Translate this page into:
Assessment of toxicity, anti-quorum sensing and anti-biofilm production effects of Hypericum triquetrifolium Turra extract on multi-drug resistant Acinetobacter baumannii
⁎Corresponding author.
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University. Production and hosting by Elsevier.
Abstract
Acinetobacter baumannii is a multidrug-resistant and an invasive bacterium and a significant contributor to nosocomial infections in the present healthcare system. It’s striking capability to survive in hospital settings on medical equipments is due to its capability of quorum sensing and biofilm formation. New medications and formulations to combat this pathogen is the need of this hour. Hypericum triquetrifolium Turra, a less famous medicinal plant is known for its antibacterial characteristics. However, there is hardly any research articles or findings to back this claim against the Acinetobacter sp. Total of 50 bacterial isolates were collected. 98% of them were resistant to Ciprofloxacin. 4% showed strong biofilm production, while 40% and 56% of the isolates revealed weak and moderate bioflim production. Biochemical analysis revealed that the ethanolic leaves extract of H. triquetrifolium Turra contains potent phytochemical compounds. Toxicity assessment of the plant extract revealed that the extract was slightly toxic where LD50 was 4.95 g/Kg. Minimum inhibitory concetrations and Sub-minimum inhibitory concetrations of H. triquetrifolium Turra leaf extract against two strongest biofilm producers and ATCC strain of A. baumannii (19606) were estimated. MICs values were 10–15 mg/ml and SICs 5–10 mg/ml. Investigations revealed significant reductions in biofilm formation when isolates were treated with SICs of the extract. Transcription expression of quorum sensing genes, abaI/abaR, down regulated when treated with H. triquetrifolium extract. One of the Biofilm regulating genes among Bap/OmpA, were found to be sharply down regulated especially, Bap gene; however, OmpA gene didn’t show noticable change in the expression. We concluded that H. triquetrifolium Turra may have promising activity against A. baumannii in it’s quorum sensing and biofilm formation, thus it could be employed as potential as an antibacterial medication.
Keywords
Antibiotic
Downregulation
Gene expression
Hypericum
LD50
Erbil
1 Introduction
Antimicrobial resistance (AMR) remains a major problem around the world. Recently, more morbidity and mortality are attributed to pathogens resistant to multiple classes of antibiotics (Wingler et al., 2022). The bacteria Acinetobacter baumannii (A. baumannii) is widespread, showing up everywhere from the natural environment such as soil and food to medical settings especially in the intensive care unit (ICU) setting, and consequently became one of the primary cause for a wide variety of hospital-acquired infections (Medioli et al., 2022). A. baumannii infections have a mortality rate that is 2 to 3 times higher than that of infections caused by the strains which are the usual suspects (Shenkutie et al., 2020). A. baumannii possesses a variety of virulence factors for activities like desiccation resistance, biofilm production, motility, surface glycoconjugates, and systems for acquiring micronutrients (Zhu et al., 2022). Several studies have publicized that the quorum sensing (QS) system, mediated by the auto-inducing mechanism in order to maintain cell density, is associated with various virulence factors, signifying that QS may have a regulatory route in bacterial virulence (Zhong et al., 2021). Its biofilm-forming ability has been hypothesized to contribute to both its persistence and virulence in hospital settings. Biofilms can be produced by A. baumannii on biotic surfaces like epithelial cells as well as abiotic surfaces. This bacterium deploys a cell with cell communication mechanism universally named as quorum sensing to control its pathogenicity. Consequently, one of the prominent secondary metabolites which are controlled by quorum sensing process plays a substantial role in controlling the expression of specific genes and their capacity to adopt to the nature of their surroundings (Saipriya et al., 2020). Evidence suggested that the expression of virulent genes such as PNAG, pili – Type 1, the QS system, Omp CarO and multiple other functional metabolism of histidine involved with biofilm and antimicrobial resistance (Gedefie et al., 2021). Furthermore, phospholipase D production and OmpA are two of A. baumannii's important virulence factors that aid in the bacterium's ability to adhere to and invade epithelial cells. In particular, it has been revealed that the virulence factors Bap and OmpA contribute to epithelial cell invasion, while phospholipase D and lipid A are involved in epithelial cell adherence (Mea et al., 2021). Two proteins, AbaI and AbaR, make up A. baumannii's quorum sensing (QS) system. Most abundant of all the AHL signals is 3-hydroxy-C12-homoserine lactones and this encoded by the the gene known as abaI gene and is catalyzed by the autoinducer synthases (Tang et al., 2020). Moreover, differentiating an actual infection from a colonization is often complicated by the polymicrobial nature of A. baumannii infections beside Pan-drug resistant A. baumannii (PDRAB) is being continues to rise with limited therapeutic options (Karakonstantis et al., 2022). Several species of the genus Hypericum including Hypericum triquetrifolium Turra (H. triquetrifolium) which belong to the family Hypericaceae, have been found to contain bioactive secondary metabolites (Dall’Acqua et al., 2021). Extractions from Hypericum triquetrifolium Turra, a plant naturally habituated to the Mediterranean and Eastern Europe, is found to be utilized extensively in herbal medicine for their significant pharmacological properties, which have been empirically proven to be possessed ingredients of anti-helminthic, anti-depressant, anti-oxidant, anti-inflammatory, and anti-microbial activities (Asan, 2021). Also, has a wide range of other compounds, including flavonoids, xanthones, hyperforin derivatives, essential oils, and fatty acids (Rouis et al., 2013). This plant is one of the most widely used herbal remedies due to the dramatic rise in its consumer base throughout the world (Al-Snafi et al., 2018). Toxicity assay is an important process to stop the growth of pathogen microbes. These processes are supported by natural plant compounds that act similarly to synthetic pharmaceuticals but are either toxic-free or have lower toxicity (Rajalakshmi et al., 2014).
