Translate this page into:
Padina boergessenii ameliorates carbon tetrachloride induced nephrotoxicity in Wistar rats
*Corresponding author. Tel.: +91 4144 243223; fax: +91 4144 243555 rajakarthis14@yahoo.co.in (S.T. Somasundaram)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.

Available online 11 March 2011
Abstract
This research investigates the ameliorative effect of brown alga Padina boergessenii against CCl4-induced nephrotoxicity in rats. Twenty-four Wistar albino rats were divided into four groups as follows: control group (saline), CCl4 group, CCl4 + P. boergessenii extract group and P. boergessenii extract alone group. At the end of the experimental period, the animals are sacrificed and tissue samples (kidney) were collected. CCl4 at a dose of 2 mL/kg, administered subcutaneously, produced significantly decreased activities of glutathione peroxidase (GSH-Px), superoxide dismutase (SOD), and catalase (CAT). Histopathological examinations showed massive centrilobular necrosis and fat accumulation in CCl4-induced animals. In the protective test, especially in animals pretreated with P. boergessenii (150 mg/kg) extract, there was a significant increase in antioxidant enzyme levels although there was no sign of abnormality in the kidney of rats pretreated with extract as evidenced by histopathological sections against CCl4-induced decrease of lipid peroxidation (TBARS). The ameliorative effect may be due to the presence of various bioactive compounds in alga. These results suggest that an extract of P. boergessenii is able to significantly alleviate the nephrotoxicity induced by CCl4 in Wistar rats.
Keywords
Padina boergessenii
CCl4
Nephrotoxicity
Antioxidant
1 Introduction
Carbon tetrachloride (CCl4), a lipid soluble, heavy, and non-flammable liquid is most widely used for experimental induction of hepato (Reinke et al., 1988; Salazar-Montes et al., 2000; Karthikeyan et al., 2010b) and nephrotoxicity (Sylvia Zhao et al., 1997). CCl4 is an organic compound widely used as a dry cleaning solvent until it was recognized as a potent carcinogen (Kovacic and Jacintho, 2001). The toxic effects of CCl4 on liver have been extensively studied and it has been attributed to the production of free radical metabolites, with the cytochrome P450s involved in the metabolism rapidly destroyed during the metabolic process. There is extensive evidence to implicate free radicals in the development of degenerative diseases and progressive decline in the immune system (Pike and Chandra, 1995). Additionally, CCl4 is classified as a probable human carcinogen, based on the development of hepatocellular carcinomas in rats, mice and hamsters (Salazar-Montes et al., 2000).
Acute and chronic renal damage are also very common pathophysiologic disturbances caused by CCl4 (Ogeturka et al., 2005). Administration of CCl4 causes an increase in lipid peroxidation products (Daniels et al., 1995; Abraham et al., 1999; Donder et al., 1999) and a decrease in the activity of enzymes protecting lipid peroxidation in the kidney. The trichloromethyl and trichloromethyl peroxy radicals are reported to enhance lipid peroxidation and protein oxidation, resulting in wide spread membrane damage and decrease in the activity of enzymes protecting lipid peroxidation in the kidney (Dogukan et al., 2003).
The anti-oxidant enzymes include superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GSH-Px) and indirectly glutathione reductase. The role of these enzymes is well known and has been investigated extensively both in vivo and in vitro in model systems. The nephrotoxic effect of CCl4 is associated with free radical formation (Feral et al., 2003) which leads to lipid peroxidation and breakdown of the membrane structure. Characteristic renal injuries from carbon tetrachloride exposure are nephritis, nephrosis, and renal failure. Kidney failure is one of the leading causes of death in CCl4 intoxication. The consequence of oxidative damage is serious, and in many cases apparent in increased activities of enzymes involved in oxygen detoxification. The negative effects of free radicals, oxidative stress may well be overcome by antioxidants (Larson, 1995).
Seaweeds received significant attention for their potential as natural antioxidants (Kumar et al., 2008; Monsuang et al., 2009). In specific, brown algae are rich sources of bioactive compounds like polyphenols, carotenoids and polysaccharides. Recently, we studied the hepatoprotective effect of P. boergesenii against CCl4 induced liver damage in Wistar rats (Karthikeyan et al., 2010b). In addition, we have evaluated the antioxidant activities of brown alga P. boergesenii with ferric nitrilotriacetate induced renal damage in circulation of rats (Karthikeyan et al., 2010a) in two different extracts. These results have encouraged us to pay more attention to this particular species of the family Phaeophyceae. The present study was designed to evaluate the putative antioxidant action of brown alga P. boergessenii extract in an experimental model of CCl4-induced nephrotoxicity in Wistar rats.
