smooth a U-shape galleries and a central axial depression. Boring galleries range from straight to curved, 2–8 mm in length and 0.25–1.5 mm in width, with aperture 1 mm in diameter.
Eleven borings of M. sulcans are well-developed on the external surface of the gastropod Conus shell (Fig. 6D, H). They are shallow, long and sinuous borings, 0.2 mm in diameter, 10 mm long and 1–2.3 mm wide. Twenty-six Trypanites are normal to the substrates of Plagiocardium, Glycymeris, Chama, and Acrosterigma, thick coral and echinoid fragments, and limestone rocky grounds (Fig. 4C, D, F). They are narrow, cylindrical, and unbranched borings, 1.2–2.8 mm in diameter. Ethologically, Trypanites is classified as a domichnion, which trace maker uses an acid to dissolve the carbonate substrates (Taylor and Wilson, 2003).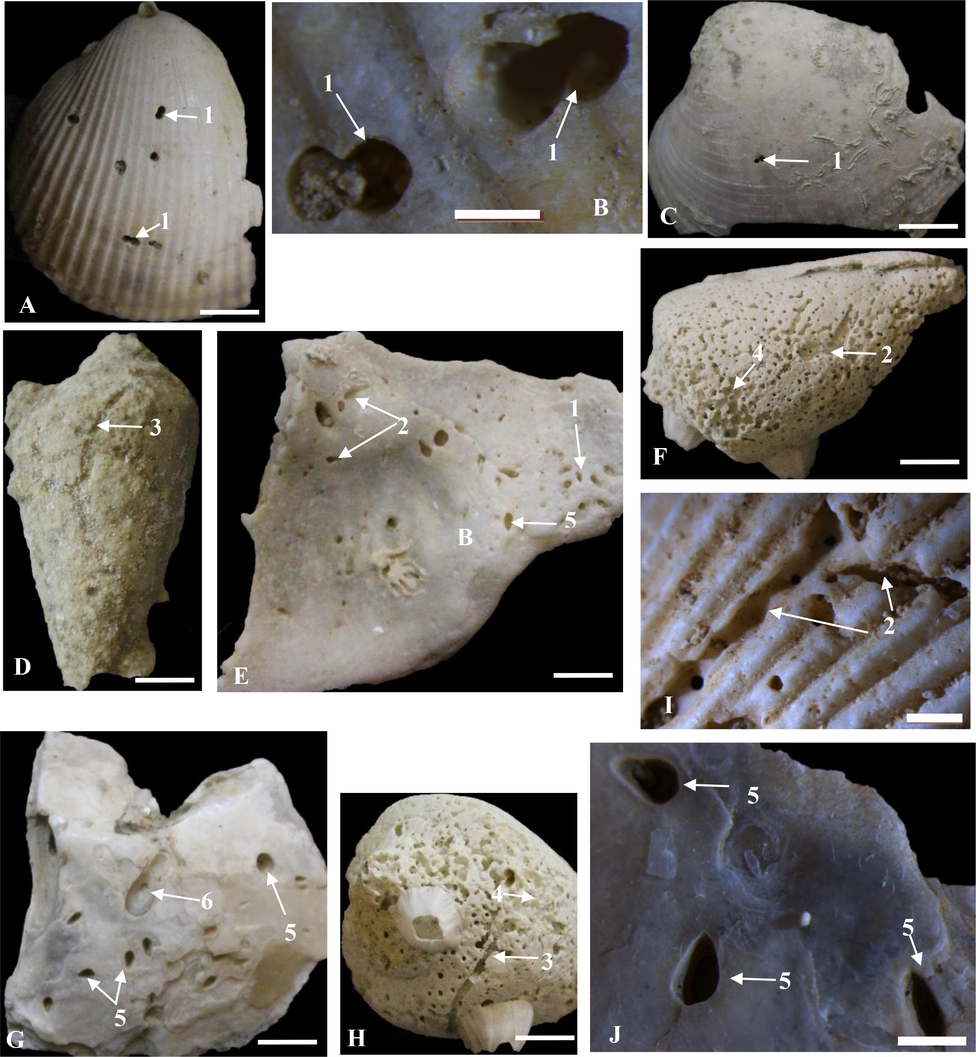
Bivalve, polychaete, barnacle, and clionaid borings. A-J. Gastrochaenolites dijugus (1), site 5, Caulostrepsis taeniola (2), sites 10, 13; Maeandropolydora sulcans (3), sites 2, 10, 13; Entobia geometrica (4), site 12; Rogerella isp., site 5 (5); Gastrochaenolites isp. (6), site 5. Traces developed on the mollusc surfaces of Acrosterigma, Fulvia, Glycymeris, Plagiocardium, Glycymeris, Chama, Pinctada and Conus. Scale bar = 10 mm, except of B, I, and J = 2 mm.