Despite its anti-quorum sensing and biofilm-preventing potential, H. triquetrifolium Turra has received surprisingly little attention from researchers. With the rising rate of AMR infections both in the community and in healthcare facilities, there is an urgent requirement for effective antibiotic therapies. In this study the primary aim is to ascertain and very carefully study the genes which are theroretically and practically said to be associated with the biofilm formation. Like for e.g abaI/abaR, ompA, and bap. Then ethanolic leaf extracts of H. triquetrifolium were tested for their toxicity, anti-quorum sensing, and anti-biofilm properties of H. triquetrifolium Turra against hospital acquired and lab grown clinical isolates of A. baumannii, and to advance alternate approaches to carefully examine the QS cascade of this bacterium to combat the spreading of difficult to treat infections.
2 Materials and methods
2.1 Plant collection
The leaves of Hypericum triquetrifolium Turra were collected during April 2021 from the Erbil in Kurdistan region of Iraq, Dr.Abdulah Shukur, a plant taxonomist at the biology department, Education College / Salahaddin university, was helping for the identification of this plant depend on the flora of Iraq (Townsend & Guest., 1980).
2.2 Plant maceration and crude extract preparation
The Hypericum triquetrifolium Turra leaves were gathered, cleaned, then thoroughly dried at room temperature at 40 to 50 °C until they gained constant weight. They were then slowly ground by grinders to get fine powder, then stored in a particular bottle. Dry powder of H. triquetrifolium Turra leaves (30.0 g) were extracted three times with stirring at regular intervals using 300 ml of 99.9% ethanol over 3 days at room temperature after being filtered through a double layer of muslin cloth and Whatman no.1 filter paper. The filtrate was then condensed by using a rotary evaporator at 45 °C under reduced pressure (Phong et al., 2022).
2.3 Specimens collection and samples sources
A total of fifty non-duplicate isolates of A. baumannii were collected from different specimens (blood, cerebrospinal fluid (CSF), pus, sputum, and wound swabs) from patients in different hospitals in Erbil city, Kurdistan Region, Iraq. The specimens were initially inoculated onto MacConkey agar medium (acumedia, Neogen, USA) and incubated for 24 h at 37 °C. Individual colonies were then identified as A. baumannii by various biochemical and conventional diagnostic tests as described previously (Tumeh and Evaluation, 2015). Bacterial isolates further identified by VITEK 2 automatic system (Biomerieux, France). The identified isolates were tested for their susceptibility to different antimicrobial agents (Colistin, Trimethoprime/Sulfamethoxazole, Amikacin, Cefazolin, Cefuroxime, Amoxilin/Clavulanic Acid, Ampicillin, Ceftazidime, Ampicilin/Sulbactum, Azithromycin, Cefepime, Cefoxitin, Ceftriaxone, Ciprofloxacin, gentamicin, Pipracillin, Imipenem, Levofloxacin, Meropenem, Piperacillin/Tazobactum, Tazobactum, Tetracycline,). Colonies were incoluated into sterile 1 ml tubes containing Tryptic Soy Broth supplemented with glycerol 30% and were stored at −70 °C after the necessary identification preocdure. An ATCC strain of A.baumannii (19606) was purchased from Medya Diagnostic center to be used as control throughout the study.
2.4 Toxicity assessment of H. Triquetrifolium Turra extract on rat and determination of lethal dose
This study was carried out at Salahaddin University, Education College, Biology Department. The female rats (Rattu norvegicus), weighed between (190–210) g. They were housed in a conventional environment with a photoperiod of 12 h of light and 12 h of darkness, temperature of 27 ± 2 °C, standard feeds, and free access to tap water. All the animals were kept in conventional plastic cages, and randomly separated into different groups (n = 5). The first group (control) only received DMSO, while the other groups were given a single dose of an ethanol extract of H. triquetrifolium Turra leaves at concentrations of 0.5, 1, 2, 4, 4.5, and 5 g/kg. The amount of extract solution that can be given orally depends on the size of the animals and should not be more than 1 ml per 100 g of their body weight. Animals were given the extract solution (16–18) hours after being deprived of food but not water and rat mortality that was recorded after 24 h was used to get the LD50 value (Taziebou et al., 2007).
2.5 Phytochemical qualitative test for H. Triquetrifolium Turra leaves
The crude extract of plant leaves was completely dissolved in its own basic solvent (mg/ml), to obtain a stock solution. The stock solutions were used for phytochemical analysis according to the standard procedure. The stock solutions were used for phytochemical test in accordance with the following procedure:
2.5.1 Detection of terpenoids
Five ml of plant extract solution, 2 ml of chloroform, and 3 ml of pure sulfuric acid were combined in small test tubes. The appearance of a reddish brown color refers to the presence terpenoids (Sheikh et al., 2013).
2.5.2 Test for alkaloids
HCl (1%) and 6 drops of Dragendroff reagent were added to the plant extract solution, the organic precipitate showed the existence of an alkaloid (Sheikh et al., 2013).
2.5.3 Phenolic compound test
A little plant extract solution was mixed with a few drops of a lead acetate solution. The presence of phenolic chemicals was shown by the formation of white precipitate (Vimalkumar et al., 2014).