2 Materials and methods
2.1 Chemicals
Reduced glutathione (GSH), oxidized glutathione (GSSG), glutathione peroxidase, thiobarbituric acid (TBA), CCl4 and diethyl ether were purchased from Sigma–Aldrich. All other chemicals and solvents of analytical grade were obtained from S.D. Fine chemicals, Chennai, India.
2.2 Collection of seaweeds
In the present study the seaweed P. boergesenii belonging to the class Phaeophyceae, of the family Dictyotaceae, was collected from Vaan island, Gulf of Mannar Biosphere in the intertidal region (Lat 11°29′N; Long 79°46′E) during April and May 2007. The algal species were hand picked and washed thoroughly with seawater to remove all the impurities like sand particles and epiphytes. Morphologically distinct thallus of algae was placed separately in new polythene bags and these were kept in an icebox containing slush ice and transported to the laboratory. Finally, the seaweeds were rinsed carefully in freshwater and shade dried then the samples were pulverized into powder (size 2 mm) using a grinder before extraction. The brown alga P. boergesenii was identified by its taxonomical features.
2.3 Preparation of seaweed extract
In general, the principle of soxhlet extraction is an extraction method by using chemical solvents (Lim et al., 2002). The 5 g of P. boergesenii grinded powder was extracted with 100 mL of diethyl ether (non-polar solvent) using a soxhlet extractor for 8 h at 35 °C. The extraction was repeated many times to obtain a sizable quantity of extract. The extracts were then concentrated under reduced pressure and the resultant residues were stored in a dark at 4 °C until further use. For the investigation of in vivo study diethyl ether residues were dissolved in 0.3% carboxyl methyl cellulose as a vehicle.
2.4 Animal and housing
The male albino rats of the Wistar strain (4–6 weeks old), weighing 125–150 g, were obtained from Central Animal House, Faculty of Medicine, Rajah Muthiah Medical College and Hospital, Annamalai University, India. Animal experiments were approved by the Institutional Ethical Committee Regulations. The Central animal house registration number is 160/1999/CPCSEA. The rats were fed with pellet diet and water ad libitum. During experimentation they were housed in the respective polypropylene cages. The animals were maintained in a controlled environment under standard conditions of temperature and humidity with alternating 12 h light/dark cycles.
2.5 Experimental induction of nephrotoxicity
All the rats were allowed to acclimatize for 1 week prior to experimentation. The treatment regimen for brown alga P. boergesenii extract was based on the preliminary in vitro studies carried out in our laboratory (Karthikeyan et al., 2010b). CCl4 dose was selected according to Janbaz et al. (2004). A total of 24 rats were used the animals were randomized and had free access to standard laboratory feed. The rats are grouped into experimental and control rats (n = 6 in each group). Group I served as saline-treated negative control and was administered with saline (0.85% NaCl) alone. Group II subcutaneously injected with CCl4 (2 mL/kg body weight) every day for 1 month. Group III were pretreated with diethyl ether extract with dose of 150 mg/kg body weight orally with the help of gavage tube before 1 h of CCl4 administration. Group IV rats were orally administered with P. boergesenii diethyl ether extract (150 mg/kg body weight) alone for 1 month. At 24 h after the last treatment of extracts or pellet feed animals on the 30th day from groups II and III received an i.p. injection of CCl4 (2 mL/kg body weight). All 24 animals were sacrificed by cervical dislocation within a period of 1 and 12 h after treatment with CCl4 or saline.
2.6 Evaluation of kidney damage
Kidney of all rats was excised immediately after collection of the blood. The right and left tissue specimens were washed twice with a cold saline solution placed into glass bottles and labeled. The right kidneys were used for biochemical estimations and the left kidney tissue specimens were used for histopathological examination.
2.7 Preparation of tissue samples
The tissue sample (kidney) was rinsed in physiological saline, and homogenized. The homogenate (25%, w/v) was in ice-cold 0.25 M sucrose, then the homogenate was deproteinized with an equal volume of ice-cold (10%, w/v) trichloroacetic acid. The ensuing supernatant fraction was analyzed for enzymatic and non-enzymatic biochemical assays.