3.1.5 Acrothoracican barnacle borings
Borings of acrothoracican barnacles are represented by ichnogenus Rogerella de Saint-Seine, 1951 (ichnofamily Echinoidea Wisshak et al., 2019). Seven traces developed on the internal shell surfaces of the bivalves Pinctada and Chama (Table 1, Fig. 6E, G, J). Rogerella shows rounded to oval-like depressions with slit-shaped apertures, 0.5–1.0 mm in width, 0.7–2.2 mm in length and 1.2–2.1 mm in depth.
3.1.6 Vermetid gastropod borings
Borings of vermetid gastropods belong to ichnogenus Renichnus Mayoral, 1987 and are repeated by Renichnus isp. (Fig. 3I). Two traces developed on the internal surface of the bivalve Amiantis. Renichnus isp. is a shallow half-moon, kidney-shaped or annular depressions. Ethologically, Renichnus is classified as praedichnia (El-Sorogy et al. 2022).
3.2 Encrustations
The invertebrate rocky shore dwellers and the lithified hardgrounds act as hard substrates for settlement by other invertebrates, including serpulids, bryozoans, barnacles, and cemented bivalves (Figs. 2, 7). Serpulids are tube-dwelling polychaetes, circular to sub-circular in cross-sections, always cryptic in habit, and tend to heavily encrust overhangs and the sides of crevices in rocky grounds (Wilson and Palmer, 1992; El-Hedeny, 2005). The serpulids are the most common encrusters throughout the studied sites. These worm tubes encrust the external and internal surfaces of the molluscs Acrosterigma, Donax, Chama, Glycymeris, Lunella, Bulla, Hexaplex, Conus, as well as coral skeletons and lithified rocky ground (Fig. 2 A, B, D, 4E, 5D, 6C, E, 7D, F, G). Barnacles were the second abundant encrusters. In the studied sites, they are solitary or form aggregates on the lithified hard-grounds, external and internal surfaces of bivalves (Acrosterigma, Amiantis, Glycymeris, Hexaplex, Circe), and the gastropod Conus shells (Fig. 2A, C, 6F, H, 7B). Bryozoans are the least abundant in the study area, encrusting the internal smooth surface and the external surface of Glycymeris, Pinctada (Fig. 7E, F). Bryozoan colonies are sheet-like representatives of Holloporella, Membranipora, Celleporaria, and Watersipora spp. Many bivalves are attached to the lithified rocky shore hardgrounds, such as Septifer and Modiolus fixed by byssus. Moreover, many of small bivalves, such as Plicatula, Brachidontes, Planaxis, Chama, and Pinctada were cemented to other large and thick bivalve shells (Fig. 2D, 7A, C). However, the settlement of these encrusters on the internal surfaces of many mullusc shells indicates a postmortem colonization. On the other hand, the settlement up on the external surfaces implying mostly syn-vivo colonization (El-Sorogy et al., 2020).