2.5.4 Detection of flavonoid
Sodium hydroxide was applied a few drops at a time to the extract. Flavonoids are indicated by the creation of a bright yellow color that fades with the addition of a few drops of diluted acid (Vimalkumar et al., 2014).
2.5.5 Detection of saponins
Plant extract was diluted to a volume of 20 ml from one ml using distilled water, and the mixture was agitated in a graduated cylinder for 15 min. The presence of saponins detects by formation of one-centimeter-thick layer of foam (Rao., 2016).
2.5.6 Tannin detection test
When 1 ml of FeCl3 (1%) were added to the plant extract solution, the coloration changed to a greenish or blackish- blue color, which served as a sign that tannin was present (Sadeq et al., 2021).
2.6 Quantitative biofilm formation assay
Microtiter plate technique which was described by (Limban et al., 2011), used with slight modifications to evaluate ability of biofilm production by the identified A. baumannii isolates. Briefly, 15 µL of the overnight cultures of the desired bacteria were inoculated into wells of the microtiter plate (MTP, Citotest Labware, China) containing 200 µL of sterile Nutrient broth (NB, Neogen, USA) supplemented with 2% glucose. Wells containing NB only served as control. Then, the inoculated MTP was incubated for 24 h in static condition at 37 °C. Following the incubation period, wells were rinsed thrice with sterile phosphate buffer saline (PBS), planktonic broth culture were discarded, plates were dried at 50 °C for 20 min and then 200 µL of 1% crystal violet staining solution was added into each well and incubated at room temperature for 10 min. Wells of the MTP were rinsed thrice with PBS, wells dried and eluted with 95% ethanol solvent to quantify the produced biofilms by mean of Elisa (Epson, Biotek, UK) at a wavelength of 490 nm. Three biological samples were analysed. The two strongest biofilm producer’s isolates (and the most resistant to the studied antibiotics) were chosen for further experiments throughout the study.
2.7 Minimum inhibitory and sub-inhibitory concentrations
A modified Broth dilution protocol was deployed to find the MIC (Minimum inhibitory concentration) of H. triquetrifolium Turra ethanolic extract for ascertaining the antagonistic action against MDR A. baumannii isolates (Wiegand et al., 2008). Ten µL of stationary-phase A. baumannii cells equilibrated to OD550 0.5 inoculated to 100 µL NB containing a range of extract concentrations (1–30 mg ml−1) in a 96-wells polystyrene MTP. Aerobic, 37 °C and 24H was the culture conditions. No growth visibility at the lowest concentration was ascertained as the MIC. Further lower concentrations below the MICs were approved to the sub-inhibitory and using these conentrations, asessment of anti-virulence and anti-biofilm activities among the isolated A. baumannii strains were carried out. Biological triplicates of various timelines were deployed in calculations.
2.8 Sub-MIC (SIC) effect of Hypericum triquetrifolium Turra on the biofilm production by A. Baumannii isolates
PVC (Polyvinyl Chloride) biofilm assay was deployed to check the extracts’ effects on the biofilm formation. Newly made NB medium was used to re-suspend overnight cultures of A. baumannii in the varying levels of SICs of the plant extract and incubated in a static condition at 37 °C for 24 h. The liquid cultures in the plate wells were removed, and washing thrice with PBS buffer was carriedout in the wells. Crystal violet dye (1%) was sused for staining and subsequently washed with distilled water and ethanol was used for the dye solubilisation and for further measurement. Surface adhesion ability was carried out using an Elisa reader (Epson, by measuring the coloured suspension density at 490 nm (Ahmed and Salih, 2019). Biological triplicates of various timelines were deployed in calculations and the calculation of the standard error was finalized.
2.9 RNA extraction and quantification of QS and biofilm related genes
Real time PCR was used to evaluate the effect of ethanol extract of H. triquetrifolium Turra at SIC values on the expression levels of the QS (abaI, abaR), biofilm attachment protein (Bap) and outer membrane protein A (ompA) genes. Total RNA was extracted from both untreated bacteria which were used as control and bacteria exposed to the plant extract following guidance provided by the manufacturer (total RNA kit, Favorgen Biotech, Taiwan). cDNA was synthesized through reverse transcription of the isolated RNA using AddScript cDNA synthesis kit according to the manufacturer protocol (addbio,Koria). RT-PCR reactions were carried out utilizing RealQ Plus 2x Master Mix Green (Ampliqon,Denmark) in the PCRmax Eco 48 RT-PCR system. Primer sequences listed in Table 4 were used to analyze candidate genes by qPCR and the results were calculated using ΔΔCt method (Livak and Schmittgen, 2001).
2.10 Statistical analysis
The assay results were analyzed using GraphPad Prism 8.0 software. Two-way analysis of variance (ANOVA) method was used for multiple comparisons. Data presented as mean ± SE.