2.8 Vitamin E in kidney
The level of vitamin E were determined by the method of reduction in Fe3+ to Fe2+ by the tocopherols with the formation of a red complex of the Fe2+ with α,α′-dipyridyl (Emmerie and Engel, 1938a,b). The transmissions were measured by calorimeter and it is expressed in terms of μg/mg protein.
2.9 Estimation of thiobarbituric acid reactive substances (TBARS)
The level of TBARS was estimated in kidney by measuring malondialdehyde and TBARS reactivity with thiobarbituric acid (TBA) to generate a pink coloured chromophore, which was read at 535 nm (Nichans and Samuelson, 1972). The transmissions were measured by calorimeter and it is expressed in terms of mM/100 g wet tissue.
2.10 Estimation of reduced glutathione
Reduced glutathione was estimated in kidney by the method of Ellman (1959). The yellow colour developed was read at 412 nm. A series of standards were treated in a similar manner along with a blank containing 3.5 mL of buffer. The amount of glutathione was expressed as mg/100 g tissue.
2.11 Superoxide dismutase
Superoxide dismutase (SOD) activity was determined by the method of reaction between superoxide radicals with nitroblue tetrazolium (NBT) in the presence of NADH and produced formazan blue (Kakkar et al., 1984). SOD removes the superoxide radicals and inhibits the formation of formazan blue. The intensity of colour is inversely proportional to the activity of the enzyme. One unit of the enzyme activity is defined as the enzyme reaction which gave 50% inhibition of NBT reduction in 1 min under the assay conditions and expressed as specific activity in units/mg protein.
2.12 Estimation of catalase
The catalase (CAT) activities were determined by the method of conversion of dichromate in acetic acid to perchromic acid and then to chromic acetate in the presence of H2O2 when subjected to heat (Claiborne, 1985). Thus formed chromic acetate was measured at 620 nm and the activities were expressed as μM of H2O2 consumed units/mL and in liver as units/mg protein.
2.13 Estimation of glutathione peroxidase (GSH-Px)
Glutathione peroxidase was estimated by the reaction of the sample with hydrogen peroxide in the presence of GSH for a specified time period. Then the remaining GSH were measured by the method of Rotruck et al. (1973). The activities were expressed as μg of GSH consumed units/mL and in liver as units/mg protein.
2.14 Histopathological examination
Dissected kidneys were fixed in 10% neutral formalin solution for at least 24 h, then embedded in paraffin wax (melting point 56–58 °C), sectioned to 4 mm thickness and stained with Mayer’s hematoxylin and eosin (H&E) for histopathological examination under light microscope and sectioned (Drury and Wallington, 1980). All sections were evaluated for the degree of tubular and glomerular injury, vascular congestion, interstitial mononuclear inflammatory cell infiltration and fibrosis by an experienced observer.
2.15 Statistical analysis
The results were analyzed among the experimental group of animals and statistical significance (∗p < 0.05, ∗∗p < 0.01) between controls and treated groups were evaluated using multivariate analysis of variance (ANOVA) followed by Duncan’s multiple range test.
3 Results and discussion
The aim of the present study was to investigate the protective effects of brown alga P. boergessenii against carbon tetrachloride induced nephrotoxicity in Wistar rats. In CCl4 toxicity, the rat’s kidney weight was increased during oxidative damage as compared to the control group. The rats pretreated with extract (150 mg/kg body weight) almost restored the kidney weight against toxicant-induced toxicity when compared to the control group (Table 1). Treatment of rats with extract alone (150 mg/kg body weight) did not show any significant changes. The ameliorative effect of P. boergessenii against CCl4 was studied by the following antioxidant assays. The treatment with P. boergessenii extract (150 mg/kg bw) was started at 1 h interval before CCl4 (2 mL/kg body weight) administration for 1 month. Each value represents the mean value ± SEM of six animals.