Abundant encrusters from the Arabian Gulf coast. A-G. Encrustation of serpulids (1), sites 2, 4, and 12; cheilostomate bryozoans (blue circle), site 5 and 7; barnacles (2), site 3; and cemented bivalves (3), site 7. All encrusted up on hard skeletons of the invertebrate rocky shore dwellers. Scale bar = 10 mm, except of E and F = 2 mm. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
4 Discussion
The lithified substrate of the rocky shores constitutes a high-energy, stable and well-oxygenated environments characterized by high productivity suitable for suspension feeders (Wilson and Palmer, 1992; El-Sorogy et al., 2019). Organisms inhabited rocky substrates are subjected to physical and biological stress factors. Physical factors, such as wave action, scouring and covering by sand, especially in the presence of sandy beaches, and exposure, to ultraviolet light, heat, desiccation, rain, runoff of fresh water from the land during low tides. The competition for space and food, predation, grazing, and scavenging are the biological factors. Cementation and infaunalization by boring or nestling are adaptations for dealing with the rigors of hard substrate life.
Degree of articulation and fragmentation is a good indicator of relative exposure time, or energy of the depositional environment (El-Sorogy, 2015). More than 95 % of the collected bivalves in the current study have been disarticulated. Articulation occurred at rocky shore areas, particularly in specimens that are physically stuck within the spaces between rock blocks and to species that have extremely thick ligaments or dentitions resistant to disarticulation. On the other hand, disarticulated valves occurred at areas with considerable water energy. In the study area, broken seashells might be found close to abrasion and bioerosion, which could lessen the density and thickness of the shells and so make breaking easier. In such an environment, predators and other species are likely fragmentation agents as well.
383 of 614 specimens belong to bivalves, gastropods, corals, and lithified rocky shore were affected by different types of bioeroders. The total materials/bioeroded materials were 273/183 for gastropods, 211/144 for bivalves, 87/41 for corals, and lithified rocky shore specimens 43/15. The abundance of the reported bioerosion traces in the studied rocky shore were in the following order: clionaid sponge (31.75 %) > endolithic bivalve (26.19 %) > naticid gastropod (24.60 %) > polychaete annelid (15.08 %) > acrothoracican barnacle (1.85 %) > vermitid gastropod (0.53 %) traces. All bioerosion traces include dwelling traces (domichnia), except for the naticid gastropods which produce traces of predation (praedichnia) (Odumodu and Okon, 2016). Most of the rocky shore dwellers were bioeroded by clionid sponges. Some parts of shells, especially the external surfaces were intensively bioeroded with Entobia geometrica and Entobia isp. (Fig. 3A, B, 4A, 6F, H), indicating that boring occurred during their life-time (El-Sorogy, 2015). However, most shells showed a few scattered borings.
The rocky shore, mollusc shells, and coral skeletons were favorable substrates for the boring bivalves, polychaetes, and acrothoracican barnacles. They are bioeroded by representatives of the genera Gastrochaenolites, Maeandropolydora, Trypanites, Caulostrepsis, and Rogerella. Moreover, these substrates were colonized by many filter-feeding epifaunal serpulid, barnacles, bryozoans, and other small bivalves. Oichnus, on the other hand, preferred thinner shells. It was produced mostly during the lifetime of molluscs, and likely have caused their death (Bromley, 1981; Hauser et al., 2008). Some shells show more than one Oichnus, either as a shallow depression or a pit within the shell (Fig. 5D, F), which may be a record of unsuccessful attempts of predation (Hauser et al., 2008; El-Sorogy et al., 2021). In order to comb microscopic food from the water, barnacles, serpulid worms, and some molluscs, such as Brachidontes and Planaxis, intensively incrusted the rocky shore blocks and solid rubble in intertidal area facing wave action.
5 Conclusions
-
Eighteen ichnospecies produced by clionaid sponges, endolithic bivalves, naticid gastropods, polychaete annelids, acrothoracican barnacles and, vermetid gastropods were identified and illustrated in the coastal area between Al-Khafji and Al-Jubail, Arabian Gulf, Saudi Arabia. Traces of clionaid sponges, polychaete annelids, endolithic bivalves were ethologically classified as domichnia, while borings produced by carnivore gastropods are ethologically classified as praedichnia.