3 Results
Toxicity assessment of H. triquetrifolium Turra ethanol leaves extract on rat and determination of lethal dose was done prior to the experiment, single oral dosages of 0.5, 1, 2, 4, 4.5, and 5 g/kg of the leaves of H. triquetrifolium Turra were administered to the treated groups, and the toxicity was measured, the LD50 value was 4.95 g/kg. The results of phytochemical qualitative test for H. triquetrifolium Turra ethanol leaves extract revealed the presence of phenolic compound, tannins, alkaloids, and terpenoids, were identified.A total of fifty isolates of A. baumannii were collected from different clinical specimens (blood, cerebrospinal fluid (CSF), pus, sputum, and wound swab) of patients in different hospitals in Erbil city, Kurdistan Region, Iraq. Sputum was the most common source of infections 36 (72%) and minimum source of infection was revealed to be from pus specimens 1 (2%). Full results of isolates distribution are summarized in Table 1. In this study, resistance patterns of all isolates to different antibacterial agents, listed in Table 2, were investigated. According to the results, the isolates showed different resistance patterns, were 48 (96%) of the isolates resistant to Ciprofloxacin, followed by 44 (88%) were resistant to Trimethoprime/Sulfamethoxazole. The full results of antibiotic resistance patterns of all A. baumannii isolates are shown in Table 2. Based on the abilities of biofilm formation, the isolates were classified as weak, moderate, and strong biofilm producers. All the 50 non-duplicate isolates of A. baumannii were examined in 96-well MTPs and biofilm forming abilities of the isolates were quantified. The results showed that 40% of the isolates produced weak biofilm, 50% exhibited moderate biofilm, and strong biofilm produced by 4% of the isolates. The results were compared to control (Fig. 1). The MICs of H. triquetrifolium Turra ethanol extract were detected to select the SICs and evaluate their effect on the bacterial growth and inhibition of QS-regulated behaviours. The MICs of the extract against the clinical isolates of A. baumannii and the ATCC (19606) strain were determined and the SICs of the extract were used throughout the study. The range of the MICs was between (8–15 mg/ml) and the least recorded SIC was 5 mg/ml (Table 3). Two of the isolates which were the most antibiotic resistant and the strongest biofilm producers along with the ATCC strain of A. baumannii (19606) were treated with for SICs of the studied plant extract and then were assessed for the effects on biofilm formation in 96-well MTPs. The results of biofilm inhibition by ethanol leaves extract of H. triquetrifolium Turra depicted that this extract reduced biofilm production in both isolates and the ATCC strain (Fig. 2).
Type of specimen
No. of specimens
Percentage (%)
Blood
5
10%
CSF
1
2%
Pus
1
2%
Sputum
36
72 %
Wound swab
7
14%
Total
50
100%
Antibacterials
Resistant (no.) Percentage (%)
Amikacin
4
8%
Amoxilin/Clavulanic Acid
1
2%
Ampicilin
1
2%
Ampicilin/Sulbactum
1
2%
Azithromycin
1
2%
Aztreonam
11
22%
Cefazolin
2
4%
Cefepime
2
4%
Cefoxitin
1
2%
Ceftazidime
43
86%
Ceftriaxone
2
4%
Cefuroxime
3
6%
Ciprofloxacin
48
96%
Colistin
37
74%
Gentamicin
45
90%
Imipenem
41
82%
Levofloxacin
37
74%
Meropenem
43
86%
Netilmicin
18
36%
Piperacilin
41
82%
Pipracilin/Tazobactum
41
82%
Tazobactam
4
8%
Tetracyclin
34
68%
Trimethoprime/Sulfamethoxazole
44
88%
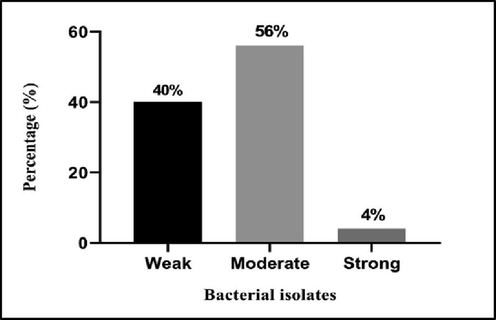
Biofilm formation ability of A. baumannii.
Bacterial isolates
MIC (mg/ml)
SUB-MIC (mg/ml)
AB-1
15
10
AB-2
8
5
ATCC
10
5
Gene
Primer Sequence (5′–3′)
Product size(bp)
Reference
Forward
Reverse
abaI
AAAGTTACCGCTACAGGG
CACGATGGGCACGAAA
435
(Dogan et al., 2019)
abaR
TCCTCGGGT CCCAATA
AAATCTACCGCATCAA
310
(Dogan et al., 2019)
Bap
AATGCACCGGTACTTGATCC
TATTGC CTGCAGGGTCAGTT
205
(Sajjadi et al., 2015)
ompA
ATGAAAAAGACAGCTATCGCGATTGCA
CACCAAAAGCACCAGCGCCCAGTTG
136
(Sajjadi et al., 2015)
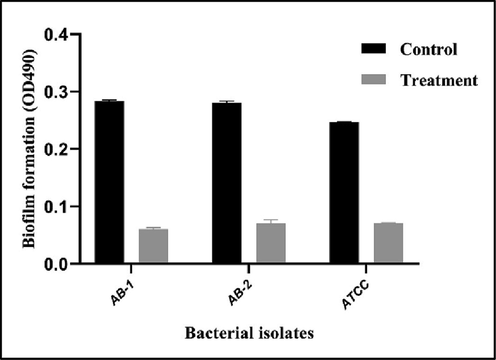
Reduction effects of SIC of H. triquetrifolium Turra ethanol extract on the quantitative measurement of A. baumannii biofilm by measuring absorbance at 490 nm. Data are presented as mean ± SE. All data are significant at P < 0.0001.