Groups
Kidney weight (g/rat)
TBARS (mM/100 g tissue)
Reduced glutathione (mM/100 g tissue)
Vitamin-E (mg/dL)
Control
2.08 ± 0.23
1.45 ± 0.21
117.75 ± 0.18
1.33 ± 0.09
CCl4
2.86 ± 0.20*
4.3 ± 0.22*
81.033 ± 0.69*
1.46 ± 0.09*
CCl4 + extract
2.53 ± 0.19**
2.7 ± 0.32**
101.66 ± 0.14**
0.67 ± 0.04**
Extract
2.11 ± 0.23
1.4 ± 0.22
127.03 ± 0.32
1.14 ± 0.08
3.1 Effect of vitamin-E levels in P. boergessenii extract on CCl4-induced nephrotoxicity
Table 1 shows the level of vitamin-E in kidney. There was a significant decrease (p < 0.01) in the relative vitamin-E level in CCl4-induced rats, when compared to the control group. Pretreatment of rats with P. boergessenii extract (150 mg/kg) for 30 consecutive day’s significantly increased (p < 0.01) the vitamin-E level, when compared to rats induced with CCl4 alone. However, there is no significant effect on rats administered with extract alone.
3.2 Effects of P. boergesenii extract on TBARS
As shown in Table 1, is the level of TBARS in kidney. The level of TBARS was significantly increased (p < 0.01) in rats treated with CCl4, when compared to the control group. P. boergesenii extract at the dose of 150 mg/kg of bw per day significantly (p < 0.05) decreased the TBARS level as compared to CCl4 induced group. There is no significant change in the level of TBARS in rats treated with extract alone. The biochemical mechanisms involved in the development of CCl4 hepatotoxicity have long been investigated but rare are the data on nephrotoxicity. It is generally believed that lipid peroxidation caused by the carbon trichloromethyl radical, CCl3. TBARS, a stable metabolite of the free radical mediated lipid peroxidation cascade, is widely used as marker of lipid peroxidation. CCl4-induced nephrotoxicity resulted in a significant increase in TBARS level, which is similar with previous findings by Feral et al. (2003) and Ogeturka et al. (2005). The data of TBARS level demonstrate that oral administration of extracts to rats suppressed ischemia–reperfusion-induced renal lipid peroxidation and tissue injury probably via the alteration of cytochrome P-450.
3.3 Effects of P. boergesenii extract on reduced glutathione content
Glutathione (GSH) constitutes the first line of defense against free radicals (Ogeturka et al., 2005). As shown in Table 1, the CCl4-induced group showed a significant reduction in GSH content (p < 0.01) (Feral et al., 2003; Ogeturka et al., 2005). Pretreatment with extract at the dose of 150 mg/kg during CCl4 administration significantly (p < 0.05) protected the GSH depletion. P. boergesenii extract can prevent the renal GSH depletion and it is also an antioxidant that reacts with free radicals. The ameliorative effects may be due to its ability to enhance glutathione production by providing more substrate for reactive intermediates that promote detoxification mechanisms. The protective quality of extract on CCl4 poisoning may be due to its action on GSH. Recently, our data have shown the antioxidant properties of P. boergesenii extract against ferric nitrilotriacetate induced renal oxidative damage (Karthikeyan et al., 2010a) and CCl4 induced hepatotoxicity in Wistar rats (Karthikeyan et al., 2010b).
3.4 Effects of P. boergesenii extract on SOD, CAT and GSH-Px
Table 2 shows the effects of P. boergesenii extract on antioxidant enzymes in CCl4-induced nephrotoxicity in rats. After injection of CCl4, the activities of GSH-Px were significantly (p < 0.01) decreased as compared to the control group (Feral et al., 2003). Pre-treatment of animals with P. boergesenii extract (150 mg/kg) for 4 weeks significantly elevated the expression of GSH-Px as compared to the rats induced with CCl4 alone (p < 0.05). The treatment with P. boergessenii extract (150 mg/kg bw) was started at 1 h interval before CCl4 (2 mL/kg body weight) administration for 1 month. Each value represents the mean value ± SEM of six animals.