-
In a descending order of abundance, the identified traces include the following: Entobia (E. geometrica, E. ovula E. cretacea, and Entobia isp.), Gastrochaenolites (G. lapidicus, G. torpedo, G. dijugus, G. ornatus, and Gastrochaenolites isp.), Oichnus (O. paraboloides, O. simplex, O. ovalis, and Oichnus isp.), Caulostrepsis taeniola, Maeandropolydora sulcans, Trypanites isp., Rogerella isp., and Renichnus isp.
-
The larger and thicker rocky shore dwellers in intertidal area offered a favorable substrate for many filter-feeding epifaunal serpulids, barnacles, bryozoans, and other bivalves, which were favorable substrates for the boring bivalves, polychaetes, and acrothoracican barnacles. On the other hand, carnivore naticid gastropods preferred thinner shells of bivalves and gastropods.
Acknowledgments
The authors extend their appreciation to Researchers Supporting Project number RSP2024R425, King Saud University, Riyadh, Saudi Arabia. Moreover, the authors thank the anonymous reviewers for their valuable suggestions and constructive comments.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Spatial distribution and metal contamination in the coastal sediments of Al-Khafji area, Arabian Gulf, Saudi Arabia. Environ. Monit. Assess.. 2017;189:634.
- [CrossRef] [Google Scholar]
- Assessment of seawater pollution of the Al-Khafji coastal area, Arabian Gulf, Saudi Arabia. Environmental Monitoring and Assessment. 2019;191:383.
- [CrossRef] [Google Scholar]
- Heavy metals in mangrove sediments of the central Arabian Gulf shoreline, Saudi Arabia. Arab. J. Geosci.. 2018;11:155.
- [Google Scholar]
- Geochemical and foraminiferal analyses of the bottom sediments of Dammam coast, Arabian Gulf, Saudi Arabia. Arab. J. Geosci.. 2015;8:11121-11133.
- [Google Scholar]
- Borings as trace fossils and Entobia cretacea Prtlock as an example. In: Crimes T.P., Harper J.C., eds. Trace Fossils. Seel House Press; 1970. p. :49-90.
- [Google Scholar]
- Bromley, R.G., 2004. A stratigraphy of marine bioerosion. In: McIlroy, D. (Ed.), The Application of Ichnology to Palaeoenvironmental and Stratigraphic Analysis, Geological Society, London, Special Publications, pp. 455–481.
- Trazas fósiles, señales de comportamiento en el registro estratigráfi. Edición Especial del Museo Egidio Feruglio: Trelew; 2002. p. :382.
- Prey selection by drilling predators: a case study from Miocene of Kutch. India. Palaeogeogr. Palaeoclimatol. Palaeoecol.. 2013;374:187-196.
- [Google Scholar]
- The beginnings of dependent life. New York State Museum Bulletin. 1908;121:146-196.
- [Google Scholar]
- Bioerosional structures from the Late Pleistocene coral reef, Red Sea coast, northwest Saudi Arabia. Turk. J. Earth Sci.. 2021;30:22-37.
- [Google Scholar]
- Taphonomic signatures on the pearl oyster Pinctada from Arabian Gulf, Saudi Arabia. Journal of King Saud University – Science. 2023;35:102870
- [CrossRef] [Google Scholar]
- Taphonomic signatures on some intertidal molluscan shells from Tarut Bay (Arabian Gulf, Saudi Arabia) Pakistan J. Zool.. 2015;47:125-132.
- [Google Scholar]
- Taphonomy and paleoecology of the Middle Miocene oysters from Wadi Sudr, Gulf of Suez. Egypt. Rev. Paléobiol.. 2005;24:719-733.
- [Google Scholar]
- Contributions to the Pleistocene coral reefs of the Red Sea coast. Egypt. Arab. Gulf. Jour. Sci. Res.. 2008;26(1/2):63-85.
- [Google Scholar]
- Taphonomic processes of some intertidal gastropod and bivalve shells from northern Red Sea coast, Egypt. Pakistan J. Zool.. 2015;47(5):1287-1296.
- [Google Scholar]
- Distribution of intertidal molluscs along Tarut Island coast, Arabian Gulf, Saudi Arabia. Pakistan J. Zool.. 2016;48(3):611-623.