Results for the expression profile of QS genes, abal, abaR, and biofilm producer genes, Bap and OmpA, in same isolates of the previous experiment, when treated with SICs ethanol extract of H. triquetrifolium Turra showed sharped down-regulation of both QS genes comparing to their expression in untreated control group. On the other hand, the expression profile of biofilm producers detected a notable reduction in the expression of Bap gene whereas no change was observed for OmpA gene (Fig. 3).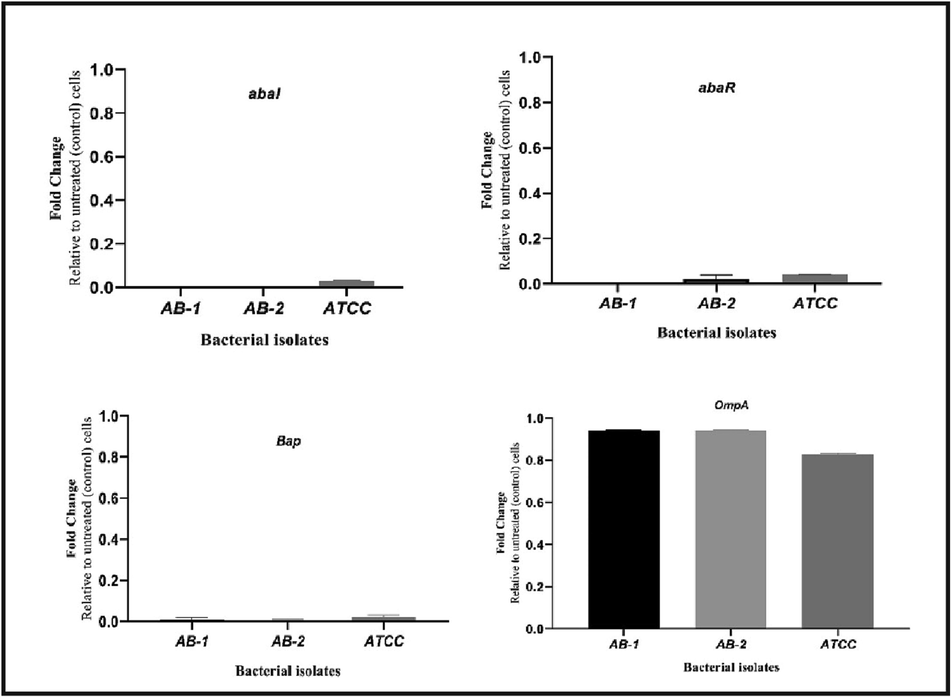
Quorum sensing and biofilm genes’ expression of A. baumannii isolates treated with SICs ethanol extract of H. triquetrifolium Turra. Transcriptional profiles were measured by RT-PCR.
4 Discussion
The emergence of pathogenic bacterial species which are antibiotic-resistant is currently one of the biggest problems facing the healthcare system. Multidrug-resistant (MDR) bacterial infections are becoming more prevalent and pose a severe threat to public health. These species were originally limited to the hospital setting, but now widespread everywhere (Vivas et al., 2019). A. baumannii is accounted to be one of the most common Gram negative multi-drug resistant bacteria. One crucial trait of pathogenic bacteria is the production of several virulence factors, which can be employed for defense and protection against the host immune system and the acquisition of significant resistance to various antimicrobials such as the production of QS and biofilms (Merghni et al., 2022). In addition to some bacterial species' capacity to evolve novel defense mechanisms via plasmids or integrons, other factors like poorly prepared food, proximity, crowdedness, and insufficient personal and general hygiene might be attributed to the spread of resistant bacteria (Nogbou et al., 2022). Medicinal plants usage has been taken in considerations by researchers to treat a variety of disorders today. In this sense, several disorders and diseases can be treated with the help of particular medicinal plant components. Due to their significance bioactive chemicals with antimicrobial and anti-oxidant properties, phenolic compounds and flavonoids have always been studied for the promotion of human health and disease prevention (Abbasi et al., 2022). In the current study, phytochemical screening of H. triquetrifolium Turra crude ethanolic leaves extract elucidated the existence of some significant bioactive compounds including terpenoid, alkaloid, phenol, flavonoid and tannin, however saponin was not detected in our studied plant’s extract, our results agreed with several other research in demonstrating the existence of many secondary metabolites which have been known for their antibacterial, antifungal activities and therapeutic activities (Rouis etal, 2013, Al-Sanfi et al., 2018). Prior to further extending our experiments, the toxicity of this plant was assessed and LD50 recorded to be 4.95 g/Kg. The toxicity scale of the H. triquetrifolium Turra extract is slightly toxic (Dawoud et al., 2015). Similar results regarding the toxicity was obtained by (Tumeh, 2015) when different extracts of H. triquetrifolium Turra induced apoptosis in several cancer and normal cell lines. It has been also proved that this plant exhibited significant protective properties against healthy cells from the toxic side effects of chemotherapeutic used in cancer treatment (Çetik Yıldız et al., 2018).
In the present study, highest percentage of A. baumannii were isolated from sputum specimens (Table 1). The isolates recorded highest resistance to Ciprofloxacin, Gentamicin, Trimethoprime/Sulfamethoxazole, Ceftazidime, Meropenem, Pipracilin/Tazobactum, and Piperacilin respectively and the isolates were resistant to the other antibiotics in different percentages (Table 2). This finding agreed with the results obtained by (Mirnejad R et al., 2013), this high resistant percentage of A. baumannii isolates to different antibiotics which are considered to be the most efficient drugs against this bacterium might be attributed to availability of the antimicrobials over the counter, without a prescription, and through unregulated supplier chains which encourages their misuse. Poverty and low educational levels have a significant root cause of antimicrobial misusage in developing countries, and non-compliance with antimicrobial use has various effects on emergence of resistant bacteria (Nogbou et al., 2022). However, this is not the case within low socioeconomical families only, even among the wealthy’s, some patients miss doses unintentionally or on purpose, especially when signs and symptoms start to fade after an initial positive therapeutic response (Hart & Kariuki., 1993). Thus, treating of A. baumannii infections became a critical therapeutic problem worldwide and seeking for new alternative treatments is urgent.Instead of using conventional bactericidal or bacteriostatic drugs, new approaches are progressively becoming evident which seek for the impact of plants natural materials on the pathogenicity of bacterial infections. Investigating the impact of natural plant metabolites on gene expression that are responsible for the new bacterial defensive methods is important to counteract bacterial resistance to traditional treatments.