Groups
Superoxide dismutase (SOD) (U/mg protein)
Catalase (CAT) (U/mg protein)
Glutathione peroxidase (GSH-Px) (U/mg protein)
Histopathology inference
Control
16.21 ± 0.87
58.5 ± 1.17
7.03 ± 0.55
Normal
CCl4
8.50 ± 0.70*
31.08 ± 3.08#
5.21 ± 0.31*
Glomerular changes and tubular dilation
CCl4 + extract
13.07 ± 0.88**
48.48 ± 3.25##
6.02 ± 0.47**
Absence of tubular dilation
Extract
15.28 ± 0.85
57.7 ± 1.87
7.23 ± 0.32
Normal
Table 2 illustrates the superoxide dismutase and catalase status in control and experimental group of rats. However, pretreatment of rats with P. boergesenii extract could restore the antioxidant capacity exhausted by CCl4. Administration of CCl4 results in oxidative damage to the lipids and proteins of the kidney in rats (Abraham et al., 1999). In the present study, the defense antioxidant enzymes were significantly altered by induction of CCl4. There was a significant decrease in SOD, GSH-Px (p < 0.01) and CAT activities (p < 0.05) (Ozen et al., 2004). The significant increase in the level of antioxidant enzymes probably occurred as a result of the rats pretreated with extracts. As reported earlier by Rush et al. (1984), the decreased activity of GSH-Px is due to changes in the level of glutathione content. Similarly, the decreased activity of SOD in CCl4-induced rats may be due to the enhanced lipid peroxidation or inactivation of the antioxidative enzymes. Overall biochemical investigations, the pretreatment of P. boergesenii extract prior to CCl4 intoxication could not only renovate the changes in lipid peroxidation and reduce GSH content, but also protected the antioxidant machineries of the rat kidney as revealed from the enhanced levels of SOD, GSH-Px (p < 0.05) and CAT (p < 0.05) activities.
3.5 Histopathological changes
The tissue sections stained with H&E were examined for the extent of kidney damage. Morphologic damage was cortical and ranged from none (control and P. boergesenii extract group) to moderate (CCl4 + P. boergesenii extract group) to severe (CCl4 group). However, extensive cortical damage and focal glomerular necrosis were observed in CCl4 challenged group (Doi et al., 1991). In addition, there are morphological changes in cortical tubules, such as dilated and flattened epithelial cells (Fig. 1). Interstitial inflammatory cell infiltration was also seen in the CCl4 group (Feral et al., 2003; Ogeturka et al., 2005). Where as, the group of rats pretreated with extract reversed the CCl4-induced nephrotoxicity; it shows the glomeruli were normal and sparse tubular changes were observed. In this group, the affected tubules showed vacuolization, dilation, and a few cell detachments.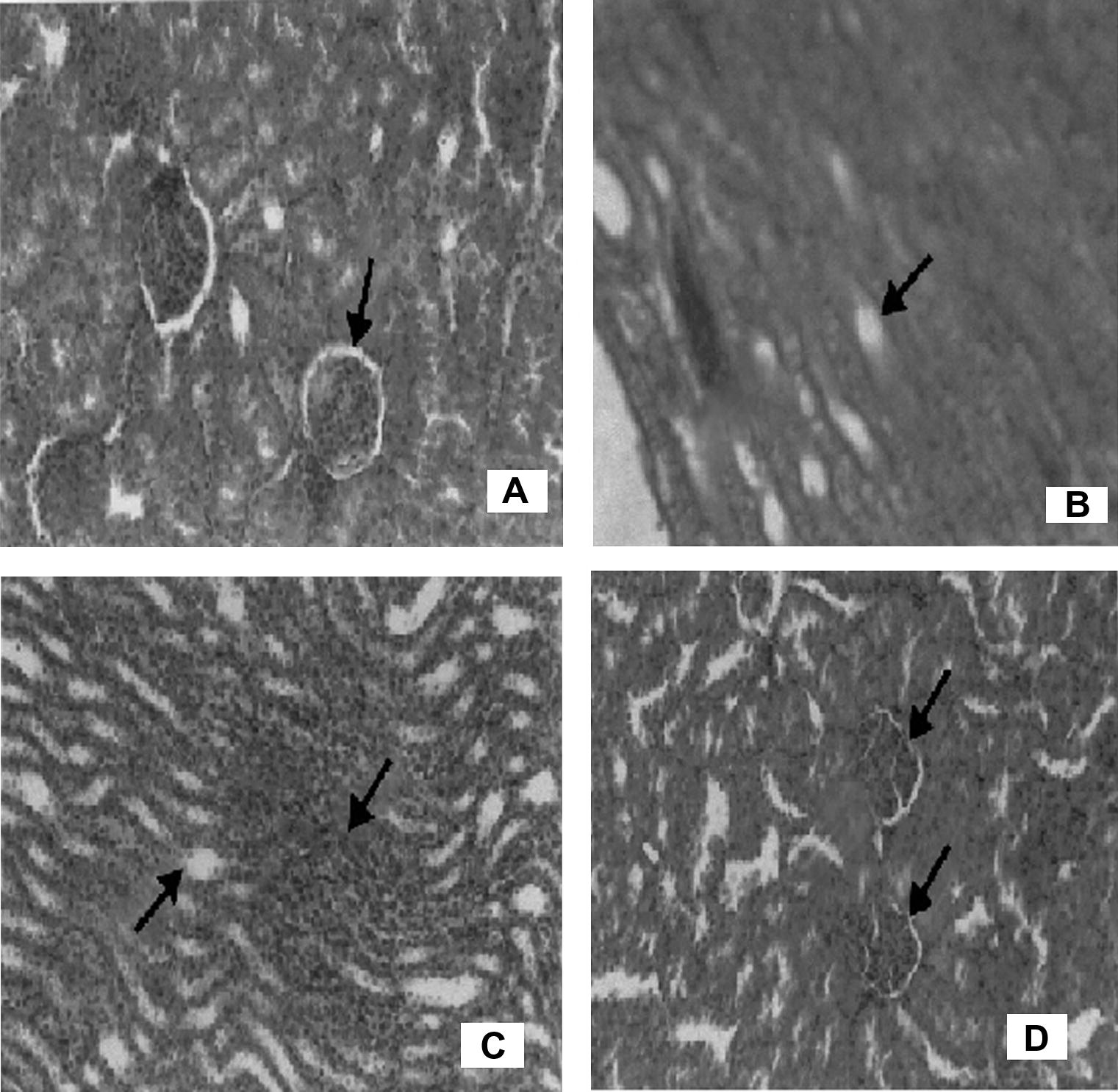
Histopathology of kidney. (A) Normal glomerulli. (B) CCl4 toxicity shows focal hyalincast. (C) Extract treated group show hemorrhage and cloudy swelling. (D) Extract alone show normal glomerulli and tubules.