- [Google Scholar]
- Bioerosion structures in high-salinity marine environments: A case study from the Al–Khafji coastline, Saudi Arabia. Estuar. Coast. Shelf Sci.. 2018;204:264-272.
- [Google Scholar]
- Molluscan assemblage as pollution indicators in Al-Khobar coastal plain, Arabian Gulf, Saudi Arabia. J. Afr. Earth Sci.. 2019;158:103564
- [CrossRef] [Google Scholar]
- Gastrochaenolites ichnofacies from intertidal seashells, Al-Khobar coastline, Saudi Arabia. J. Afr. Earth Sci.. 2020;171:103943
- [Google Scholar]
- Taphonomic signatures on modern molluscs and corals from Red Sea coast, southern Saudi Arabia. Palaeoworld 2021
- [CrossRef] [Google Scholar]
- Gibert, J.M.de, Domènech, R., Martinell, J., 2004. An ethological framework for animal bioerosion trace fossils upon mineral substrates with proposal of a new class, fixichnia. Lethaia, 37, 429–437.
- Natural casts of Early Eocene Entobia from the Kachchh Basin. India. Ichnos. 2018;25(4):261-268.
- [Google Scholar]
- Taphonomic signatures on modern Caribbean bivalve shells as indicators of environmental conditions (Belize, central America) PALAIOS. 2008;23:586-600.
- [Google Scholar]
- Lopes, R.P., 2012. Bioerosion and bioincrustation in body fossils from the coastal plain of Rio Grande do SulState, Southern Brazil. In: Ichnology of Latin America. Netto, RG, Carmona, NB, Tognoli, FM (Editors). Sociedade Brasileira de Paleontologia, 179–194.
- The paleoenvironmental significance of trace fossils from the Paleocene Imo Formation, South-eastern Nigeria. Journal of Natural Sciences Research. 2016;6(22):2224-3186.
- [Google Scholar]
- Richiano, S., Aguirre, M., Farinati, E., 2012. Bioerosion structures in Quaternary marine molluscs from Argentina. In: Ichnology of Latin America. Netto; Carmona and Tognoli (Editors). Sociedade Brasileira de Paleontologia. pp.159–177.
- Bioerosion versus colonization on Bivalvia: A case study from the upper Miocene of Cacela (southeast Portugal) Geobios. 2008;41:43-59.
- [Google Scholar]
- A new terminology for marine organisms inhabiting hard substrates. PALAIOS. 2002;17:522-525.
- [Google Scholar]
- Palaeoecology and evolution of marine hard substrate communities. Earth Sci. Rev.. 2003;62:1-103.
- [Google Scholar]
- Relative sea-level changes recorded in borings from a Miocene rocky shore of the Mut Basin from a Miocene rocky shore of the Mut Basin, southern Turkey. Ann. Soc. Geol. Pol.. 2002;72:263-270.
- [Google Scholar]
- Macroborings, their tracemakers and nestlers in clasts of a fan delta: The Savignone Conglomerate (Lower Oligocene), Northern Apennines, Italy. Neues Jahrb. Geol. Palaontol. Abh.. 2017;283(1):35-51.
- [Google Scholar]
- Verde, M., 2007. Bioerosão. In: Carvalho, I.S., Fernandes, A.C.S. (eds.) Icnologia. Sociedade Brasileira de Geologia, Série Textos, 3, 108–117.
- Über parasitische Polychaeten in Kreide-Austern sowie einige andere in Muschelschalen bohrende Würmer. Paläontol. Z.. 1965;39:193-211.
- [Google Scholar]
- Borings as trace fossils, and the processes of marine bioerosion. In: Frey R.W., ed. The Study of Trace Fossils. Springer-Verlag; 1975. p. :181-226.
- [Google Scholar]
- Hargrounds and hardground faunas. Aberystwyth: University of Wales. Institute of Earth studies Publications; 1992. p. :1-131.
- Distribution of mercury in molluscs, seawaters and coastal sediments of Tarut Island, Arabian Gulf, Saudi Arabia. J. Afr. Earth Sci.. 2016;124:365-370.
- [Google Scholar]