Quorum sensing has been defined to be the mechanism by which bacteria organize their biological activities including motility, conjugation, biofilm formation, production of virulence factors, and pathogenic processes. Biofilm production enables A. baumannii to colonize different surfaces including biotic and abiotic subjects (Papenfort & Bassler., 2016).
In the present study we characterized the biofilm formation ability of all isolates (Fig. 1), we found that 4% of the isolates exhibited strong biofilm production capability and those were among the most antibiotic resistant isolates. However, more than half of the isolates were moderate biofilm producers. As expected, robust biofilm producer isolates exhibit strongest antibiotic resistance and were isolated from sputum specimens. These findings were in agreement with the results obtained by (Zhang et al., 2016), where isolates from sputum revealed strong biofilm development and the potential biofilm formation was strongly contributed to their pathogenicity and survival. The exceptional survival proprieties and high resistance to antibiotics are closely related to biofilm production, hence, antibiotics might be ineffective when used directly for bacterial treatment because they can create biofilms under stressful conditions (Dogan et al., 2019). Thus in seeking for innovative alternate curing strategy, it is believed that interrupting bacterial cell–cell communication without inducing stress, leave them more vulnerable and harmful. By using quorum sensing inhibitors (QSIs), this would limit the release of virulence factors and development of biofilm production by targeting the expression of those genes which are responsible for QS and biofilm production. For this purpose, the antimicrobial activity of H. triquetrifolium Turra ethanol leaves extract was assesed for their ability to interfer with QS system and biofilm producer genes. Two of the isolates which were the most antibiotic resistant and the strongest biofilm producers and the ATCC strain of A. baumannii (19606) were chosen for further experiments. MICs and SICs antimicrobial activity of ethanol extract of H. triquetrifolium Turra were measured against MDR A. baumannii isolates and the ATCC strain (Table 3).
To further investigate and address the QS and biofilm inhibitory effects, bacterial isolated were treated with SICs of ethanol extract of H. triquetrifolium Turra. Our results revealed significant attenuation in biofilm productions by the treated A. baumannii isolates comparing to untreated group (Fig. 2). Chemical compositions and antibacterial activity of different species of Hypericum were illustrated in different studies (Kizil et al., 2008, (Sajjadi et al., 2015). It has been reported previously that the essential oil obtained from H. triquetrifolium Turra has antibacterial effects against Bacillus brevis, Bacillus cereus, Escherichia coli PBR 322, Escherichia coli PUC 9, Pseudomonas aeruginosa and Staphylococcus aureus (Rouis et al., 2013). Several studies reported that ethanolic extract of H. perforatum showed efficient antimicrobial activities against most of the oral pathogens and this was attributed to that most of the components of this extract were found to be phenol, flavonoids, hypericins and hyperforins, which have been known for their antibacterial activities (Rouis et al., 2013); (Süntar et al., 2016).
The ability of A. baumannii’s biofilm formation and related genes have been studied by many researchers (Tang et al., 2020); (Amin et al., 2019). It has been known that QS system controls virulence factors production and formation of biofilms (Papenfort & Bassler., 2016). Mutation in abaI gene resulted in reduced QS and in turn biofilm production (Yang et al., 2019). The QS system and biofilm formation in A. baumannii have been recorded to include abaI, abaR, Bap and OmpA genes respectively (Niu et al., 2008). Transcriptional profiles expression of QS and biofilm genes of A. baumannii isolates when treated with SICs ethanol extract of H. triquetrifolium Turra revealed that the studied plant’s extract down regulated quorum sensing genes (abaI and abaR) expression dramatically which are considered criticals for cell–cell communications and further regulates biofilm production, its been obsereved that QS in A. baumannii could be inhibited with different plant extracts (Saipriya et al., 2020); (Subhadra et al., 2016). It has been reported that suppresing the expression of abaI was required for biofilm development (Niu et al., 2008). Same group also found that AbaR is required for abaI promotor activation. It has been shown that applying natural plant products including different species of Hypericum interfered with AHL receptors, resulting in imparing the QS signals (Dogan et al., 2019); (Roy et al., 2022). Anti-biofilm effects of H. perforatum against different oral bacteria was assessed and found to impair biofilm formation (Suntar et al., 2016). We further assessed the effects of abaI and abaR down regulations on biofilm formation genes, our SICs plant extract exhibited drastic down regulation expression on Bap, which was found to be harbored and expressed among strong biofilm produces and that Bap represents the key protein factors for biofilm initiation attachment and maturation of the biofilm by A. baumannii, in that it affects both biofilm thickness and bio-volum. This results came in line with a recent study (Amin et al., 2016). On the other hand, OmpA gene expression didn’t show pronounced change when bacterial isolates were treated with the SICs extract of H. triquetrifolium Turra. However, it has been demonstrated by Gaddy and colleagues that OmpA has partial role in the development of strong biofilm and that its required mostly for biofilm formation on abiotic surfaces (Gaddy et al., 2016). Another study revealed that Bap and OmpA genes influence biofilm formation and antibiotic resistance patterns, however same study did not provide further verification to ascertain that OmpA gene is pivotal in biofilm production (Yang et al., 2016). This finding suggests that in spite of OmpA expression, this could not result in a robust biofilm production which means biofilm might be formed independently of OmpA. These effects could be attributed to the presence of plenty of natural components within the extracts which can exhibit different activities against bacterial species including antibacterial and anti-biofilm productions.