Results suggest that the extract possesses protective action against CCl4 induced nephrotoxicity. This has been confirmed by antioxidant assays and histopathological observations. In our earlier report, the protective nature of P. boergessenii was confirmed by damage in the structural integrity of cell membrane in liver and leakage of transaminases from the cytoplasm into the blood circulation (Karthikeyan et al., 2007).
In conclusion, P. boergesenii extract effectively protect the kidney from oxidative damage against CCl4 through free radical scavenging and avoiding the oxidation of detoxifying enzymes. Moreover, not only CCl4-induced hepatotoxicity in our earlier findings (Karthikeyan et al., 2010b) but also nephrotoxicity was effectively alleviated by the P. boergesenii-pretreatment showed in these study experiments. Therefore, the brown alga P. boergesenii is beneficial in reducing free radical damage. While it is unclear to what extent each mechanism may contribute, all these reactions may provide a fundamental protective milieu for kidney in coping against CCl4 induced nephrotoxicity, by favoring the removal of oxidants as well as by increasing the amount of anti-oxidants from P. boergessenii extract. At this stage we identified and characterized the presence of polyphenols in P. boergessenii (data not shown) and still the investigations are underway for structural elucidation of bioactive compounds.
In summary, the results of this study suggest that P. boergesenii could prevent renal damage by improving the lipid peroxidation products through the scavenging activity of free radicals induced by CCl4. Further, the improvement of antioxidant enzymes may be another possible mechanism of action of brown alga P. boergesenii.
Acknowledgements
We are grateful to Prof. Venugopal P. Menon, Director of Research, Annamalai University for purveying this opportunity to do this research work. The authors wish to thank Department of Science and Technology (DST), India for the financial support. Finally, we would like to thank Dr. P. Subramanian, Reader and Dr. T. Manivasagam, Lecturer, Department of Biochemistry and Biotechnology, Annamalai University for their valuable support in technical aspects.
References
- Oxidative damage to the lipids and proteins of the lungs, testis and kidney of rats during carbon tetrachloride intoxication. Clin. Chim. Acta. 1999;289:177-179.
- [Google Scholar]
- Catalase activity. In: Green Wald R.A., ed. CRC Hand book of Methods for Oxygen Radical Research. Boca Raton, FL: CRC Press; 1985. p. :283-284.
- [Google Scholar]
- Melatonin counteracts lipid peroxidation induced by carbon tetrachloride but does not restore glucose-6 phosphatase activity. J. Pineal Res.. 1995;19:1-6.
- [Google Scholar]
- Protective effect of interferon-alpha on carbon protective effect of interferon-alpha on carbon tetrachloride-induced nephrotoxicity. J. Nephrol.. 2003;16:81-84.