To the best of our knowledge, the present study is the first work reporting the toxicity, antimicrobial, anti-QS and anti-biofilm activities of H. triquetrifolium Turra against A. baumannii. We concluded that this plant excrets slight toxicity against experimental rats. This plant showed promissing activities against the bacterial isolates in QS system and biofilm formation due to the valuable bioactive components. The antibiotic resistant bacteria continue to be the main health problem globally. As bacteria progressively generate recsistace against different bactericidal and bacteristaic antibiotics, thus innovating alternative materials apart from the conventional drugs was made. Results of biofilm inhibition assay showed that H. triquetrifolium Turra is capable to exhibit anti-biofilm activity against A. baumannii. Furthermore, we concluded that QS and biofilm producing genes were down regulated by the studied plant. Future analysis could be done in order to seek for the most potent components. Thus H. triquetrifoliumTurracould be used as a potential antimicrobial drugs.
5 Authors’ contributions
AA & JR designed research; AA & JR conducted research, AA analyzed data; TM, AA, AG and JR wrote and edited the paper; AA, JR, AG, TM had primary responsibility for final content. All authors read and approved the final manuscript.
Institutional Review Board Statement: The study was carried out in accordance with the Declaration of Helsinki and approved by the Ethics Committee at the Pharmacy College-Hawler Medical University (number: 20202508–209 HMU.PH.EC).
Funding
The current research received no funding.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Loranthus europaeus Jacq. Uçucu Yağ ve Özütünün Fitokimyasal Analizi ve Acinetobacter baumannii, Staphylococcus aureus ve Pseudomonas aeruginosa Üzerine Antimikrobiyal Etkisi. Kafkas Universitesi Veteriner Fakultesi Dergisi 2022
- [CrossRef] [Google Scholar]
- Quercus infectoria gall extracts reduce quorum sensing-controlled virulence factors production and biofilm formation in Pseudomonas aeruginosa recovered from burn wounds. BMC Complement. Altern. Med.. 2019;19:177.
- [CrossRef] [Google Scholar]
- Chemical constituents and pharmacological effects of Hypericum triquetrifolium. Indo American Journal of Pharmaceutical Sciences. 2018;5:1757-1765.
- [Google Scholar]
- Association Between Biofilm Formation, Structure, and the Expression Levels of Genes Related to biofilm formation and Biofilm-Specific Resistance of Acinetobacter baumannii Strains Isolated from Burn Infection in Ahvaz. Iran. Infect Drug Resist. 2019;12:3867-3881.
- [CrossRef] [Google Scholar]
- Asan, H.S. 2021. Phenolic compound contents of hypericum species from Turkey. In: Propagation and genetic manipulation of plants. Springer. Pp. 43-68.
- Çetik Yıldız, S., Keskin, C., Şahintürk, V. et al. Cardioprotective effects of Hypericum triquetrifolium Turra. against cyclophosphamide related cardiotoxicity in rats. Journal of Research in Pharmacy 2018; 22: 46-58. DOI: 10.12991/jrp.2018.77.
- Hypericum triquetrifolium and H. neurocalycinum as Sources of Antioxidants and Multi-Target Bioactive Compounds: A Comprehensive Characterization Combining In Vitro Bioassays and Integrated NMR and LC-MS Characterization by Using a Multivariate Approach. Front. Pharmacol.. 2021;12:660735
- [Google Scholar]
- Dawoud, A.D.H. 2015. Acute Toxicity Studies of Ethanolic Extract of Eucalyptus Camaldulensis Dehnh Leaves. Journal of Network Communications and Emerging Technologies (JNCET) www jncet org 2.
- Anti-quorum sensing and anti-biofilm activities of Hypericum perforatum extracts against Pseudomonas aeruginosa. J Ethnopharmacol. 2019;235:293-300.
- [CrossRef] [Google Scholar]
- Acinetobacter baumannii Biofilm Formation and Its Role in Disease Pathogenesis: A Review. Infect Drug Resist. 2021;14:3711-3719.
- [CrossRef] [Google Scholar]
- In search for a synergistic combination against pandrug-resistant A. baumannii; methodological considerations. Infection 2022:1-13.
- [Google Scholar]
- Antioxidant Activities of Ethanol Extracts of Hypericum triquetrifolium. and Hypericum scabroides. Pharm. Biol.. 2008;46:231-242.
- [Google Scholar]
- Synthesis, spectroscopic properties and antipathogenic activity of new thiourea derivatives. Molecules. 2011;16:7593-7607.
- [CrossRef] [Google Scholar]
- Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods. 2001;25:402-408.
- [Google Scholar]
- An overview of Acinetobacter baumannii pathogenesis: Motility, adherence and biofilm formation. Microbiol. Res.. 2021;247:126722
- [Google Scholar]
- Is It Possible to Eradicate Carbapenem-Resistant Acinetobacter baumannii (CRAB) from Endemic Hospitals? Antibiotics. 2022;11:1015.
- [Google Scholar]
- Comparative Study of Antibacterial, Antibiofilm, Antiswarming and Antiquorum Sensing Activities of Origanum vulgare Essential Oil and Terpinene-4-ol against Pathogenic Bacteria. 2022;12
- [CrossRef]
- Antibiotic resistance and carriage class 1 and 2 integrons in clinical isolates of Acinetobacter baumannii from Tehran. Iran. Asian Pacific Journal of Tropical Biomedicine. 2013;3:140-145.