- [Google Scholar]
- Systemic histopathology of rats with CCl4-induced hepatic cirrhosis. Lab. Anim.. 1991;25:21-25.
- [Google Scholar]
- Investigation of antioxidant effect of melatonin against carbon tetrachloride toxicity in various tissues. Biomed. Res.. 1999;10:141-145.
- [Google Scholar]
- Carleton’s Histological Technique. New York: Oxford University Press; 1980.
- Calorimetric determination of alpha tocopherol (vitamin E) Rev. Trav. Chim.. 1938;57:1351.
- [Google Scholar]
- Calorimetric determination of tocopherol. II: Absorption experiments. Nature. 1938;142:873.
- [Google Scholar]
- Carbon tetrachloride-induced nephrotoxicity and protective effect of betaine in Sprague–Dawley rats. Urology. 2003;62(2):353-356.
- [Google Scholar]
- Studies on the protective effects of caffeic acid and quercetin on chemical-induced hepatotoxicity in rodents. Photomed. J.. 2004;11:424-430.
- [Google Scholar]
- A modified spectrophotometric assay of superoxide dismutase. Indian J. Biochem. Biophys.. 1984;21:130-132.
- [Google Scholar]
- Antioxidant activity of brown alga Padina boergesinii against CCl4 induced liver fibrosis in rats. Seaweed. Res. Utiln.. 2007;30(Special issue):157-163.
- [Google Scholar]
- Rajamani Karthikeyan, Manivasagam, T., Anantharaman, P., Balasubramanian, T., Somasundaram, S.T., 2010a. Chemopreventive effect of Padina boergesenii extracts on ferric nitrilotriacetate (Fe-NTA)-induced oxidative damage in Wistar rats. J. Appl. Phycol. (August 18).
- Hepatoprotective activity of brown alga P. boergesenii against CCl4 induced oxidative damage in Wistar rats. Asian Pac. J. Trop. Med.. 2010;3:696-701.
- [Google Scholar]
- Mechanisms of carcinogenesis: focus on oxidative stress and electron transfer. Curr. Med. Chem.. 2001;8:773-796.
- [Google Scholar]
- Antioxidant potential of solvent extracts of Kappaphycus alvarezii (Doty) Doty – an edible seaweed. Food Chem.. 2008;107:289-295.
- [Google Scholar]
- Plant defenses against oxidative stress. Arch. Insect Biochem. Physiol.. 1995;29:175-186.
- [Google Scholar]
- Evaluation of antioxidative activity of extracts from a brown seaweed, Sargassum siliquastrum. J. Agric. Food. Chem.. 2002;50(13):3862-3866.
- [Google Scholar]
- Antioxidant activities of four edible seaweeds from the southern coast of Thailand. Plant Foods Hum. Nutr.. 2009;64:218-223.
- [Google Scholar]
- Formation of malondialdehyde from phospholipid arachidonate during microsomal lipid peroxidation. Eur. J. Biochem.. 1972;6:126-130.
- [Google Scholar]
- Caffeic acid phenethyl ester protects kidneys against carbon tetrachloride toxicity in rats. J. Ethnopharmacol.. 2005;97:273-280.
- [Google Scholar]
- Role of caffeic acid phenethyl ester, an active component of propolis, against cisplatin-induced nephrotoxicity in rats. J. Appl. Toxicol.. 2004;24:27-35.
- [Google Scholar]
- Effect of vitamin and trace element supplementation on immune indices in healthy elderly. Int. J. Vitam. Nutr. Res.. 1995;65:117-120.
- [Google Scholar]
- Ethanol feeding stimulates trichloromethyl radical formation from carbon tetrachloride in liver. Xenobiotica. 1988;18:1311-1318.
- [Google Scholar]
- Selenium: biochemical role as a component of glutathione peroxidase. Science. 1973;179:588-590.
- [Google Scholar]
- Chemically induced nephrotoxicity: role of metabolic activation. Crit. Rev. Toxicol.. 1984;13:99-160.
- [Google Scholar]
- Differential gene expression of pro-inflammatory and anti-inflammatory cytokines in acute and chronic liver injury. Hepatol. Res.. 2000;16:181-194.
- [Google Scholar]
- The prevention of ferric nitrilotriacetate-induced nephro- and hepatotoxicity by methylenedioxybenzene antioxidants. Chem. Biol. Interact.. 1997;108:107-118.
- [Google Scholar]