- [CrossRef] [Google Scholar]
- Isolation and characterization of an autoinducer synthase from Acinetobacter baumannii. J. Bacteriol.. 2008;190:3386-3392.
- [Google Scholar]
- Antibacterial Activities of Monsonia Angustifolia and Momordica Balsamina Linn Extracts against Carbapenem-Resistant Acinetobacter Baumannii. Plants. 2022;11:2374.
- [Google Scholar]
- Quorum sensing signal-response systems in Gram-negative bacteria. Nat Rev Microbiol. 2016;14:576-588.
- [CrossRef] [Google Scholar]
- Phytochemical screening, total phenolic, flavonoid contents, and antioxidant activities of four spices commonly used in Vietnamese traditional medicine. Mater. Today:. Proc.. 2022;56:A1-A5.
- [Google Scholar]
- Toxicity Analysis of different medicinal plant extracts in Swiss Albino Mice. Pharmacol. Toxicol.. 2014;1:1-6.
- [Google Scholar]
- Evaluation of the cytotoxic effect and antibacterial, antifungal, and antiviral activities of Hypericum triquetrifolium Turra essential oils from Tunisia. BMC Complement. Altern. Med.. 2013;13:1-13.
- [Google Scholar]
- Convergence of Biofilm Formation and Antibiotic Resistance in Acinetobacter baumannii Infection. Front Med (Lausanne). 2022;9:793615
- [CrossRef] [Google Scholar]
- Sadeq, O., Mechchate, H., Es-Safi, I. et al. 2021. Phytochemical Screening, Antioxidant and Antibacterial Activities of Pollen Extracts from Micromeria fruticosa, Achillea fragrantissima, and Phoenix dactylifera. Plants (Basel). 10. DOI: 10.3390/plants10040676.
- Quorum-sensing system in Acinetobacter baumannii: a potential target for new drug development. J. Appl. Microbiol.. 2020;128:15-27.
- [Google Scholar]
- Essential oil composition of Hypericum triquetrifolium Turra growing wild in Iran. Research in Pharmaceutical Sciences. 2015;10:9.
- [Google Scholar]
- Phytochemical screening to validate the ethnobotanical importance of root tubers of Dioscorea species of Meghalaya, North East India. Journal of Medicinal Plants. 2013;1:62-69.
- [Google Scholar]
- Shenkutie, A,M., Yao, M.Z., Siu, G.K.-h. et al. 2020. Biofilm-induced antibiotic resistance in clinical Acinetobacter baumannii isolates. Antibiotics 9, 817.
- Quorum sensing in Acinetobacter: with special emphasis on antibiotic resistance, biofilm formation and quorum quenching. AIMS Microbiol. 2016;2:27-41.
- [Google Scholar]
- Antimicrobial effect of the extracts from Hypericum perforatum against oral bacteria and biofilm formation. Pharm. Biol.. 2016;54:1065-1070.
- [Google Scholar]
- Contribution of the AbaI/AbaR Quorum Sensing System to Resistance and Virulence of Acinetobacter baumannii Clinical Strains. Infect Drug Resist. 2020;13:4273-4281.
- [CrossRef] [Google Scholar]
- Acute and subacute toxicity of Aspilia africana leaves. Afr. J. Tradit. Complement. Altern. Med.. 2007;4:127-134.
- [Google Scholar]
- Townsend, C., Guest, E. 1980. Flora of Iraq Ministry of Agriculture, Baghdad.
- Tumeh, A.M.A. 2015. In Vitro Evaluation of Apoptotic Induction of Hypericum triquetrifolium and Arum palaestinum Plant Extracts on Cancer Cell lines.
- Comparative preliminary phytochemical analysis of ethanolic extracts of leaves of Olea dioica Roxb., infected with the rust fungus Zaghouania oleae (EJ Butler) Cummins and non-infected plants. Journal of Pharmacognosy and phytochemistry 2014:3.
- [Google Scholar]
- Multidrug-resistant bacteria and alternative methods to control them: an overview. Microb. Drug Resist.. 2019;25:890-908.
- [Google Scholar]
- Agar and broth dilution methods to determine the minimal inhibitory concentration (MIC) of antimicrobial substances. Nat Protoc. 2008;3:163-175.
- [CrossRef] [Google Scholar]
- Defining effective durations of antibiotic therapy for community-acquired pneumonia and urinary tract infections in hospitalized children. Curr. Opin. Infect. Dis.. 2022;35:442-451.
- [Google Scholar]
- Biofilm Formation in Acinetobacter Baumannii: Genotype-Phenotype Correlation. Molecules 2019:24.
- [CrossRef] [Google Scholar]
- Biological features of biofilm-forming ability of Acinetobacter baumannii strains derived from 121 elderly patients with hospital-acquired pneumonia. Clin Exp Med. 2016;16:73-80.
- [CrossRef] [Google Scholar]
- Quorum Sensing Inhibition or Quenching in Acinetobacter baumannii: The Novel Therapeutic Strategies for New Drug Development. Front. Microbiol. 2021:12.
- [CrossRef] [Google Scholar]
- Insight into carbapenem resistance and virulence of Acinetobacter baumannii from a children’s medical centre in eastern China. Ann. Clin. Microbiol. Antimicrob.. 2022;21:47.
- [CrossRef] [Google Scholar]







